
- •Contents
- •Preface
- •Contributors
- •1 Vessels
- •1.1 Aorta, Vena Cava, and Peripheral Vessels
- •Aorta, Arteries
- •Anomalies and Variant Positions
- •Dilatation
- •Stenosis
- •Wall Thickening
- •Intraluminal Mass
- •Perivascular Mass
- •Vena Cava, Veins
- •Anomalies
- •Dilatation
- •Intraluminal Mass
- •Compression, Infiltration
- •1.2 Portal Vein and Its Tributaries
- •Enlarged Lumen Diameter
- •Portal Hypertension
- •Intraluminal Mass
- •Thrombosis
- •Tumor
- •2 Liver
- •Enlarged Liver
- •Small Liver
- •Homogeneous Hypoechoic Texture
- •Homogeneous Hyperechoic Texture
- •Regionally Inhomogeneous Texture
- •Diffuse Inhomogeneous Texture
- •Anechoic Masses
- •Hypoechoic Masses
- •Isoechoic Masses
- •Hyperechoic Masses
- •Echogenic Masses
- •Irregular Masses
- •Differential Diagnosis of Focal Lesions
- •Diagnostic Methods
- •Suspected Diagnosis
- •3 Biliary Tree and Gallbladder
- •3.1 Biliary Tree
- •Thickening of the Bile Duct Wall
- •Localized and Diffuse
- •Bile Duct Rarefaction
- •Localized and Diffuse
- •Bile Duct Dilatation and Intraductal Pressure
- •Intrahepatic
- •Hilar and Prepancreatic
- •Intrapancreatic
- •Papillary
- •Abnormal Intraluminal Bile Duct Findings
- •Foreign Body
- •The Seven Most Important Questions
- •3.2 Gallbladder
- •Changes in Size
- •Large Gallbladder
- •Small/Missing Gallbladder
- •Wall Changes
- •General Hypoechogenicity
- •General Hyperechogenicity
- •General Tumor
- •Focal Tumor
- •Intraluminal Changes
- •Hyperechoic
- •Hypoechoic
- •Nonvisualized Gallbladder
- •Missing Gallbladder
- •Obscured Gallbladder
- •4 Pancreas
- •Diffuse Pancreatic Change
- •Large Pancreas
- •Small Pancreas
- •Hypoechoic Texture
- •Hyperechoic Texture
- •Focal Changes
- •Anechoic Lesion
- •Hypoechoic Lesion
- •Isoechoic Lesion
- •Hyperechoic Lesion
- •Irregular (Complex Structured) Lesion
- •Dilatation of the Pancreatic Duct
- •Marginal/Mild Dilatation
- •Marked Dilatation
- •5 Spleen
- •Nonfocal Changes of the Spleen
- •Diffuse Parenchymal Changes
- •Large Spleen
- •Small Spleen
- •Focal Changes of the Spleen
- •Anechoic Mass
- •Hypoechoic Mass
- •Hyperechoic Mass
- •Splenic Calcification
- •6 Lymph Nodes
- •Peripheral Lymph Nodes
- •Head/Neck
- •Extremities (Axilla, Groin)
- •Abdominal Lymph Nodes
- •Porta Hepatis
- •Splenic Hilum
- •Mesentery (Celiac, Upper and Lower Mesenteric Station)
- •Stomach
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •Small/Large Intestine
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •8 Peritoneal Cavity
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Wall Structures
- •Smooth Margin
- •Irregular Margin
- •Intragastric Processes
- •Intraintestinal Processes
- •9 Kidneys
- •Anomalies, Malformations
- •Aplasia, Hypoplasia
- •Cystic Malformation
- •Anomalies of Number, Position, or Rotation
- •Fusion Anomaly
- •Anomalies of the Renal Calices
- •Vascular Anomaly
- •Diffuse Changes
- •Large Kidneys
- •Small Kidneys
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Irregular Structure
- •Circumscribed Changes
- •Anechoic Structure
- •Hypoechoic or Isoechoic Structure
- •Complex Structure
- •Hyperechoic Structure
- •10 Adrenal Glands
- •Enlargement
- •Anechoic Structure
- •Hypoechoic Structure
- •Complex Echo Structure
- •Hyperechoic Structure
- •11 Urinary Tract
- •Malformations
- •Duplication Anomalies
- •Dilatations and Stenoses
- •Dilated Renal Pelvis and Ureter
- •Anechoic
- •Hypoechoic
- •Hypoechoic
- •Hyperechoic
- •Large Bladder
- •Small Bladder
- •Altered Bladder Shape
- •Intracavitary Mass
- •Hypoechoic
- •Hyperechoic
- •Echogenic
- •Wall Changes
- •Diffuse Wall Thickening
- •Circumscribed Wall Thickening
- •Concavities and Convexities
- •12.1 The Prostate
- •Enlarged Prostate
- •Regular
- •Irregular
- •Small Prostate
- •Regular
- •Echogenic
- •Circumscribed Lesion
- •Anechoic
- •Hypoechoic
- •Echogenic
- •12.2 Seminal Vesicles
- •Diffuse Change
- •Hypoechoic
- •Circumscribed Change
- •Anechoic
- •Echogenic
- •Irregular
- •12.3 Testis, Epididymis
- •Diffuse Change
- •Enlargement
- •Decreased Size
- •Circumscribed Lesion
- •Anechoic or Hypoechoic
- •Irregular/Echogenic
- •Epididymal Lesion
- •Anechoic
- •Hypoechoic
- •Intrascrotal Mass
- •Anechoic or Hypoechoic
- •Echogenic
- •13 Female Genital Tract
- •Masses
- •Abnormalities of Size or Shape
- •Uterus
- •Abnormalities of Size or Shape
- •Myometrial Changes
- •Intracavitary Changes
- •Endometrial Changes
- •Fallopian Tubes
- •Hypoechoic Mass
- •Anechoic Cystic Mass
- •Solid Echogenic or Nonhomogeneous Mass
- •14 Thyroid Gland
- •Diffuse Changes
- •Enlarged Thyroid Gland
- •Small Thyroid Gland
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Circumscribed Changes
- •Anechoic
- •Hypoechoic
- •Isoechoic
- •Hyperechoic
- •Irregular
- •Differential Diagnosis of Hyperthyroidism
- •Types of Autonomy
- •15 Pleura and Chest Wall
- •Chest Wall
- •Masses
- •Parietal Pleura
- •Nodular Masses
- •Diffuse Pleural Thickening
- •Pleural Effusion
- •Anechoic Effusion
- •Echogenic Effusion
- •Complex Effusion
- •16 Lung
- •Masses
- •Anechoic Masses
- •Hypoechoic Masses
- •Complex Masses
- •Index
Intracavitary Changes
Female Genital Tract

Vagina
Uterus
Abnormalities of Size or Shape
Myometrial Changes
Intracavitary Changes
Endometrial Changes
Fallopian Tubes
Ovaries
Foreign Body (IUD)
Mucocele, Serometra, Pyometra, Hematometra
Pregnancy
Ectopic Pregnancy
Missed Abortion, Incomplete Abortion, Cervical Pregnancy
Endometrial Polyps, Cervical Polyps, Placental Polyps
Foreign Body (IUD) 

















































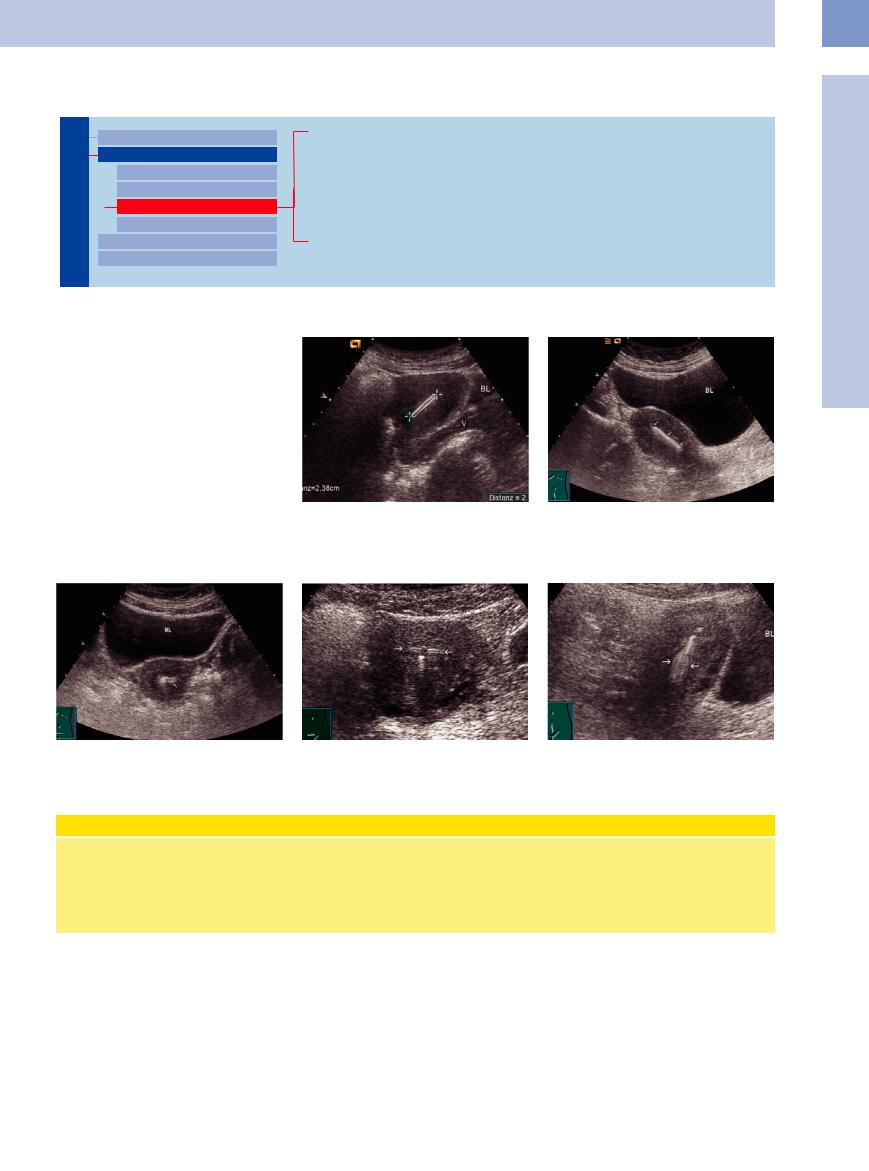
An intrauterine contraceptive device (IUD) appears as an intensely echogenic band with slightly indistinct margins at the center of the uterus. The shape and sharpness of the foreign body are variable, depending on the type of IUD (Figs. 13.33, 13.34, 13.35, 13.36, 13.37). The echogenicity changes with the angle of the scan.
Fig. 13.33 IUD (cursors): very echogenic structure in the |
Fig. 13.34 Uterus (sagittal scan) with a very echogenic |
uterine cavity. Correct placement is indicated if the dis- |
IUD (arrows) in the uterine cavity. |
tance between the endometrium and IUD is less than |
|
5 mm. UT = uterus; BL = bladder; V = vagina. |
|
Fig. 13.35 Uterus (transverse scan) with an echogenic IUD |
Fig. 13.36 T-shaped copper IUD, transverse scan. Arrows: |
Fig. 13.37 T-shaped copper IUD, sagittal scan. Blurring |
(arrows) in the fundus. BL = bladder. |
horizontal parts. |
vertical part (arrows). BL = bladder. |
Contraceptive Protection |
|
|
Today the IUD is the second most widely |
should be checked regularly to ensure con- |
be scheduled for midcycle when the uterus |
used contraceptive in the world. Before an |
traceptive protection. The distance of the |
has a thick, proliferative endometrium. |
IUD is inserted, transvaginal sonography |
IUD from the fundus is normally 1 cm and |
The incidence of ectopic pregnancy is 4–5 |
should be performed to measure the size |
should not exceed 2 cm. In a uterus with |
times higher in IUD wearers. |
of the uterine cavity and exclude an ob- |
myomas, it is difficult to confirm correct |
|
structing myoma. The position of the IUD |
IUD placement. The examination should |
|
13
Uterus
451
13
Female Genital Tract
Mucocele, Serometra, Pyometra, Hematometra
Pyometra, Hematometra
Mucocele. A mucocele is a collection of secretions retained in the uterine cavity, especially around menopause. The cause of the obstructed drainage may be a carcinoma.
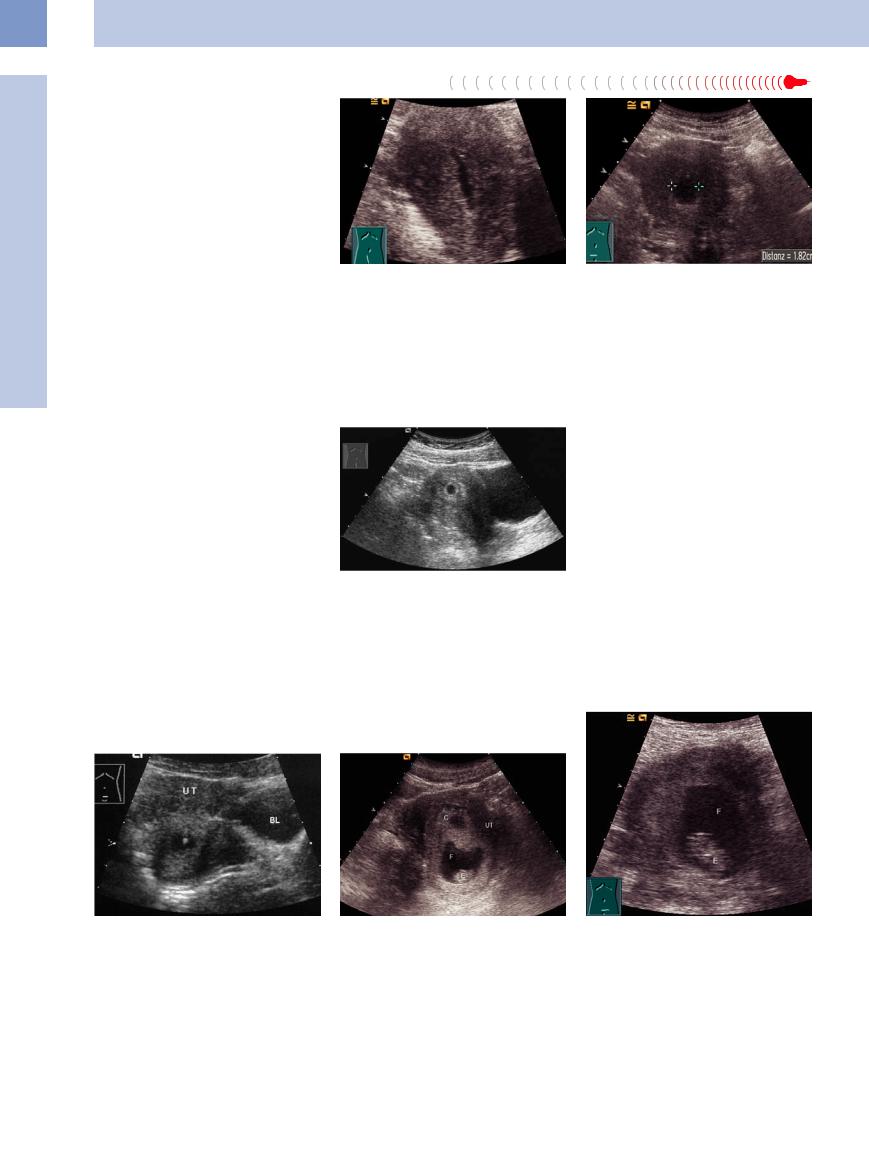
Serometra. This refers to an excessive accumulation of endometrial discharge due to intermittent cervical obstruction. The discharge may have a malignant or inflammatory cause (Fig.13.38, Fig.13.39).
Pyometra. Pyometra occurs as a complication of endometrial carcinoma owing to stenosis of the cervical canal. Its sonographic hallmark is a dilated, hypoechoic uterine cavity. Underlying endometrial carcinoma is present in more than 50% of cases.
Fig. 13.38 Serometra: fluid collection (hypoechoic mass) in the uterine cavity; sagittal scan.
Fig. 13.39 Serometra: fluid collection (cursors: hypoechoic mass); transverse scan.
Pregnancy






















































Pregnancy should be considered in patients undergoing ultrasound for lower abdominal pain or unexplained vomiting, and an ectopic pregnancy should be excluded.
In an intrauterine pregnancy, the normal anechoic chorionic cavity can be seen at an eccentric location in the uterus as early as 2 weeks’ gestational age (Fig.13.40). Its size increases by approximately 1 mm/day. By the fifth week, a ringlike structure can be seen in the chorionic cavity, representing the secondary yolk sac. This structure is of embryonic origin and affords proof of intrauterine pregnancy (Fig.13.41). The normal average diameter of the chorionic cavity is 5.5 cm (3–7 cm). Deviations from the norm signify a developmental anomaly, and karyotyping should be performed. The chorionic cavity is surrounded
by the hyperechoic chorion, which displays a typical ringlike vascular pattern. In later stages, the gestational sac develops from the secondary yolk sac and the chorionic cavity disappears (Fig.13.42).
Fig. 13.41 Early pregnancy with a chorionic cavity and |
Fig. 13.42 Early pregnancy; C = chorionic cavity, F = am- |
hyperechoic conceptus. UT = uterus; BL = bladder. |
nionic sac; E = embryo; UT = uterus. |
Fig. 13.40 Anechoic chorionic cavity in the third week of pregnancy, surrounded by the hyperechoic chorion.
As gestation progresses, the product of conception becomes visible and embryofetal details can be defined and evaluated (Figs. 13.43, 13.44, 13.45, 13.46).
Fig. 13.43 Intact pregnancy in the 10th week after spontaneous abortion of one twin; real-time ultrasound demonstrated the heartbeats. F = amnionic sac; E = embryonic area.
452
13
Uterus
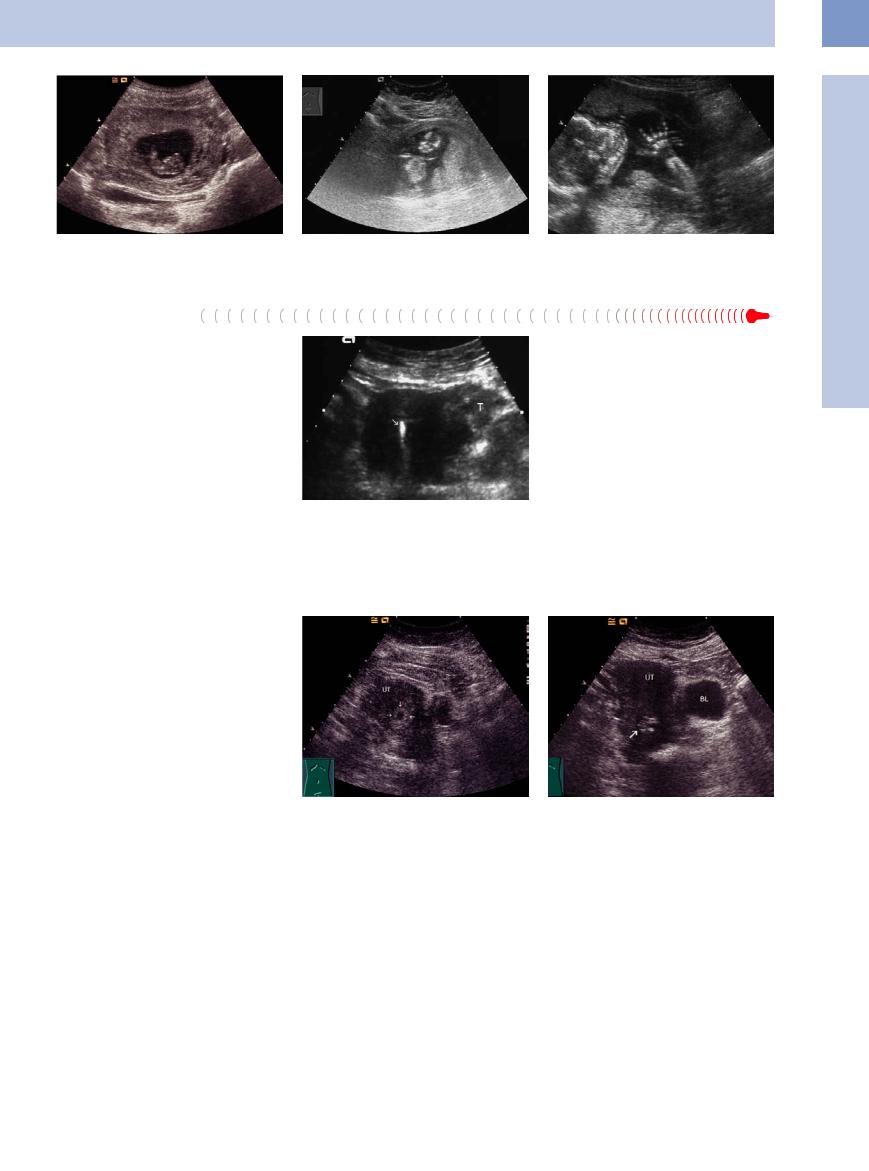
Fig. 13.44 Early pregnancy: The embryo is very well de- |
Fig. 13.45 Embryo in the 12th week of pregnancy. |
Fig. 13.46 Fetus, last trimester, arms and legs very well |
tectable in its amnionic sac. |
|
detectable. |
Ectopic Pregnancy
In an ectopic pregnancy (1:100 intrauterine pregnancies),4 a pseudogestational sac may appear at the center of the uterus. This fluid collection is caused by a decidual reaction within the slit-like uterine cavity. The sac rarely exceeds 2 cm in diameter, lacks an echogenic rim, and does not enlarge. A mass with a hyperechoic ring and anechoic center can be demonstrated in the adnexal region. Since this usually represents a tubal abortion, a conceptus is not present.
Ectopic pregnancy occurs more frequently when an IUD is in place (Fig.13.47). It is extremely rare for an intrauterine pregnancy to coexist with an IUD: 1:35 000 pregnancies.4
Fig. 13.47 Ectopic pregnancy: thickened, hypoechoic fallopian tube (T); IUD in the uterine cavity (high-level echo, arrow).
Missed Abortion, Incomplete Abortion, Cervical Pregnancy
Abortion, Cervical Pregnancy 



























In a missed abortion, either the chorionic cavity does not contain a conceptus or the conceptus is rudimentary and fails to develop further. The adnexa appear normal (Fig.13.48).
In an incomplete abortion, structures with irregular outlines and varying echogenicity can be demonstrated within the uterine cavity. The hyperechoic areas are retained products of conception, while hypoechoic areas represent small pockets of blood.
In some cases the remnants of an incomplete abortion may be found entirely in the cervical region (Fig.13.49). Differentiation is then required from an abnormal cervical pregnancy, which is recognized by a club-like expansion of the cervix.4
Fig. 13.48 Early pregnancy. Suspicion of missed abortion in the fifth week of pregnancy. The chorionic cavity is surrounded by the hyperechoic chorion (arrows), an embryonic area is not detectable; no progress in size. UT = fundus of the uterus.
Fig. 13.49 Incomplete abortion: remnants of the embryonic area are still inside the uterus, visible as a nonhomogeneous formation in the cervix (arrow). In the following time curettage. BL = bladder; UT = uterus.
Endometrial  Polyps,
Polyps, Cervical
Cervical Polyps,
Polyps, Placental Polyps
Placental Polyps 






























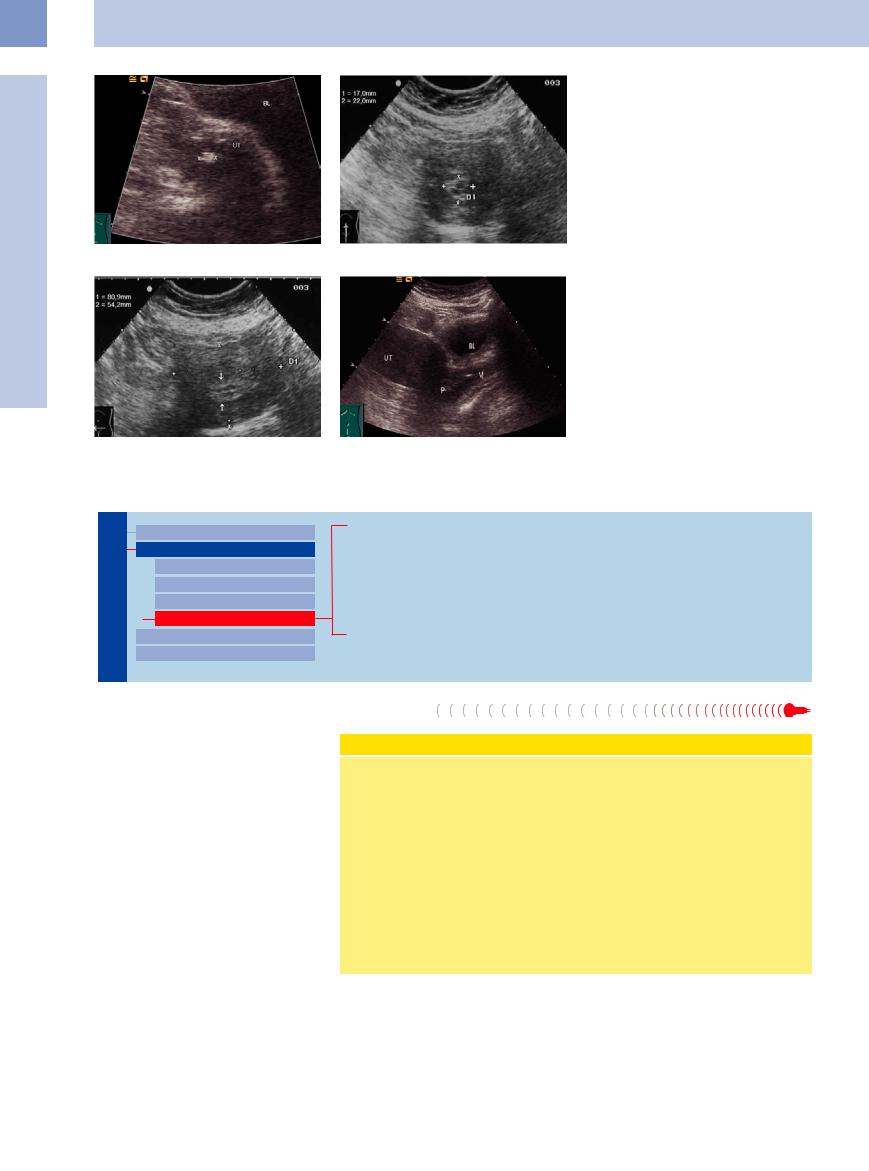
Endometrial polyps. Like adenomas, polyps arise from local sites of mucosal hypertrophy and project into the uterine cavity. They are usually benign (Fig.13.50). They may be pedunculated, and the pedicle may be so long that the polyp protrudes from the cervical os. Endometrial polyps are usually solitary. Large polyps can form in response to increased estrogen production after menopause. Treatment in
bleeding patients consists of dilatation and curettage. Further surveillance is indicated because of the risk of carcinoma.
Sonographically, the endometrium is hyperechoic and thickened with indistinct borders (Fig.13.51, Fig.13.52). If a mucocele or serometra is present, the polyps will be clearly delineated within the anechoic or hypoechoic uterine cavity.
Cervical polyps. More common than endometrial polyps, cervical polyps are lentil-size to cherry-size growths (Fig.13.53) that may be seen in the cervical os during speculum examination. Removal of these polyps should be followed by dilatation and curettage. Cervical polyps should not be removed during pregnancy.
453
13
Female Genital Tract
Fig. 13.50 Corpus polyp: hyperechoic mass (cursors), 7.5 mm in size, 80-year-old woman. UT = uterus, BL = bladder.
f Fig. 13.51 Endometrial polyp (cursors; histological finding): thickened, hyperechoic endometrium in a menopausal patient on tamoxifen. Nonhomogeneous uterine cavity.
Fig. 13.52 Endometrial polyp, transverse section: thickened endometrium (arrows); uterus normal in size. Cursors: uterine body.
f Fig. 13.53 Small cervical polyp (P) stretching from the portio into the vagina (V). UT = uterus; BL = bladder.
Endometrial Changes
Female Genital Tract

Vagina
Uterus
Abnormalities of Size or Shape
Myometrial Changes
Intracavitary Changes
Endometrial Changes
Fallopian Tubes
Ovaries
Menstrual Cycle: Proliferative Endometrium
Cystic Glandular and Atypical Adenomatous Hyperplasia
Endometritis, Cervicitis
Corpus Carcinoma
Chorioepithelioma
Cervical Carcinoma
Menstrual Cycle: Proliferative Endometrium
Proliferative Endometrium
The endometrium is more echogenic than the myometrium and appears as an oblong or elliptical structure with homogeneous echogenicity and smooth margins at the center of the uterus. At the beginning of the cycle the endometrium appears as a thin echogenic layer between the ventral and dorsal halves of the uterus.
Its thickness varies during the menstrual cycle, as well as its increase in thickness. Endometrial thickness is measured sonographically as twice the single-wall thickness, or the sum of both visible endometrial layers minus the intervening cavity.
Normal values of endometrial thickness. The normal values are hormone-dependent:
●Postmenopausal (without hormone replacement) <8 mm
●Postmenstrual 0 mm
●Premenstrual 8–15 mm
The menstrual history is helpful in interpreting the image correctly.
Cyclic Endometrial Changes
●After menstruation: no visible endometrium.
●Proliferative phase: central echogenic line 3 mm thick (Fig. 13.54), increasing hypoechoic boundary with the myometrium (Fig. 13.55).
●Midcycle: hyperechoic boundary with the myometrium, bright central echo, gaping cervical canal; graafian follicle may be detectable (Figs. 13.56, 13.57, 13.58).
●Secretory phase: After ovulation, luteal hormones stimulate increasing transformation of the tubular glands and glan-
dular secretions, giving the endometrium a porous, edematous appearance. The mucosa initially shows alternating hypoechoic and echogenic layers. The overall echogenicity of the endometrium increases during this phase while the bright central echo disappears (Fig. 13.59, Fig. 13.60), culminating in a well-defined hypoechoic center due to collected secretions (Fig. 13.61).
●Postmenopause: The postmenopausal endometrium appears at most as an echogenic band less than 8 mm thick, usually without a detectable cavity.
454
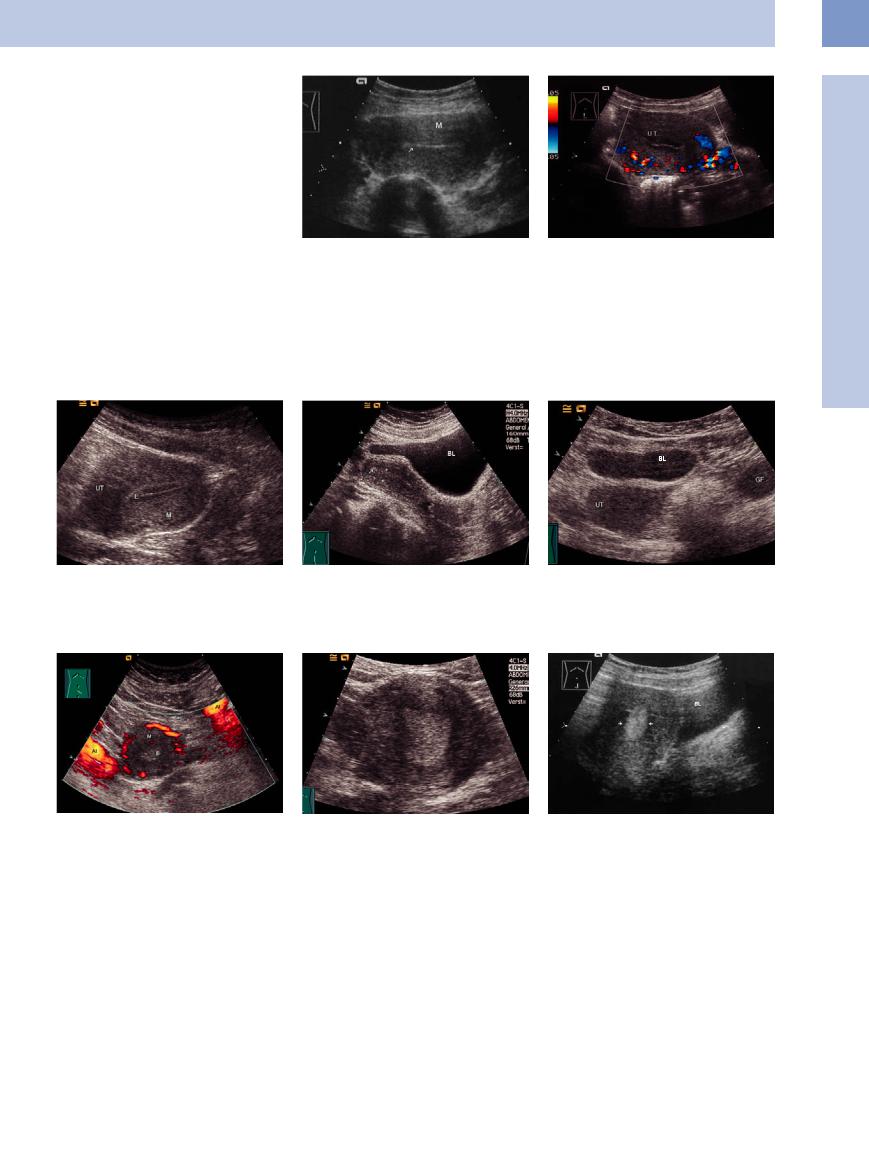
Pathological findings in the endometrium. The principal findings are as follows:
●Thickness >15 mm, or > (5–)8 mm after menopause.
●Nonhomogeneous echo pattern with microcystic or macrocystic changes, which may be benign or malignant. Cysts < 3 mm are typically benign.
●Indistinct endometrial/myometrial boundary.
A small amount of fluid in the uterine cavity does not have pathological significance.
A homogeneous endometrium more than 8 mm thick warrants ultrasound surveillance. The differential diagnosis includes hyperplasia and early endometrial carcinoma. Cytological examination is inconclusive, and the diagnosis is established by curettage.
Fig. 13.56 Endometrial layers at midcycle: hyperechoic – hypoechoic – hyperechoic – hypoechoic – hyperechoic (not illustrated). UT = uterus; M = myometrium.
Fig. 13.59 Uniformly echogenic endometrium (E) during the secretory phase after ovulation, with no central echo. Normal, circular myometrium blood flow pattern is clearly defined by power Doppler.
Fig. 13.54 Proliferative phase: endometrium with a fine central echo (arrow; first half of cycle). M = myometrium.
A bright internal echo in the uterine cavity with a homogeneous endometrium, seen particularly well in the presence of serometra, may be an endometrial polyp. This is confirmed by removing the lesion for histology.
Fig. 13.57 Endometrium at midcycle, day 14. Cursors: thickened endometrium; arrows: middle echo; BL = bladder.
Fig. 13.60 Thick, proliferative endometrium, second half of the cycle; regular thickness; endometrial thickness 20 mm.
Fig. 13.55 Endometrial layers at midcycle: hypoechoic – hyperechoic – hypoechoic. Proliferation phase.
It is normal after ovulation to find a small amount of free fluid in the rectouterine pouch, but other causes of ascites should be excluded.
Fig. 13.58 Mature graafian follicle (GF), day 14. BL = bladder; UT = uterus.
Fig. 13.61 Thick, proliferative endometrium (arrows), hyperechoic with a hypoechoic center. BL = bladder.
13
Uterus
455
13
Female Genital Tract
Cystic Glandular and Atypical
and Atypical Adenomatous
Adenomatous Hyperplasia
Hyperplasia





























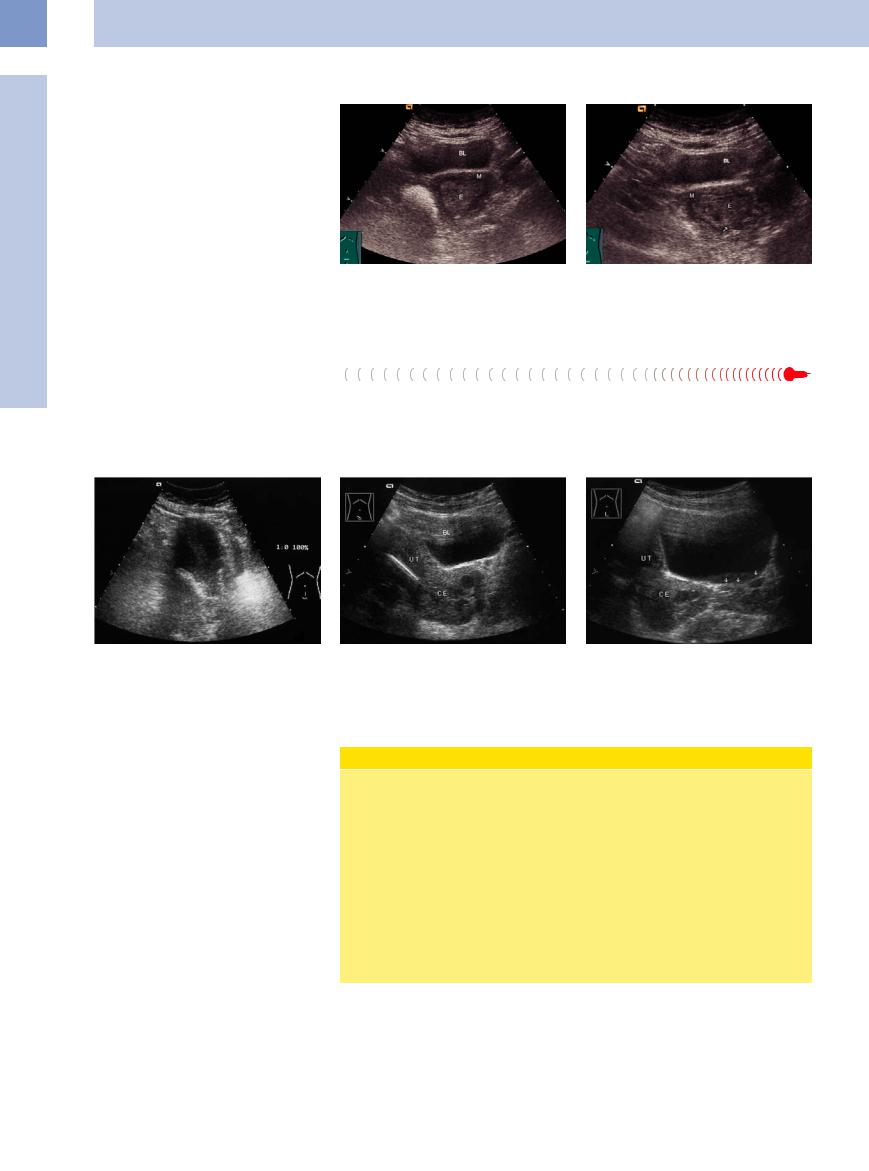
Cystic glandular hyperplasia occurs in an anovulatory cycle with a persistent follicle.7
Atypical adenomatous hyperplasia is diagnosed histologically by curettage. It is a precancerous lesion that progresses to corpus carcinoma in 20% of cases (Fig. 13.62, Fig.13.63).
Women who take tamoxifen (antiestrogen) are at increased risk of developing endometrial carcinoma.
Fig. 13.62 Pathologically thickened endometrium (E) well |
Fig. 13.63 Endometrium (E), 78-year-old woman, not en- |
defined from the myometrium; 78-year-old woman; his- |
tirely separated (arrow) from the myometrium (M); pos- |
tologically confirmed adenomatous hyperplasia, not yet |
sible transition to a carcinoma. |
carcinoma. BL = bladder; M = myometrium. |
|
Endometritis, Cervicitis





Inflammation of the endometrium due to tu- |
13.65, 13.66). Estrogen production promotes |
berculosis or other diseases spreads by a de- |
inflammatory changes. One-third of cases of |
scending route and may also involve the cervix |
tuberculous salpingitis occur during puberty. |
(cervicitis) and vagina (colpitis) (Figs. 13.64, |
The most common cause is an infected IUD. |
Fig. 13.64 Endometritis, myometritis.
a Hypoechoic uterus with indistinct borders, a retrouterine abscess, marked tenderness to pressure, and a foulsmelling discharge.
Fig. 13.65 Posterior to the bladder (BL), the cervix (CE) is |
Fig. 13.66 Cervicitis (CE) with parauterine lymph nodes |
markedly expanded and contains hypoechoic foci signify- |
(arrows). |
ing microabscesses. The uterus (UT) contains an IUD. |
|
Corpus Carcinoma


















































Histology. Carcinoma of the uterine corpus is generally an endometrial cancer. The majority are adenocarcinomas (40%). Corpus carcinoma arises from the endometrium of the uterine fundus, usually in the cornual region. A typical lesion is adenocarcinoma of the endometrium with extensive squamous cell metaplasia, found in 28% of cases (these tumors are called adenocancroids or adenocanthomas). Squamous cell carcinoma of the uterine corpus is rare, accounting for less than 1% of lesions. Stromal cancers (mucosal sarcomas) can also arise from the endometrium.
Risk factors. Tumor growth is estrogen-de- pendent and is mediated by estrogen receptors on the glandular epithelial and stromal cells. This is why years of unopposed estrogen stimulation is thought to be associated with a higher risk of endometrial carcinoma (ovarian tumors, polycystic ovaries, hepatic cirrhosis, etc.).
FIGO and TNM Staging for Corpus Carcinoma
0/Tis Carcinoma in situ
I/T1 Tumor confined to the uterine corpus IA/T1a Tumor confined to the endometrium
IB/T1b Tumor invades less than half of the myometrium IC/T1c Tumor invades half or more of the myometrium II/T2 Tumor invades the cervix
IIA/T2a Spread limited to the endocervix IIB/T2b Tumor also invades the cervical stroma III/T3 Spread in the lesser pelvis
IIIA/T3a Tumor involves the serosa and/or adnexa, and/or tumor detected in ascites IIIB/T3b Vaginal involvement
IIIC/N1 Pelvic or para-aortic lymph node metastases IVA/T4 Invasion of the bladder and/or rectum
IVB/M1 Distant lymph node metastases outside the lesser pelvis
An increased risk exists in endometrial carcinoma syndrome with overweight, hypertension, and glucose intolerance or diabetes mellitus. The principal risk factor is age, and
456
13
Uterus
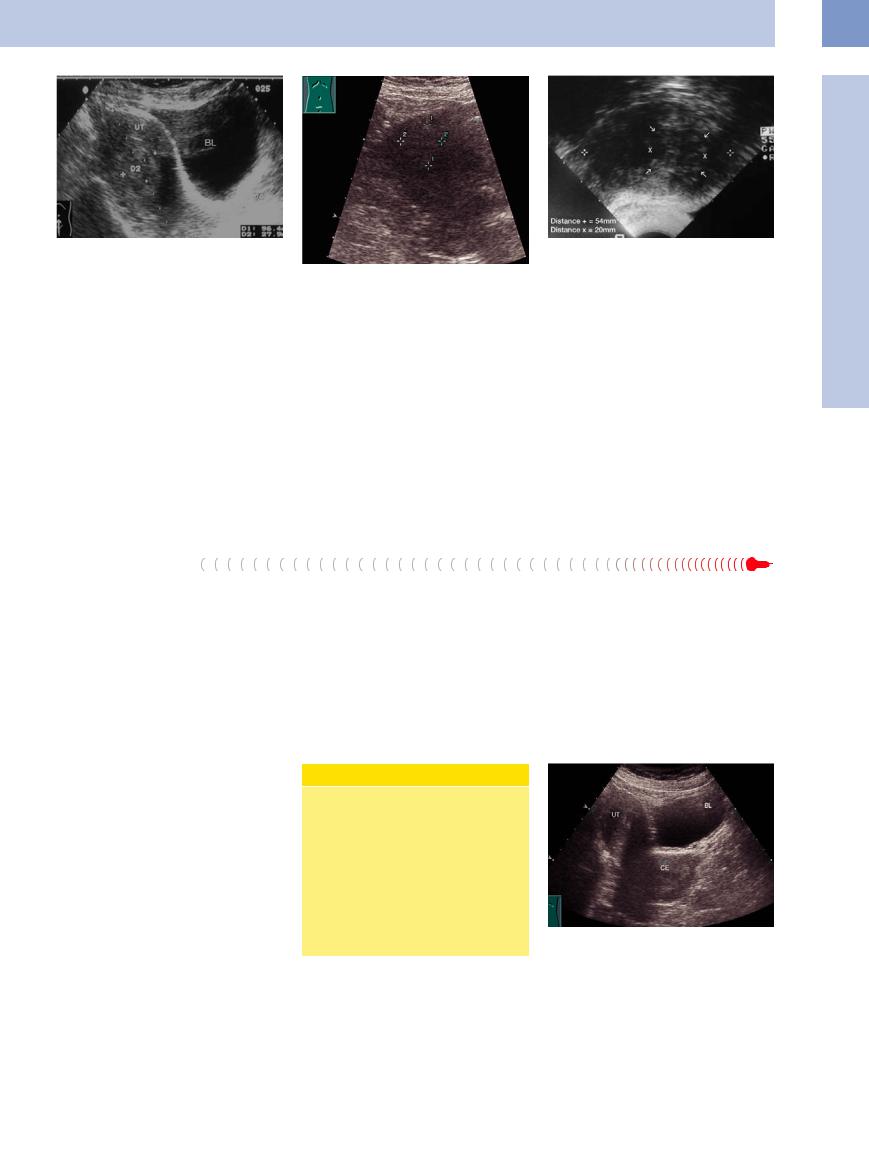
Fig. 13.67 Endometrial carcinoma (D 2: 27.9 mm) in a postmenopausal patient. The uterus (UT, 96.4 mm long) is still just distinguishable from the myometrium by its greater echogenicity. BL = bladder.
women with a higher socioeconomic status seem to be predisposed.
Growth pattern. Corpus carcinoma grows slowly on the mucosa or may form a polypoid lesion projecting into the uterine cavity; thus it stays confined to the mucosa for some time. It eventually invades the myometrium by contiguous spread but rarely penetrates the serosa. Metastasis occurs much later than with cervical carcinoma and only after the tumor has permeated the outer third of the myometrium.
Chorioepithelioma
Choriocarcinoma (malignant chorioepithelioma) arises from syncytial and Langhans cells of the placenta following a birth (20%), miscarriage (25%), or molar pregnancy (50%). It may develop after a latent period of several years. The overall incidence of chorioepithelioma is very low (1:100 000 births).
Fig. 13.68 Endometrial carcinoma: the tumor (cursors) is just distinguishable by its higher echogenicity in the transabdominal scan.
Distant metastases are rare and occur at a late stage. Bleeding usually directs attention to the cancer while it is still confined to the uterine corpus.
Sonographic features. The sonographic hallmarks of endometrial carcinoma are as follows:
●Thick, hyperechoic endometrium
●Macrocystic changes
●Ill-defined margins
●Expansion of the uterine cavity by tumor with an exophytic growth pattern
The tumor begins with a nodule in the endometrium, which grows rapidly into the uterine wall. Chorioepithelioma is extremely malignant and metastasizes early to lung, brain, kidney, bone, etc. Bleeding is a frequent complication. Serum levels of chorionic gonadotropins and α-hCG are elevated.
Fig. 13.69 Transvaginal scan: 2 cm carcinoma (arrows) with ill-defined margins. Uterine width is 54 mm.
The uterus may be larger than normal for age. Endometrial carcinoma appears more echogenic than the myometrium. The precise assessment of myometrial invasion has a crucial bearing on the prognosis. Transabdominal ultrasound is not sufficiently precise for this purpose, but transvaginal scanning can define the myometrium with great clarity (Figs. 13.67, 13.68, 13.69).
Ultrasound shows inadequate postpartum involution of the uterus in addition to an enlarging intracavitary mass. Because of the chorionic gonadotropins from chorioepithelioma, the ovaries contain corpora lutea at various developmental stages as well as granulosa and theca-lutein cysts. As a result, cystic ovaries are frequently noted at ultrasonography.
Cervical Carcinoma 

















































Cervical carcinoma is usually a nonkeratinizing squamous cell carcinoma that metastasizes early to regional lymph nodes. Even with a microcarcinoma (FIGO IA2, TNM T1a2) not exceeding 5 cm in depth and 7 mm in its superficial extent, metastases are already present in 10% of cases. The tumor spreads contiguously to the vagina, uterus, and parametria. Distant metastases are rare and occur at a late stage.
Sonographic features. The carcinoma initially appears hyperechoic. Transabdominal ultrasound can detect the tumor only after it has reached an advanced stage (at least IB/T1b) (Fig. 13.70, Fig.13.71). With endophytic tumors, the cervix may appear greatly thickened; with exophytic tumors (less common), a cauliflower-like growth can be seen (Fig.13.72). A barrel-like distention of the cervix is most often seen with cancers of the cervical cavity. Over time the tumor may become less echogenic, particularly in response to
FIGO Staging for Cervical Carcinoma
0Carcinoma in situ
IStrictly confined to the cervix
IIExtends into the parametria (but not onto the pelvic sidewall) or the upper two-thirds of the vagina
IIIHas spread onto the pelvic sidewall and/or involves the lower third of the vagina
IV Has spread beyond the lesser pelvis to distant organs and/or has invaded the bladder, rectum, or both
treatment (Fig.13.73). Transvaginal sonography (TVS) yields more accurate findings and permits an earlier diagnosis.
A sharply circumscribed, globular type of tumor requires differentiation from a uterine
Fig. 13.70 Cervical carcinoma: balloonlike enlarged uterine cervix (CE), well defined to the surrounding tissue. BL = bladder; UT = uterus.
myoma. An extensive ovarian cancer can also mimic cervical carcinoma.
457
