
- •Contents
- •Preface
- •Contributors
- •1 Vessels
- •1.1 Aorta, Vena Cava, and Peripheral Vessels
- •Aorta, Arteries
- •Anomalies and Variant Positions
- •Dilatation
- •Stenosis
- •Wall Thickening
- •Intraluminal Mass
- •Perivascular Mass
- •Vena Cava, Veins
- •Anomalies
- •Dilatation
- •Intraluminal Mass
- •Compression, Infiltration
- •1.2 Portal Vein and Its Tributaries
- •Enlarged Lumen Diameter
- •Portal Hypertension
- •Intraluminal Mass
- •Thrombosis
- •Tumor
- •2 Liver
- •Enlarged Liver
- •Small Liver
- •Homogeneous Hypoechoic Texture
- •Homogeneous Hyperechoic Texture
- •Regionally Inhomogeneous Texture
- •Diffuse Inhomogeneous Texture
- •Anechoic Masses
- •Hypoechoic Masses
- •Isoechoic Masses
- •Hyperechoic Masses
- •Echogenic Masses
- •Irregular Masses
- •Differential Diagnosis of Focal Lesions
- •Diagnostic Methods
- •Suspected Diagnosis
- •3 Biliary Tree and Gallbladder
- •3.1 Biliary Tree
- •Thickening of the Bile Duct Wall
- •Localized and Diffuse
- •Bile Duct Rarefaction
- •Localized and Diffuse
- •Bile Duct Dilatation and Intraductal Pressure
- •Intrahepatic
- •Hilar and Prepancreatic
- •Intrapancreatic
- •Papillary
- •Abnormal Intraluminal Bile Duct Findings
- •Foreign Body
- •The Seven Most Important Questions
- •3.2 Gallbladder
- •Changes in Size
- •Large Gallbladder
- •Small/Missing Gallbladder
- •Wall Changes
- •General Hypoechogenicity
- •General Hyperechogenicity
- •General Tumor
- •Focal Tumor
- •Intraluminal Changes
- •Hyperechoic
- •Hypoechoic
- •Nonvisualized Gallbladder
- •Missing Gallbladder
- •Obscured Gallbladder
- •4 Pancreas
- •Diffuse Pancreatic Change
- •Large Pancreas
- •Small Pancreas
- •Hypoechoic Texture
- •Hyperechoic Texture
- •Focal Changes
- •Anechoic Lesion
- •Hypoechoic Lesion
- •Isoechoic Lesion
- •Hyperechoic Lesion
- •Irregular (Complex Structured) Lesion
- •Dilatation of the Pancreatic Duct
- •Marginal/Mild Dilatation
- •Marked Dilatation
- •5 Spleen
- •Nonfocal Changes of the Spleen
- •Diffuse Parenchymal Changes
- •Large Spleen
- •Small Spleen
- •Focal Changes of the Spleen
- •Anechoic Mass
- •Hypoechoic Mass
- •Hyperechoic Mass
- •Splenic Calcification
- •6 Lymph Nodes
- •Peripheral Lymph Nodes
- •Head/Neck
- •Extremities (Axilla, Groin)
- •Abdominal Lymph Nodes
- •Porta Hepatis
- •Splenic Hilum
- •Mesentery (Celiac, Upper and Lower Mesenteric Station)
- •Stomach
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •Small/Large Intestine
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •8 Peritoneal Cavity
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Wall Structures
- •Smooth Margin
- •Irregular Margin
- •Intragastric Processes
- •Intraintestinal Processes
- •9 Kidneys
- •Anomalies, Malformations
- •Aplasia, Hypoplasia
- •Cystic Malformation
- •Anomalies of Number, Position, or Rotation
- •Fusion Anomaly
- •Anomalies of the Renal Calices
- •Vascular Anomaly
- •Diffuse Changes
- •Large Kidneys
- •Small Kidneys
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Irregular Structure
- •Circumscribed Changes
- •Anechoic Structure
- •Hypoechoic or Isoechoic Structure
- •Complex Structure
- •Hyperechoic Structure
- •10 Adrenal Glands
- •Enlargement
- •Anechoic Structure
- •Hypoechoic Structure
- •Complex Echo Structure
- •Hyperechoic Structure
- •11 Urinary Tract
- •Malformations
- •Duplication Anomalies
- •Dilatations and Stenoses
- •Dilated Renal Pelvis and Ureter
- •Anechoic
- •Hypoechoic
- •Hypoechoic
- •Hyperechoic
- •Large Bladder
- •Small Bladder
- •Altered Bladder Shape
- •Intracavitary Mass
- •Hypoechoic
- •Hyperechoic
- •Echogenic
- •Wall Changes
- •Diffuse Wall Thickening
- •Circumscribed Wall Thickening
- •Concavities and Convexities
- •12.1 The Prostate
- •Enlarged Prostate
- •Regular
- •Irregular
- •Small Prostate
- •Regular
- •Echogenic
- •Circumscribed Lesion
- •Anechoic
- •Hypoechoic
- •Echogenic
- •12.2 Seminal Vesicles
- •Diffuse Change
- •Hypoechoic
- •Circumscribed Change
- •Anechoic
- •Echogenic
- •Irregular
- •12.3 Testis, Epididymis
- •Diffuse Change
- •Enlargement
- •Decreased Size
- •Circumscribed Lesion
- •Anechoic or Hypoechoic
- •Irregular/Echogenic
- •Epididymal Lesion
- •Anechoic
- •Hypoechoic
- •Intrascrotal Mass
- •Anechoic or Hypoechoic
- •Echogenic
- •13 Female Genital Tract
- •Masses
- •Abnormalities of Size or Shape
- •Uterus
- •Abnormalities of Size or Shape
- •Myometrial Changes
- •Intracavitary Changes
- •Endometrial Changes
- •Fallopian Tubes
- •Hypoechoic Mass
- •Anechoic Cystic Mass
- •Solid Echogenic or Nonhomogeneous Mass
- •14 Thyroid Gland
- •Diffuse Changes
- •Enlarged Thyroid Gland
- •Small Thyroid Gland
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Circumscribed Changes
- •Anechoic
- •Hypoechoic
- •Isoechoic
- •Hyperechoic
- •Irregular
- •Differential Diagnosis of Hyperthyroidism
- •Types of Autonomy
- •15 Pleura and Chest Wall
- •Chest Wall
- •Masses
- •Parietal Pleura
- •Nodular Masses
- •Diffuse Pleural Thickening
- •Pleural Effusion
- •Anechoic Effusion
- •Echogenic Effusion
- •Complex Effusion
- •16 Lung
- •Masses
- •Anechoic Masses
- •Hypoechoic Masses
- •Complex Masses
- •Index

6 Lymph Nodes
Lymph Nodes 233
|
|
|
Peripheral Lymph Nodes |
241 |
||||||||
|
|
|
||||||||||
|
|
|
|
|
|
|
|
Head/Neck |
241 |
|||
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
Inflammatory Lymph Nodes |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Metastases |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Malignant Lymphoma |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Other Structures |
|
|
|
|
|
|
|
Extremities (Axilla, Groin) |
245 |
|||||
|
|
|
|
|
|
|
|
|
|
|
Inflammatory Lymph Nodes |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Metastases |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Malignant Lymphoma |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Other Structures |
|
|
|
Abdominal Lymph Nodes |
247 |
|||||||||
|
|
|||||||||||
|
|
|
|
|
|
|
Porta Hepatis |
247 |
||||
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
Inflammatory Lymph Nodes |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Metastases |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Malignant Lymphoma |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Other Structures |
|
|
|
|
|
|
|
Splenic Hilum |
249 |
|||||
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
Inflammatory Lymph Nodes |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Metastases |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Malignant Lymphoma |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Other Structures |
|
|
|
|
|
|
|
Mesentery (Celiac, Upper and |
|
|||||
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
Lower Mesenteric Station) |
251 |
|||||
|
|
|
|
|
|
|
|
|
|
|
Inflammatory Lymph Nodes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Metastases |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Malignant Lymphoma |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Structures |
|
|
|
|
|
|
|
Retroperitoneum (Para-Aortic, |
|
|||||
|
|
|
|
|
|
Paracaval, Aortointercaval, |
|
|||||
|
|
|
|
|
|
and Iliac Station) |
254 |
|||||
Inflammatory Lymph Nodes
Metastases
Malignant Lymphoma
Other Structures


6Lymph Nodes
C. Goerg
Together with the spleen and the mucosa-as- sociated lymphoid tissue (MALT), lymph nodes form the essential barrier of the secondary immune system. The usual lymph node diameter varies between 0.2 cm and 2.5 cm and depends on the functional state of the lymph node as well as the age of the patient. The lymph nodes are in connection with the lymphatics.
Assessment of possible lymph node malignancy is based primarily on the clinical picture (history, workplace, and age of patient; location of lymph node in question; palpation of finding; experience of examining physician) and only secondarily on the ultrasound findings. A definite diagnosis has to await confirmation by histology. Sonographic follow-up has proved to be invaluable in terms of assessing possible malignancy as well as documenting the response to therapy. The diagnostic armamentarium for the follow-up and detection of recurrence, particularly in diseases of the peripheral lymph nodes, has gained immeasurably by the addition of ultrasound.1–6
A summary of possible differential diagnosis in pathological lymph nodes is listed in Table 6.1.
Table 6.1 Differential diagnosis in lymphadenopathy2
Infectious disease
●Viral infection:
infectious mononucleosis (EBV), cytomegalic inclusion disease, infectious hepatitis, AIDS, rubella, human herpesvirus 3 (varicella-zoster)
●Bacterial infection:
streptococci, staphylococci, salmonellae, Tropheryma whipplei (Whipple disease), Francisella tularensis (tularemia)
●Mycotic infection:
histoplasmosis, coccidioidomycosis, blastomycosis
●Chlamydial infection:
lymphogranuloma venereum, cat-scratch disease, trachoma
●Mycobacterial infection:
toxoplasmosis, trypanosomiasis, microfilariae
●Spirochetosis: syphilis, leptospirosis
Malignancy
●Hodgkin disease, non-Hodgkin lymphoma, chronic lymphocytic leukemia, acute lymphocytic leukemia
●Myelogenous leukemia (chloromas = extramedullary manifestation of myelogenous leukosis): acute myelogenous leukemia, blast crisis in chronic myelogenous leukemia
●Metastasis of epithelial or mesenchymal tumors
Immunological disease
●Rheumatoid arthritis
●Systemic lupus erythematosus
●Dermatomyositis
●Allergic reaction to drugs such as phenytoin, hydralazine, allopurinol
Other
●Sarcoidosis
●Lipid storage disease (Gaucher disease, Niemann–Pick disease)
Anatomy























































The trabeculae extend from the capsule, con- |
made up of the cortex, the paracortex, and the |
|
sisting of connective tissue and a few elastic |
medulla where the immune response takes |
|
fibrils, into the substance of the lymph node, |
place. The hilum is rich in connective tissue |
|
resulting in a |
radial configuration. From the |
and houses the afferent and efferent lym- |
outside to the |
inside, the cortical substance is |
phatics and blood vessels. |
Topography 





















































Overall, the normal patient harbors approxi- |
Each of these major drainage regions is subdi- |
Lymph node regions of the head/neck |
||
mately 1000 lymph nodes. The lymphatic sys- |
vided into several lymphatic subregions, the |
(Fig. 6.1) |
||
tem in mammals may be grouped into five |
names of which are primarily taken from the |
I |
Submental, submandibular |
|
large regions, and the lymph of each drains |
adjacent blood vessels. For didactic as well as |
II |
Lymph nodes draining into the upper third |
|
into a major lymphatic trunk: |
differential diagnostic reasons, the lymph |
|
of the jugular vein |
|
1. |
Head/neck |
nodes should be classified as peripheral or non- |
III Lymph nodes draining into the middle third |
|
2. |
Axilla (including ipsilateral arm, mammary |
peripheral. Mediastinal and pulmonary lymph |
|
of the jugular vein |
|
gland, and thoracic wall) |
nodes are excluded from this discussion since |
IV Lymph nodes draining into the lower third |
|
3. |
Groin (including ipsilateral leg and abdomi- |
transcutaneous sonography cannot visualize |
|
of the jugular vein (including the supracla- |
|
nal wall) |
them by conventional means. |
|
vicular lymph nodes) |
4. |
Intra-abdominal and pelvic organs |
Peripheral lymph nodes include the fol- |
V |
Nuchal |
5. |
Intrathoracic organs |
lowing regions: |
|
|
|
|
● Head/neck |
|
|
|
|
● Axilla/groin |
|
|
6
Lymph Nodes
233
6
Lymph Nodes
Relations
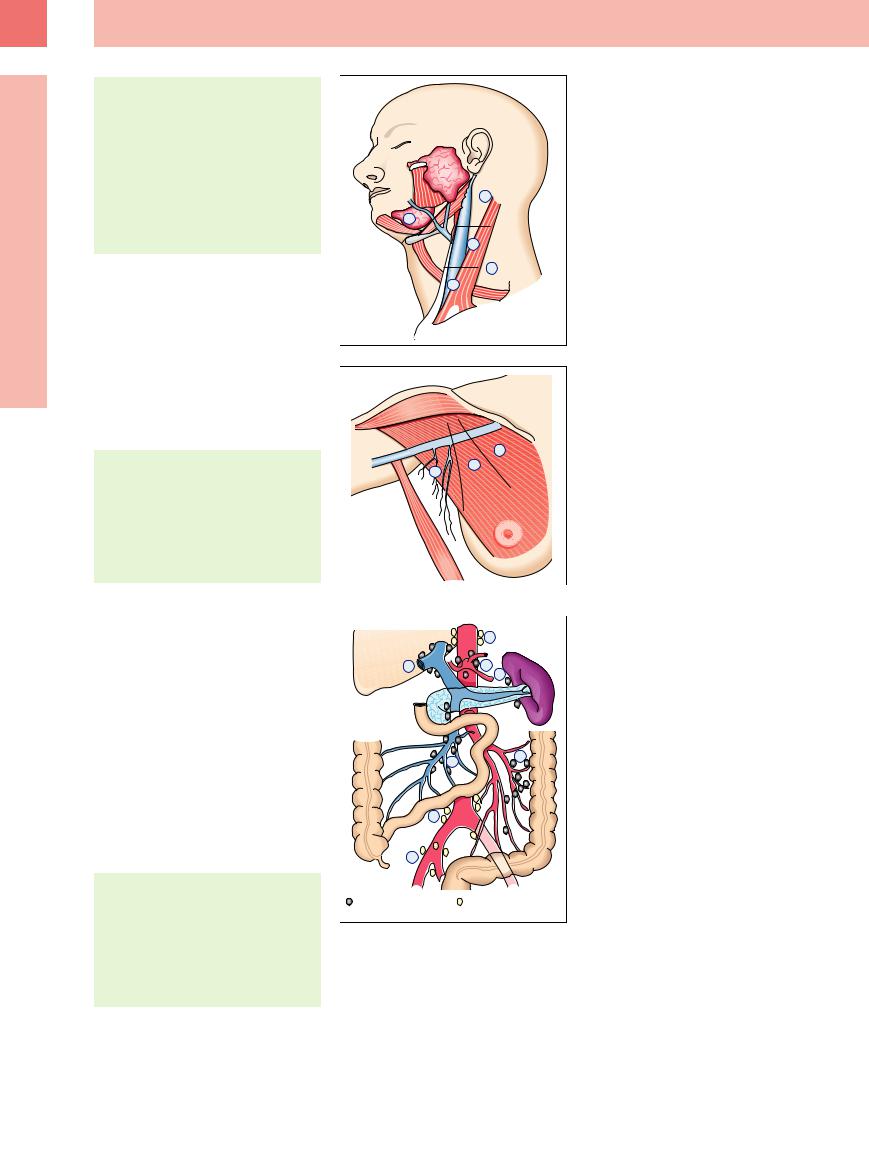
●Cervical lymph nodes along the jugular vein and carotid artery
●Submandibular lymph nodes along the muscles of the root of the tongue
●Nuchal lymph nodes posterior to the sternocleidomastoid muscle
Landmark structure in ultrasound
●Jugular vein
●Carotid artery
●Subclavian artery and vein
Regions of the axillary lymph nodes (Fig. 6.2)
I Lymph nodes of the lower axillary region
IILymph nodes of the central axillary region III Lymph nodes of the upper axillary region (including the infraclavicular lymph nodes)
Relations
●Lymph nodes along the axillary vessels
●Lymph nodes anterior, along, and posterior to the pectoralis major muscle
Landmark structure in ultrasound
●Axillary vessels
●Pectoralis major muscle
Nonperipheral lymph nodes. The abdominal (nonperipheral) lymph nodes comprise the following regions (Fig. 6.3):
●Intraperitoneal (splanchnic) lymph nodes (hepatic hilum, splenic hilum, mesentery)
●Retroperitoneal (parietal) lymph nodes (para-aortic, iliac)
Regions of the intraperitoneal lymph nodes
I Lymph nodes at the hepatic hilum II Lymph nodes at the splenic hilum III Mesenteric lymph nodes
Regions of the retroperitoneal lymph nodes
I. Para-aortic/paracaval lymph nodes II. Iliac lymph nodes
Relations
●Intraperitoneal along the blood vessels leading to liver and spleen
●Along the mesenteric blood vessels
●Along the retroperitoneal iliac blood vessels
Landmark structures for ultrasound
● Arteries and veins
Fig. 6.1 Topography of the lymph nodes in the head and neck.
Fig. 6.2 Topography of the axillary lymph nodes.
Fig. 6.3 Topography of the abdominal lymph nodes: 1 = porta hepatis
2 = hilum of spleen
3 = celiac group
4 = upper and lower mesenteric group
5 = para-aortic group
6 = iliac group
234
Sonographic Morphology
Morphology














































Normal lymph node morphology
●Size < 1 cm
●Discernible hilum
●Cortex and hilum distinctly different
●Ellipsoid shape (length:width ≥ 2)
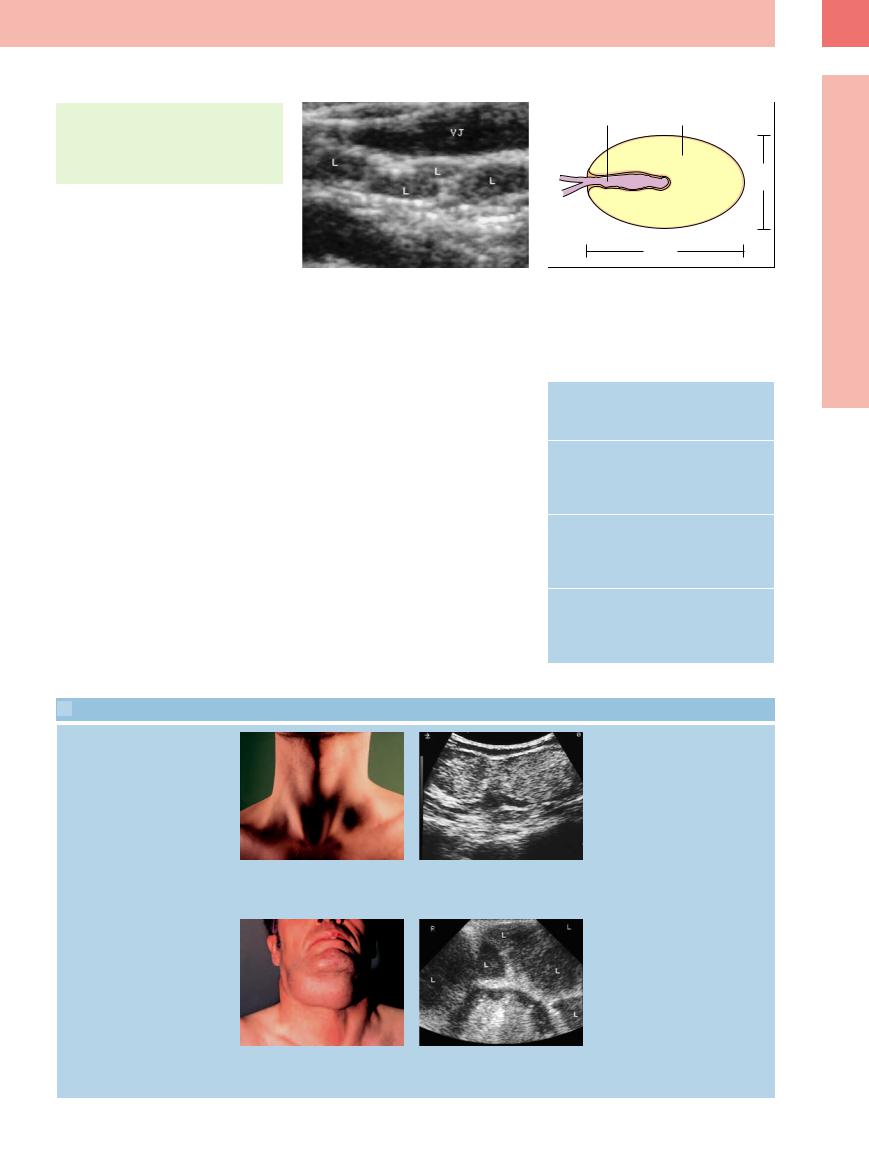
Usually, only enlarged lymph nodes will be visualized for certain, but normal lymph nodes can be detected down to a size of only 2–3 mm (Fig. 6.4). Lymph nodes of the head and neck measuring more than 10 mm across are considered as being pathologically enlarged. Quite often a central hyperechoic ovoid hilum can be differentiated from a peripheral hypoechoic concentric cortex (Fig. 6.5).
The sonographic parameters for lymph node assessment are listed in Table 6.2.
Location. The location alone of a lymph node may point the way to the etiology (supraclavicular sentinel, or Virchow’s, node in gastric cancer, painful inguinal lymphadenopathy in erysipelas (alarm-bell sign), unilateral axillary lymphadenopathy in breast cancer;  6.1a–f).
6.1a–f).
One important criterion for assessing possible malignancy is the pattern of involvement, i. e., whether this is localized or generally spread.
The relation with the blood vessels is of particular importance in ENT since it could determine the difference between surgery and inoperability. If ultrasound demonstrates contact between the tumor and the vascular wall over a distance of more than 3.5 cm or more than half the circumference, invasion of the vessel no longer amenable to resection is indicated.4
Fig. 6.4 An 8 mm long enlarged lymph node (L) of homogeneous echogenicity in an otherwise asymptomatic patient. The lymphadenopathy was considered to be reactive. VJ = jugular vein.
Morphology. Apart from the size ( 6.1 g–i), morphological assessment has to consider the shape of the lymph nodes as well (
6.1 g–i), morphological assessment has to consider the shape of the lymph nodes as well ( 6.1j–l). The parameter commonly used is the ratio of length (L) to width (W); a L/W ratio greater than 2 is more indicative of reactive changes, whereas a ratio less than 2 suggests malignancy (Fig. 6.5). The information gained by palpation can be supplemented by ultrasound because of its ability to differentiate between a well-defined lymph node and festoon-like lymphadenopathy or lymphomas in close contact like potatoes in a bag (
6.1j–l). The parameter commonly used is the ratio of length (L) to width (W); a L/W ratio greater than 2 is more indicative of reactive changes, whereas a ratio less than 2 suggests malignancy (Fig. 6.5). The information gained by palpation can be supplemented by ultrasound because of its ability to differentiate between a well-defined lymph node and festoon-like lymphadenopathy or lymphomas in close contact like potatoes in a bag ( 6.1 m–o).
6.1 m–o).
Structural parameters. Regarding the structural parameters, the importance of any hilar sign (present or missing) is generally accepted, and it supposedly is indicative of malignant lymphadenopathy ( 6.2a–f).
6.2a–f).
The assessment of the echogenicity of any lymph node should always be based on an “invivo” reference, usually the adjacent tissue
Fig. 6.5 Schematic illustration of a lymph node.
Table 6.2 Sonographic parameters for lymph node assessment
Location
●Region involved
●Pattern of involvement
Morphology
●Size
●Shape
●Delineation from adjacent tissue
Structural parameters
●Hilar sign
●Echogenicity
●Homogeneity
Vascularization
●Concentration of blood vessels
●Pattern of vascularization
●Flow parameters
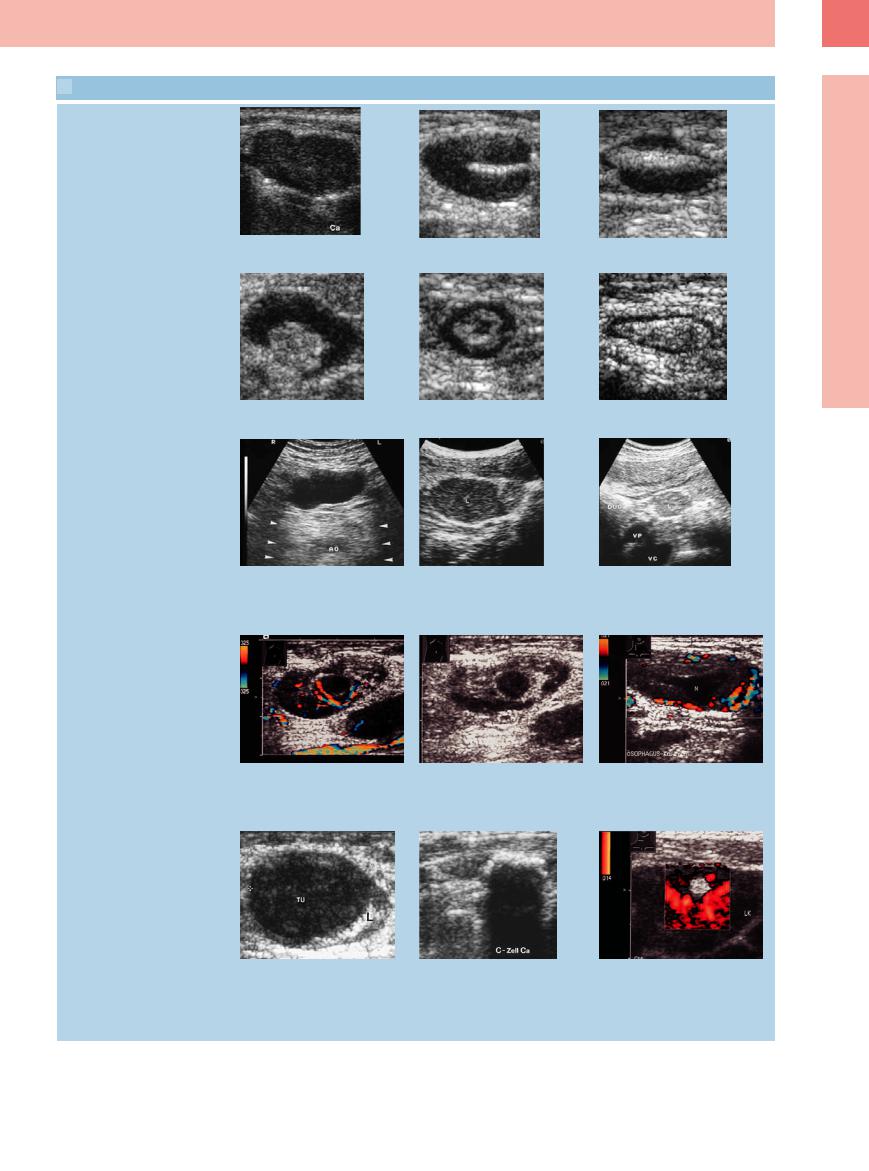
 6.1 Location and Morphology of Abnormal Lymph Nodes
6.1 Location and Morphology of Abnormal Lymph Nodes
Location
6
Lymph Nodes
a and b A 29-year-old patient with lymph node in left supraclavicular fossa (“Virchow’s sentinel node”). Ultrasound demonstrates two large hyperechoic lymph node metastases stuck together (gastroscopy confirmed the suspected diagnosis of gastric cancer). This localization signals malignancy.
c and d Chronic lymphocytic leukemia
c A 63-year-old patient with submandibular tumor formation.
d Ultrasound demonstrates large lymph nodes (L) invaded by tumor and stuck together.
235

6
Lymph Nodes
 6.1 Location and Morphology of Abnormal Lymph Nodes (Continued)
6.1 Location and Morphology of Abnormal Lymph Nodes (Continued)
e and f A 51-year-old patient with erysipelas. Enlarged lymph node in the left groin with wide hyperechoic hilum and thin hypoechoic parenchymal border; reactive lymphadenopathy.
Size
g–i Lymph nodes of di erent size invaded by tumor.
g Small lymph node metastasis in the left groin in malignant melanoma.
Form
j–l Peripheral lymph nodes of di erent shape.
j Elongated hypoechoic reactive lymph node in erysipelas.
Delineation from surrounding tissue
m Multiple round hypoechoic tumor-infil- trated well-defined lymph nodes (L) in lung cancer.
h Lymph node metastasis of the lower jugular region in lung cancer.
k Ovoid hypoechoic lymph node invaded by malignant lymphoma.
n Multiple ill-defined lymph nodes (L) invaded by malignant lymphoma.
i Malignant lymphoma invading an inguinal lymph node.
l Round hypoechoic lymph node invaded by carcinoma.
o Multiple lymphomas stuck together in Hodgkin disease.
( 6.2 g–i). It has been stated that hypoechoic nodes may be typical for invasion by malignant lymphoma, while hyperechoic parenchyma seems to be more common in cancer metastasis and lymph nodes with chronic inflammation or regression scars.1
6.2 g–i). It has been stated that hypoechoic nodes may be typical for invasion by malignant lymphoma, while hyperechoic parenchyma seems to be more common in cancer metastasis and lymph nodes with chronic inflammation or regression scars.1
The normal parenchyma is homogeneous but there may be clusters of masses that are anechoic ( 6.2k,l), hypoechoic (
6.2k,l), hypoechoic ( 6.2j,m), or hyperechoic (
6.2j,m), or hyperechoic ( 6.2o). Anechoic lesions are seen in granulomatous or suppurating inflammation corresponding with necrosis, but also in cancer metastases where the lesions would correspond with colliquation. Hypoechoic lesions may be indicative of early malignant
6.2o). Anechoic lesions are seen in granulomatous or suppurating inflammation corresponding with necrosis, but also in cancer metastases where the lesions would correspond with colliquation. Hypoechoic lesions may be indicative of early malignant
lymph node invasion. Hyperechoic regions in lymph nodes are seen in low-grade malignant lymphoma and could correspond with regressive changes. Calcification may represent lymph node scarring. However, sometimes lymph node metastases tend to calcify as well (e. g., medullary thyroid carcinoma) ( 6.2n).
6.2n).
Vascularization. CDS, especially power Doppler can demonstrate lymph node vascularization only indirectly by the presence or absence of flow phenomena.
The afferent and efferent hilar vessels in a normal-sized lymph node can be visualized by color-flow Doppler imaging. Depending on the
state of the lymph node, the cortical region will display a spoke-like or increased parenchymal vascularization with a markedly hilar aspect ( 6.3a–f). Semiquantitative assessment of the concentration of blood vessels differentiates between increased vascularization (
6.3a–f). Semiquantitative assessment of the concentration of blood vessels differentiates between increased vascularization ( 6.3 g,h), and a focal decrease or lack of flow phenomena in the lymph node. “Increased vascularization” is said to be indicative of reactive lymphadenopathy in acute inflammation (
6.3 g,h), and a focal decrease or lack of flow phenomena in the lymph node. “Increased vascularization” is said to be indicative of reactive lymphadenopathy in acute inflammation ( 6.3i,j), but it is also seen in malignant lymphoma. A focal lack of flow phenomena in lymph nodes has frequently been observed in metastasis.7 In addition, this lack of flow signals is also quite common post therapy (
6.3i,j), but it is also seen in malignant lymphoma. A focal lack of flow phenomena in lymph nodes has frequently been observed in metastasis.7 In addition, this lack of flow signals is also quite common post therapy ( 6.3k,l).
6.3k,l).
236
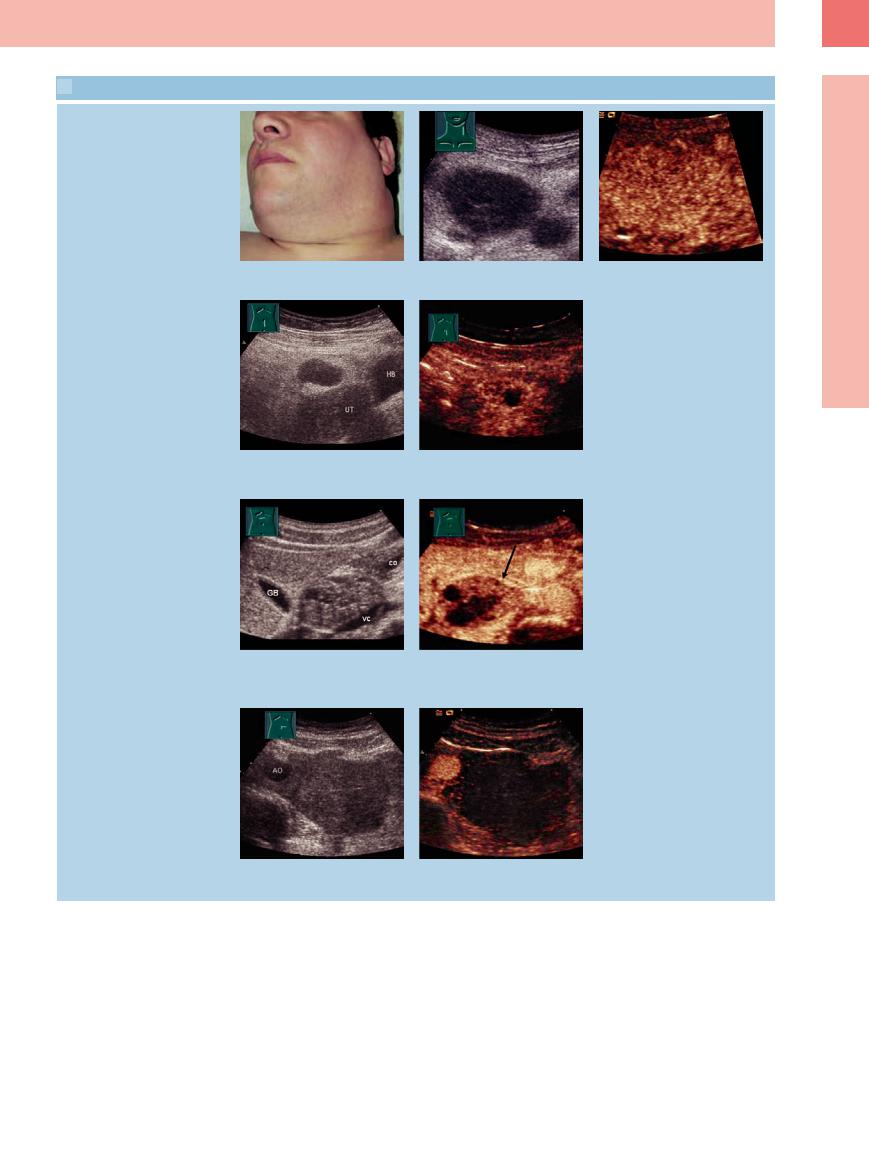
 6.2 Structural Parameters of Abnormal Lymph Nodes
6.2 Structural Parameters of Abnormal Lymph Nodes
Hilar sign
a–c Peripheral lymph nodes with different levels of the hilar sign. Lymph node with malignant invasion and no or just a faint hilar sign (a).
d–f Enlarged reactive lymph node with mild to marked hilar sign.
Echogenicity
g–i Enlarged lymph nodes displaying dif- |
h Hypoechoic cervical lymph node meta- |
i Hyperechoic reactive lymph node (L) in |
ferent levels of echogenicity. |
stasis in ovarian cancer. |
HIV-infected patient; located in the liver |
g Hypoechoic to “anechoic” mesenteric |
|
hilum. VC = caval vein, VP = portal vein. |
lymph node invaded by malignant lym- |
|
Duo = duodenum. |
phoma. |
|
|
Inhomogeneity
j Structural inhomogeneity of a lymphoma |
k Colliquation of an enlarged cervical |
l Lymph node (N) with liquefied center |
infiltration in a groin lymph node with |
lymph node (N) in bacterial lymphadenitis. |
invaded by cancer of the esophagus. |
hyperand hypoechoic parts in chronic |
|
|
lymphocytic leukemia (regressive |
|
|
changes). |
|
|
m Enlarged lymph node (L) with focal hy- |
n Large plaquelike calcification in medul- |
o CLL. Focal hyperechoic masses in an |
poechoic lesions; beginning lymph node |
lary thyroid carcinoma. |
enlarged lymph node (LK) invaded by |
metastasis in malignant melanoma. The |
|
lymphoma. The pathomorphological |
hypoechoic regions correspond with the |
|
substrate of the hyperechoic lesion re- |
malignant tissue (TU). |
|
mains unclear. Color-flow Doppler scan- |
|
|
ning shows a hypervascularization of the |
|
|
lymph node. |
6
Lymph Nodes
237

6
Lymph Nodes
 6.3 Lymph node with Changed Vascularization Visualized by Power Doppler Sonography
6.3 Lymph node with Changed Vascularization Visualized by Power Doppler Sonography
a and b Reactive lymph node enlarge- |
b On color-flow Doppler scanning hilar |
c and d Reactive lymph node enlargement |
d Color-flow Doppler scanning demon- |
ment. |
flow signal (1) and regular intraparenchy- |
(resolved during follow-up). |
strates increased vascularization. 1 = hilar |
a Small ovoid hypoechoic lymph node, |
mal vascular branches (2, 3). |
c Enlarged lymph node in the upper jug- |
vessel, 2 = longitudinal vessel, 3 = intra- |
diameter approximately 15 mm. |
|
ular region, hilar sign present. |
parenchymal vascular branches. |
e and f Infectious mononucleosis confirmed by serology. Enlarged lymph node.
i and j Reactive lymph node enlargement in autoimmune vasculitis.
i Lymph node enlargement.
m and n Gastric cancer.
m Lymph nodes invaded by tumor.
f Color-flow Doppler scanning demonstrates hypervascularization in reactive lymph node enlargement.
j On color-flow Doppler scanning marked vascularization with demonstration of hilar (1), longitudinal (2), and peripheral vessels
(3).
n On color-flow Doppler scanning subcapsular (1) and aberrant (2) vessels.
g and h Cancer of the prostate. g Lymph node metastasis.
k and l Hodgkin disease.
k Persistent lymph node enlargement after chemotherapy.
h Hypervascularization on color-flow Doppler scanning.
l On color-flow Doppler scanning no flow signals within the lymph node parenchyma. The nodular enlargement was regarded as being reactive.
Apart from the intranodal vascular bed visualized on ultrasound in peripheral lymph nodes, vascular displacement, aberrant vessels, focal lack of vessels, subcapsular vessels, and so-called “paint spots” all point more toward
malignant |
lymphadenopathy |
(Fig. 6.6, |
6.3 m,n).8 |
Enlarged reactive |
lymph nodes |
tend to be characterized by more horizontal hilar or longitudinal parenchymal vessels as well as treelike branching.
Flow parameters, such as the resistance indices measured in the lymph node arteries (resistance index, RI; pulsatility index, PI), seem to offer some benefit in the differential
diagnosis of enlarged lymph nodes. A large resistance index (RI >85) is considered indicative of a lymph node invaded by metastasis.
The vascularization of lymph nodes can be assessed by contrast-enhanced ultrasound (CEUS). This is mainly the case for abdominal lymph nodes. The significance of CEUS in the evaluation of malignancy of lymph nodes remains to be seen. Since nonvascularized areas are clearly delineated in CEUS, this method of examination might be of significance in assessing vital from nonvital lymph nodes ( 6.4a–i).
6.4a–i).
Abdominal lymph nodes. Compared with the peripheral lymph nodes, sonographic parameters of the vascularization in abdominal lymph nodes are difficult to obtain. Because they are located far away from the probe, B-mode imaging yields less detailed sonographic resolution of the parenchymal structures; this implies that any differential diagnosis has to rely primarily on location and morphology, with structural parameters being relegated to a minor role. The above criteria for the diagnosis of possible malignancy in peripheral lymph nodes have been confirmed far better than for abdominal lymph nodes, since their sonographic
238
 6.4 Lymph Node with Changed Vascularization Visualized by CEUS
6.4 Lymph Node with Changed Vascularization Visualized by CEUS
Hodgkin disease
a–c Patient with cervical lymphoma in Hodgkin disease; B-scan sonography shows enlarged lymph nodes, CEUS shows a heightened vascularization of the cervical lymph node.
Tuberculosis
d and e Patient with abdominal lymph node tuberculosis after chemotherapy. A grayscale image shows a homogeneous hypoechoic mesenteric lymph node. CEUS shows a inhomogeneous vascularization with areas without absorption of contrast agent.
Testicular germ cell tumor
f and g Patient with testicular germ cell tumor after chemotherapy. B-scan sonography shows an inhomogeneous hyperechoic paraaortal lymph node. CEUS shows a inhomogeneous vascularization. Areas free of contrast agent are indications of a still vital lymph node.
h and i Patient with testicular gem cell tumor after first cycle of chemotherapy. B-scan sonography shows a big homogenous para-aortal lymph node. CEUS shows no signs of contrast agent absorptions indicating a response to chemotherapy.
pattern of involvement and the ultrasound visualization differ substantially from those of the peripheral lymph nodes. The one important factor in abdominal lymph nodes is their size. In most cases it becomes impossible to demonstrate the hilum, and thus in many patients selective visualization of the cortex is also not feasible.
Ultrasound distinguishes four basic patterns in the invasion of abdominal lymph nodes (Fig. 6.7). The diffuse speckled micronodular invasion (primarily of the mesenteric lymph nodes) is quite different from the focal enlargement of lymph nodes (micronodular and macronodular). The “bulky” formation is characterized by a large mass of tumor, which may rep-
resent confluent lymphomas, or excessive growth of individual lymph nodes (Fig. 6.8).
Ultrasonography of abdominal lymph nodes has the following disadvantages:
●Only enlarged lymph nodes will be detected.
●Specificity is rather low in lumbar and iliac lymph nodes.
6
Lymph Nodes
239

6
Lymph Nodes
●Intestinal gas can limit the usefulness of the study.
●Misdiagnosis is possible in solitary lymphomas.
●It requires substantial experience of the operator in terms of technique and assessment.
Fig. 6.6 Schematic illustration of various vascular patterns in peripheral lymph nodes on color-flow Doppler scanning (from Schmidt and Görg8).
Fig. 6.7 Schematic illustration of the different patterns of invasion in abdominal lymph nodes around the celiac axis.
Fig. 6.8 Different patterns of invasion in abdominal lymph nodes (L) around the celiac axis.
a Individual enlarged lymph nodes in hepatitis. AH = hepatic artery; AL = splenic artery; TR = celiac axis.
b Plaquelike micronodular lymph node metastasis in gastric cancer (TU). AO = aorta.
c Macronodular lymph node invasion in a malignant lymphoma.
d Homogeneously confluent lymph node masses (L; arrows) in a malignant lymphoma. VC = vena cava, AO = aorta; TR = celiac trunk.
240
