
- •Contents
- •Preface
- •Contributors
- •1 Vessels
- •1.1 Aorta, Vena Cava, and Peripheral Vessels
- •Aorta, Arteries
- •Anomalies and Variant Positions
- •Dilatation
- •Stenosis
- •Wall Thickening
- •Intraluminal Mass
- •Perivascular Mass
- •Vena Cava, Veins
- •Anomalies
- •Dilatation
- •Intraluminal Mass
- •Compression, Infiltration
- •1.2 Portal Vein and Its Tributaries
- •Enlarged Lumen Diameter
- •Portal Hypertension
- •Intraluminal Mass
- •Thrombosis
- •Tumor
- •2 Liver
- •Enlarged Liver
- •Small Liver
- •Homogeneous Hypoechoic Texture
- •Homogeneous Hyperechoic Texture
- •Regionally Inhomogeneous Texture
- •Diffuse Inhomogeneous Texture
- •Anechoic Masses
- •Hypoechoic Masses
- •Isoechoic Masses
- •Hyperechoic Masses
- •Echogenic Masses
- •Irregular Masses
- •Differential Diagnosis of Focal Lesions
- •Diagnostic Methods
- •Suspected Diagnosis
- •3 Biliary Tree and Gallbladder
- •3.1 Biliary Tree
- •Thickening of the Bile Duct Wall
- •Localized and Diffuse
- •Bile Duct Rarefaction
- •Localized and Diffuse
- •Bile Duct Dilatation and Intraductal Pressure
- •Intrahepatic
- •Hilar and Prepancreatic
- •Intrapancreatic
- •Papillary
- •Abnormal Intraluminal Bile Duct Findings
- •Foreign Body
- •The Seven Most Important Questions
- •3.2 Gallbladder
- •Changes in Size
- •Large Gallbladder
- •Small/Missing Gallbladder
- •Wall Changes
- •General Hypoechogenicity
- •General Hyperechogenicity
- •General Tumor
- •Focal Tumor
- •Intraluminal Changes
- •Hyperechoic
- •Hypoechoic
- •Nonvisualized Gallbladder
- •Missing Gallbladder
- •Obscured Gallbladder
- •4 Pancreas
- •Diffuse Pancreatic Change
- •Large Pancreas
- •Small Pancreas
- •Hypoechoic Texture
- •Hyperechoic Texture
- •Focal Changes
- •Anechoic Lesion
- •Hypoechoic Lesion
- •Isoechoic Lesion
- •Hyperechoic Lesion
- •Irregular (Complex Structured) Lesion
- •Dilatation of the Pancreatic Duct
- •Marginal/Mild Dilatation
- •Marked Dilatation
- •5 Spleen
- •Nonfocal Changes of the Spleen
- •Diffuse Parenchymal Changes
- •Large Spleen
- •Small Spleen
- •Focal Changes of the Spleen
- •Anechoic Mass
- •Hypoechoic Mass
- •Hyperechoic Mass
- •Splenic Calcification
- •6 Lymph Nodes
- •Peripheral Lymph Nodes
- •Head/Neck
- •Extremities (Axilla, Groin)
- •Abdominal Lymph Nodes
- •Porta Hepatis
- •Splenic Hilum
- •Mesentery (Celiac, Upper and Lower Mesenteric Station)
- •Stomach
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •Small/Large Intestine
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •8 Peritoneal Cavity
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Wall Structures
- •Smooth Margin
- •Irregular Margin
- •Intragastric Processes
- •Intraintestinal Processes
- •9 Kidneys
- •Anomalies, Malformations
- •Aplasia, Hypoplasia
- •Cystic Malformation
- •Anomalies of Number, Position, or Rotation
- •Fusion Anomaly
- •Anomalies of the Renal Calices
- •Vascular Anomaly
- •Diffuse Changes
- •Large Kidneys
- •Small Kidneys
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Irregular Structure
- •Circumscribed Changes
- •Anechoic Structure
- •Hypoechoic or Isoechoic Structure
- •Complex Structure
- •Hyperechoic Structure
- •10 Adrenal Glands
- •Enlargement
- •Anechoic Structure
- •Hypoechoic Structure
- •Complex Echo Structure
- •Hyperechoic Structure
- •11 Urinary Tract
- •Malformations
- •Duplication Anomalies
- •Dilatations and Stenoses
- •Dilated Renal Pelvis and Ureter
- •Anechoic
- •Hypoechoic
- •Hypoechoic
- •Hyperechoic
- •Large Bladder
- •Small Bladder
- •Altered Bladder Shape
- •Intracavitary Mass
- •Hypoechoic
- •Hyperechoic
- •Echogenic
- •Wall Changes
- •Diffuse Wall Thickening
- •Circumscribed Wall Thickening
- •Concavities and Convexities
- •12.1 The Prostate
- •Enlarged Prostate
- •Regular
- •Irregular
- •Small Prostate
- •Regular
- •Echogenic
- •Circumscribed Lesion
- •Anechoic
- •Hypoechoic
- •Echogenic
- •12.2 Seminal Vesicles
- •Diffuse Change
- •Hypoechoic
- •Circumscribed Change
- •Anechoic
- •Echogenic
- •Irregular
- •12.3 Testis, Epididymis
- •Diffuse Change
- •Enlargement
- •Decreased Size
- •Circumscribed Lesion
- •Anechoic or Hypoechoic
- •Irregular/Echogenic
- •Epididymal Lesion
- •Anechoic
- •Hypoechoic
- •Intrascrotal Mass
- •Anechoic or Hypoechoic
- •Echogenic
- •13 Female Genital Tract
- •Masses
- •Abnormalities of Size or Shape
- •Uterus
- •Abnormalities of Size or Shape
- •Myometrial Changes
- •Intracavitary Changes
- •Endometrial Changes
- •Fallopian Tubes
- •Hypoechoic Mass
- •Anechoic Cystic Mass
- •Solid Echogenic or Nonhomogeneous Mass
- •14 Thyroid Gland
- •Diffuse Changes
- •Enlarged Thyroid Gland
- •Small Thyroid Gland
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Circumscribed Changes
- •Anechoic
- •Hypoechoic
- •Isoechoic
- •Hyperechoic
- •Irregular
- •Differential Diagnosis of Hyperthyroidism
- •Types of Autonomy
- •15 Pleura and Chest Wall
- •Chest Wall
- •Masses
- •Parietal Pleura
- •Nodular Masses
- •Diffuse Pleural Thickening
- •Pleural Effusion
- •Anechoic Effusion
- •Echogenic Effusion
- •Complex Effusion
- •16 Lung
- •Masses
- •Anechoic Masses
- •Hypoechoic Masses
- •Complex Masses
- •Index
13
Female Genital Tract
Fig. 13.71 Cervical carcinoma, transverse scan; markedly enlarged and nonhomogeneous cervix (CE).
Complications. Ultrasound can detect the following complications due to tumor invasion:
●Rectal stenosis with bowel obstruction
●Ureteral obstruction with hydroureter and hydronephrosis
Fig. 13.72 Cervical carcinoma, hypoechoic (arrows) spreading to the uterine corpus and vagina (arrows). Ulcerated tumor with free fluid (FL). UT = uterus; BL = bladder.
Other complications may arise:
●Infiltration of the sciatic nerve, causing sciatica
●Obstruction of the iliac vessels with inflow stasis of the vulva and lower extremities
●Infections, septic pyonephritis, and uremia
Fig. 13.73 Cervical carcinoma (stage IIIB, following irradiation and cisplatin chemotherapy): hypoechoic broadening of the cervix with tapered posterior margin due to tumor infiltration.
■ Fallopian Tubes
The fallopian tubes can be identified at their origin from the uterus, to the right and left of the uterine fundus. Their further course can be visualized mainly if pathological changes are present (Figs. 13.74, 13.75, 13.76).
Fig. 13.74 The fallopian tubes (arrows) branch off laterally from the hypoechoic uterus. Bowel gas (D) may reduce the visibility.
Fig. 13.75 Uterine fundus and fallopian tube (T) cranial to the bladder (BL); transverse scan. M = myometrium, E = endometrium.
Fig. 13.76 Tortuous right fallopian tube (T), lateral to the uterus. M = myometrium; E = endometrium.
Hypoechoic Mass
Tract |
Vagina |
||||
|
|
|
|||
|
|
|
Uterus |
||
|
|
|
|||
|
|
|
Fallopian Tubes |
||
GenitalFemale |
|||||
|
Hypoechoic Mass |
||||
|
|
|
|
||
|
|
|
Ovaries |
||
|
|
|
|||
Sactosalpinx, Hematosalpinx, Pyosalpinx
Tubal Carcinoma
Benign Tumors
Tubal Pregnancy
458
Sactosalpinx, Hematosalpinx, Pyosalpinx 





































Definitions. Sactosalpinx is a hypoechoic mass between the uterus and ovary caused by a fluid collection in the fallopian tube due to various causes (Fig.13.77).
Hematosalpinx is a collection of blood extending into the fallopian tube, e. g., due to hymenal atresia.
Pyosalpinx is a collection of pus secondary to an inflammatory stenosis.
Sonographic features. The tubal wall appears sonographically as a pair of echogenic lines. With an abscess, the wall shows echogenic thickening (Fig.13.78). The fallopian tube runs a tortuous course. When it contains a collection of clear fluid, the lumen is dilated and anechoic. With blood or pus in the tube, the lumen may appear echogenic and nonhomogeneous (Fig.13.79). An infection of the fallopian tube often encroaches on the ovaries by direct continuation. The inflamed organs (tube/ovaries) generally cannot be sufficiently differentiated.
Differentiation. Color duplex examination can differentiate a fallopian tube from varicose vessels. Bowel loops generally have a hypoechoic wall that distinguishes them from the fallopian tube. Malignant masses tend to be painless, whereas inflammatory processes cause severe pain. Tubo-ovarian cysts are not true neoplasms but residua from pelvic inflammatory disease.
13
Fallopian Tubes
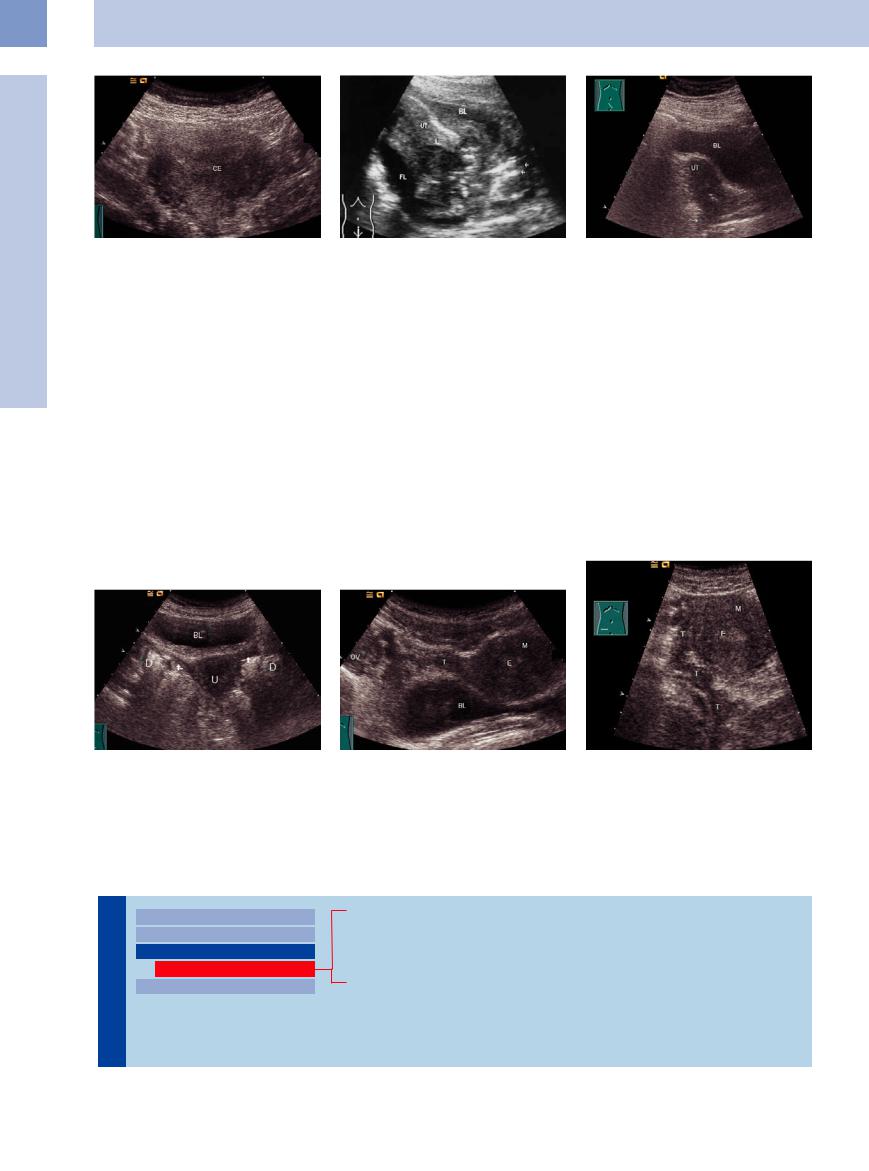
Fig. 13.77 Saktosalpinx; enlarged anechoic fallopian tubes
(T) lateral to the uterus (UT).
Fig. 13.78 Tubo-ovarian abscess, transvaginal ultrasound, lower abdominal pain. The uterus (UT) is homogeneous and smoothly margined, the regular tube (T) coming out of the uterus is easily detectable. The tube is ballooned in its course.
Fig. 13.79 Tubo-ovarian abscess. Transvaginal scan shows a hypoechoic mass with ill-defined margins (49.8 mm × 27 mm) in the left adnexa. UT = uterus.
Tubal Carcinoma
Only 0.3% of gynecological cancers are primary malignant tumors of the fallopian tube. The most common type is adenocarcinoma. Patients present clinically with pain on the affected side, discharge (50%), and bleeding (25%). The periodic drainage of large fluid volumes that is commonly seen with tubal carcinoma is termed hydrops tubae profluens. Thus, vaginal discharge assumes special significance
in postmenopausal women and should always be investigated. Tumor cells are not found in material obtained by dilatation and curettage (D&C), but the cytological examination of cervical secretions will reveal cancer cells. Given the copious lymphatic drainage of the fallopian tube region, metastases from tubal carcinoma are frequent. The prognosis is the same as for ovarian carcinoma.
Sonographic features. Tubal carcinomas are unilateral, round-to-oval lesions with a hypoechoic to hyperechoic appearance. The tube is often dilated distally and is tender to manual pressure. In its advanced stages, the tumor has the same appearance as ovarian carcinoma.
Benign Tumors
Benign tumors of the fallopian tubes are very |
● Myomas |
● Dermoids |
||
rare. The following types occur: |
● |
Fibromas |
● |
Lymphangiomas |
|
● |
Lipomas |
● |
Adenomas |
Tubal Pregnancy




























More than 90% of ectopic pregnancies are tubal pregnancies. The affected tube appears thickened and hypoechoic at transabdominal ultrasound (Fig.13.80). Transvaginal sonography is definitely superior to transabdominal scanning in the diagnosis of tubal pregnancy.
With a ruptured tubal pregnancy, ultrasound will generally detect free fluid in the abdomen (Fig.13.81).
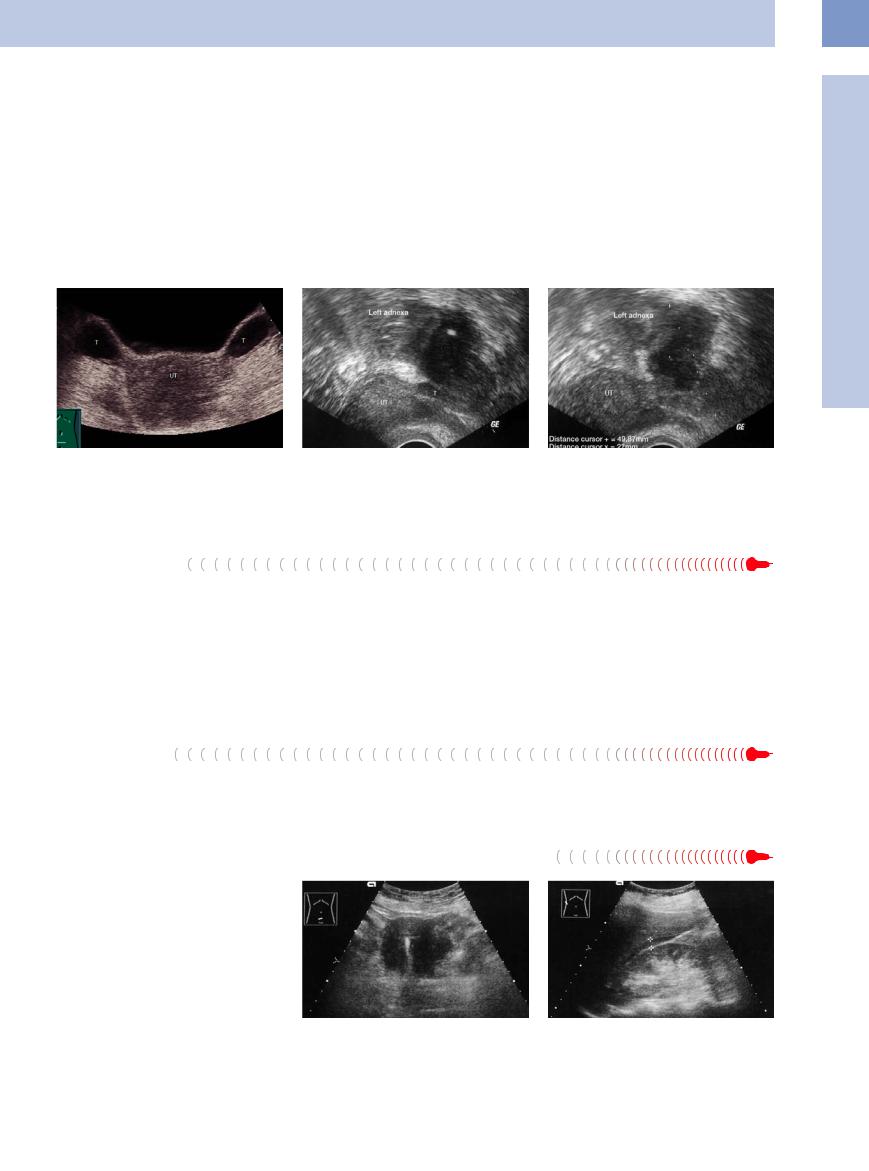
Fig. 13.80 Tubal pregnancy: thickened, hypoechoic fallopian tube with an IUD in place (high-level echo in the uterine cavity).
Fig. 13.81 Tubal rupture: free fluid (cursors) in the hepatorenal sinus following a tubal rupture due to ectopic pregnancy.
459
13
Female Genital Tract
■ Ovaries
The ovaries undergo both structural and func- |
Due to biological heterogeneity the ovaries |
tional changes in response to gonadotropin |
are highly variable in size, shape, echogenicity, |
production and aging. The sex hormones are |
and position. They are often difficult to visual- |
synthesized in the ovaries. |
ize with ultrasound; transvaginal sonography |
Anatomy and Histology
and Histology





The ovaries are small, firm, almond-shaped organs lying in the lesser pelvis. They are surrounded by a thin fibrous capsule (tunica albuginea). Their surface is smooth initially but becomes irregular over time owing to the pitting and scarring that result from follicular maturation and degeneration. The ovaries become scarred and atrophic after menopause.
The follicles (primordial follicles, primary and secondary follicles) are located in the very cellular cortical zone of the ovary, along
with the corpora lutea and corpora albicantia. The cortical zone is continuous with the smaller medullary zone, with no distinct boundary between the two. The medullary zone is composed of connective tissue with occasional smooth muscle cells and vessels.
Most of the ovary is intraperitoneal. Vessels enter the ovary at the hilum, which is extraperitoneal. The ovary is supplied by the ovarian arteries, which arise directly from the aorta below the renal artery, and by the ovarian
is the best available imaging technique. Diseases of the ovaries are often detected at
an extremely late stage owing to the delayed onset of symptoms.
branch of the uterine artery. The left ovarian artery occasionally arises from the left renal artery. The blood vessels run to the hilum in the ovarian suspensory ligament.
Arborizing valveless veins form a network, the pampiniform plexus (ovarian venous plexus), that surrounds the organs. The right ovarian vein opens directly into the vena cava, while the left ovarian vein drains into the left renal vein.
Topography 





















































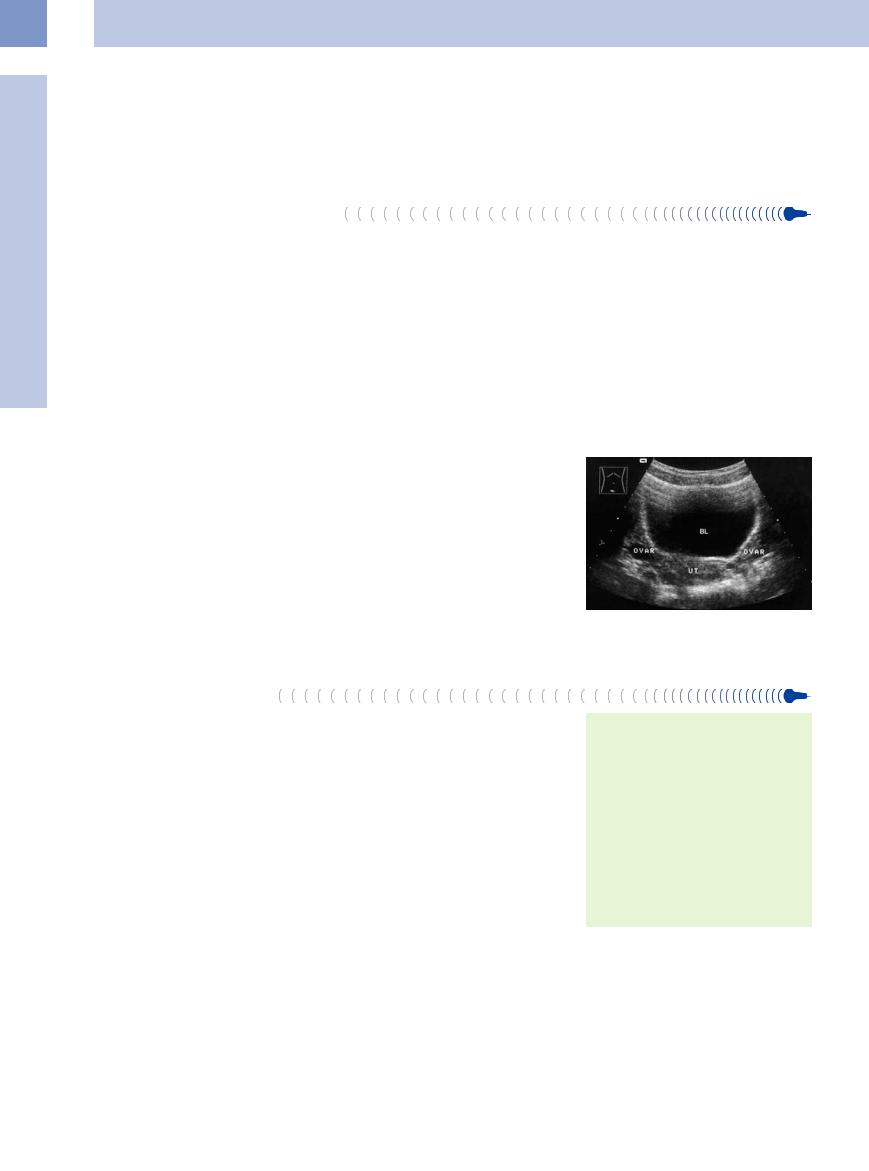
The ovaries of the nullipara lie in a small groove (ovarian fossa) in the lateral pelvic wall on both sides of the uterus. They are located anterior to the ureter in the bifurcation of the common iliac artery (Fig.13.82).
The ovaries are attached to the pelvic wall by the ovarian suspensory ligament and to the cornual area of the uterus by the proper ovar-
ian ligament. They are attached posteriorly to the mesovarium by the broad ligament. During pregnancy, the enlarged uterus displaces the ovaries into the abdomen. After pregnancy, the ligaments are so stretched that the ovaries become mobile and can vary greatly in their position.
Fig. 13.82 Ovaries (Ovar) flanking the uterus (UT) posterior to the bladder (BL) in a nullipara.
Ultrasound Morphology
Morphology
The ovaries can usually be visualized in women of reproductive age (Fig.13.83). Both ovaries can be identified in approximately 80% of cases. The ovaries can rarely be visualized after menopause, when they are smaller and less echogenic.
On ultrasound the ovaries in fertile women measure approximately 2 × 3 × 1.5 cm,3 which corresponds closely to the anatomic dimensions of 2.5 cm long, 2 cm wide, and 1 cm thick.2 They may be slightly less echogenic than the uterus and display anechoic cystic changes during the ovarian cycle.
The ovarian cortex initially contains several rounded, anechoic masses up to 10 mm in diameter. Approximately 7 days after menstruation, one dominant follicle appears while the others regress. The dominant follicle grows to a graafian follicle by the time of ovulation, measuring approximately 2 cm in size and projecting above the organ surface. An echogenic ringlike structure, the cumulus oophorus, appears in the follicle. After ovulation, free fluid can be demonstrated in the cul-de-sac. Now the follicle is misshapen or no longer visualized. When bleeding occurs in the corpus luteum, the follicle becomes uniformly hyperechoic.
Size and shape
●2 × 3 × 1.5 cm
●Almond-shaped
Structure
●Medullary zone
●Cortical zone with follicles, corpora lutea, and corpora albicantia
●Capsule (tunica albuginea)
Landmarks
●Bladder
●Uterus
●Pelvic wall
●Iliac vessels
460
Anechoic Cystic Mass
Tract |
Vagina |
||||
|
|
|
|||
|
|
|
Uterus |
||
|
|
|
|||
|
|
|
Fallopian Tubes |
||
Genital |
|||||
Ovaries |
|||||
|
|
|
|||
|
|
|
|
Anechoic Cystic Mass |
|
|
|
|
|
||
Female |
|
Solid Echogenic or |
|||
|
Nonhomogeneous Mass |
||||
|
|
|
|
||
Simple Follicles
Functional Cysts (Follicular and Corpus Luteum Cysts)
Theca-Lutein Cyst
Paraovarian Cyst
Polycystic Ovaries (PCO Syndrome, Stein–Leventhal Syndrome)
Cystic Ovarian Tumors
Simple Follicles 



















































Simple follicles appear as unilocular, anechoic |
approximately 2 cm and projects above the |
masses with sharply circumscribed margins. |
ovarian surface. The cumulus oophorus is visi- |
When intraluminal hemorrhage is present, |
ble as a ringlike structure of high echogenicity. |
they appear hyperechoic. Simple follicles re- |
After ovulation, the follicle normally appears |
gress spontaneously. |
distorted in its shape or is no longer visible |
The dominant follicle grows to a graafian |
(Figs. 13.83, 13.84, 13.85). |
follicle by the time of ovulation. It enlarges to |
|
13
Ovaries
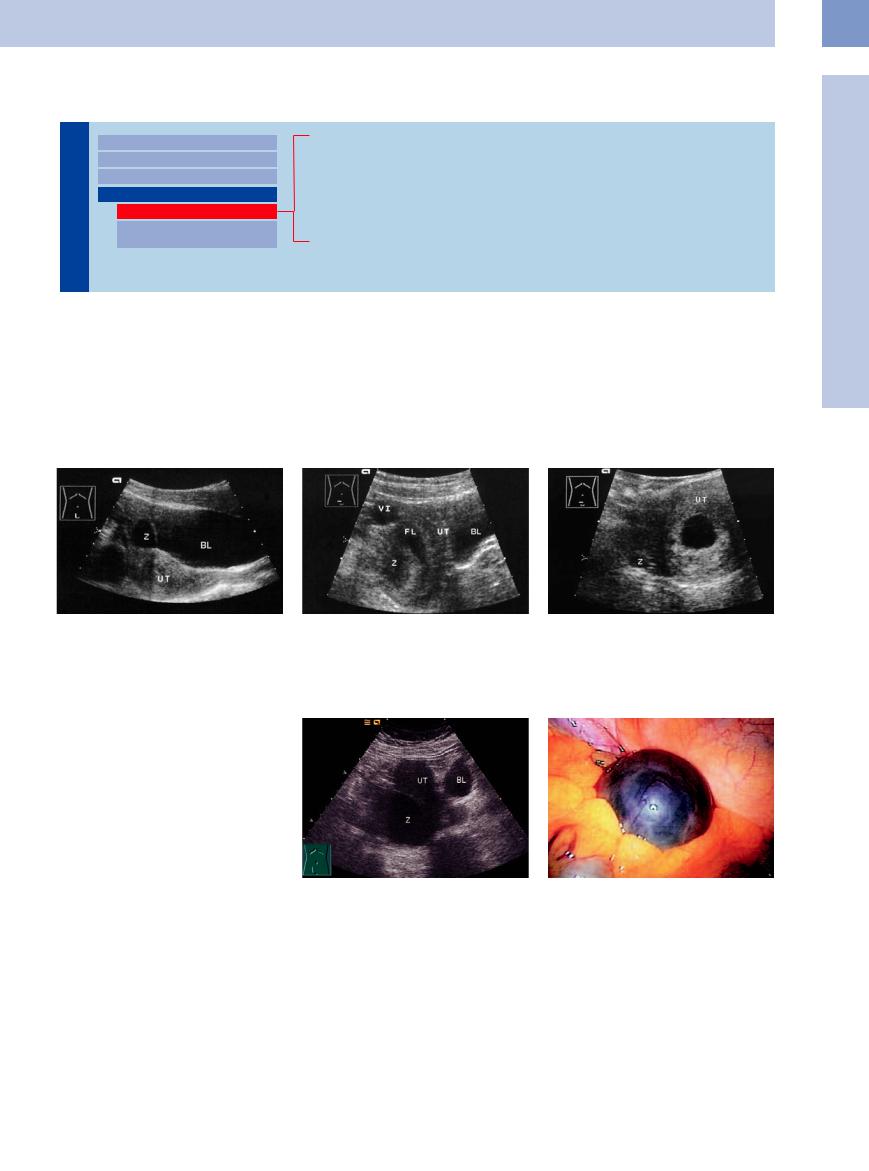
Fig. 13.83 Mature graafian follicle. BL = bladder; UT = uterus; C = follicle appearing as a cystic mass.
Fig. 13.84 Follicular rupture. Residual follicle (C); FL = free fluid; VI = iliac vein; UT = uterus; BL = bladder.
Fig. 13.85 Persistent follicle: early pregnancy with a chorionic cavity in the uterus (UT) and a follicle (C) in the ovary.
Functional  Cysts
Cysts (Follicular
(Follicular  and
and Corpus Luteum
Corpus Luteum Cysts)
Cysts) 






























Most ovarian cysts are “functional cysts” such as follicular and corpus luteum cysts. Functional cysts are the response of the ovaries to elevated gonadotropin levels (FSH and LH) in the blood. A cyst no larger than 3–6 cm in a woman under the age of 30 may be a functional cyst. If it does not regress by the next menstruation or grows even larger, it should be surgically removed.
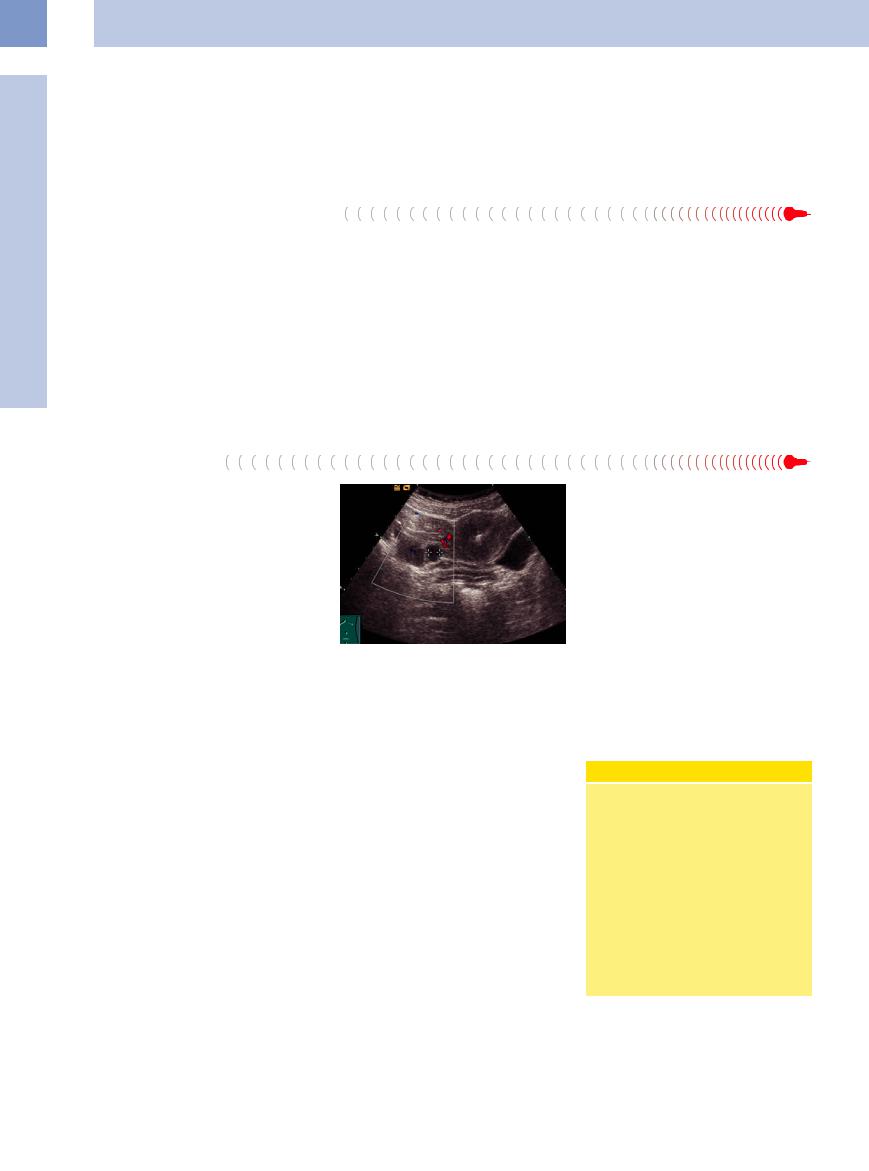
Follicular cyst. A follicular cyst is the most common ovarian cyst and can occur at any age. It is common after menarche and in the climacteric and is rare after menopause. A follicular cyst develops in response to estrogen stimulation when ovulation fails to occur. Generally the cyst is detected incidentally, but patients occasionally present with abdominal pain, a palpable mass, or signs of estrogen activity. The cysts may rupture when palpated, which can lead to hemorrhage, peritoneal shock, and spillage of the cyst contents into the abdominal cavity (Fig. 13.86, Fig.13.87).
Fig. 13.86 Hemorrhagic ovarian cyst (Z): painful hypoechoic to cystic mass lateral to the uterus (UT). BL = bladder.
The sonographic criteria of a follicular cyst are as follows:
● Thin-walled
● Usually no larger than 5 cm (maximum
8 cm)
●Solitary, rarely multiple
●Unilateral, rarely bilateral
●Contains clear watery fluid, rarely hemorrhagic
Fig. 13.87 Hemorrhagic ovarian cyst: laparoscopy demonstrates a balloonlike red-blue mass in the lower pelvis.
Retention cysts. Retention cysts are formed by the accumulation of fluids such as blood and secretions in follicular cysts. Unlike cytomas, they grow no larger than fist-size.
Corpus luteum cyst. A corpus luteum cyst results from cystic enlargement of the corpus luteum. It develops when hemorrhage occurs within the corpus luteum and the blood is
461
13
Female Genital Tract
reabsorbed, leaving a cavity that contains se- |
pus luteum cyst is often present during preg- |
rous fluid. Corpus luteum cysts are rarer than |
nancy and regresses spontaneously. |
follicular cysts. |
The sonographic criteria of a corpus luteum |
Most patients are clinically asymptomatic or |
cyst are as follows: |
present with irregular menstrual periods due |
● Unilateral |
to the overproduction of progesterone. A cor- |
● Up to 5 cm in diameter |
|
● Anechoic to echogenic contents |
Theca-Lutein Cyst
Cyst 








Lutein cysts develop from atretic follicles. They represent follicular cysts that have theca-lutein cells on their inner surface. Lutein cysts result from hyperstimulation by gonadotropins and therefore occur in diseases and conditions that are associated with increased α-hCG formation, i. e., in 20% of molar pregnancies, in chorioepithelioma patients, and in multiple pregnancies. For this reason they are also commonly seen in patients with polycystic ovaries and in women treated for infertility. They may contain progesterone in high doses and regress
after the cause is eliminated, although this may take several months.
Patients may become symptomatic owing to complications such as cyst torsion, intracystic hemorrhage, and rupture with hemorrhage. Ruptures and infarctions are an indication for operative treatment.
Sonographic features. Theca-lutein cysts can range from 6 to 20 cm in size.8 A single cyst is rarely larger than a lemon, but because lutein cysts tend to be multiple and bilateral, the ovaries may be substantially enlarged. The
cyst wall resembles an abscess membrane. The ovarian parenchyma is edematous. The cysts rupture easily, and pedunculated cysts often undergo torsion. Ascites may occur.
The sonographic criteria of a theca-lutein cyst are:
●Multiple
●Bilateral
●Thin-walled
●Anechoic contents
●Echogenic with intracystic hemorrhage
●Moderate to massive ovarian enlargement
Paraovarian Cyst
Paraovarian cysts arise from fetal tissues bordering the ovaries (epoöphoron, vestiges of the caudal mesonephric duct in the mesovary). They are more accurately classified as retention cysts rather than true neoplasms. They arise in the mesovary, are invariably benign, and occur at any age. Paraovarian cysts are variable in size; they may be pedunculated, and those with long pedicles tend to undergo torsion (Fig.13.88).
Their sonographic criteria are as follows:
●Elliptical shape
●Intraligamentous
●Well delineated from the ovary, may be pedunculated
Fig. 13.88 Paraovarian cyst: near-anechoic mass (cursors) between uterus (UT) and ovary (OV); BL = bladder.
Polycystic Ovaries (PCO
(PCO Syndrome, Stein–
Syndrome, Stein– Leventhal Syndrome)
Leventhal Syndrome)

























PCO syndrome develops during puberty as a result of increased androgen production by the adrenal cortex following adrenarche. It affects up to 7% of the female population. The initiating cause is poorly understood.
Stein–Leventhal syndrome develops when the hyperandrogenemia9 gives rise to a symptom complex of polycystic ovaries, hirsutism, menstrual abnormalities, and obesity.
The following are additional symptoms of PCO:
●Sterility and infertility (74%)
●Hirsutism (50–70%)
●Amenorrhea and oligomenorrhea (approximately 50%)
●Dysfunctional bleeding (approximately 30%)
●Overweight (40%)
The estrogen stimulation of the endometrium induces hyperplasia and can also lead to welldifferentiated endometrial carcinoma.
Sonographic features. The ovaries are generally larger than the uterine corpus. The smooth tunica albuginea shows echogenic thickening, and the cortex contains anechoic, usually equal-sized follicles up to 5 mm in diameter (rarely >1 cm) arranged in a circumferential pattern. Follicular maturation does not occur. Since ovulation is absent, there is no surface pitting and no corpora albicantia.
The sonographic criteria are as follows (Fig.13.89, Fig.13.90):
●Bilateral occurrence
●Enlarged ovaries
●Hyperechoic center
●Multiple cysts (anechoic follicles up to 5 mm in the cortex)
●Endometrial hyperplasia usually present
Pathogenesis of PCO Syndrome
PCO syndrome is characterized by a vicious cycle in which a stressful situation with increased ACTH secretion stimulates the formation of androstenedione in the adrenal cortex. This hormone is metabolized in the fat to estrone and sensitizes the pituitary to LHRH. The result is excessive LH secretion (FSH is unaffected), causing a nonphysiological stimulation of the ovaries and increased androgen production (androstenedione) in the theca follicles and stromal cells. The androgen is again converted to estrone in the fatty tissue, and the cycle continues.
462
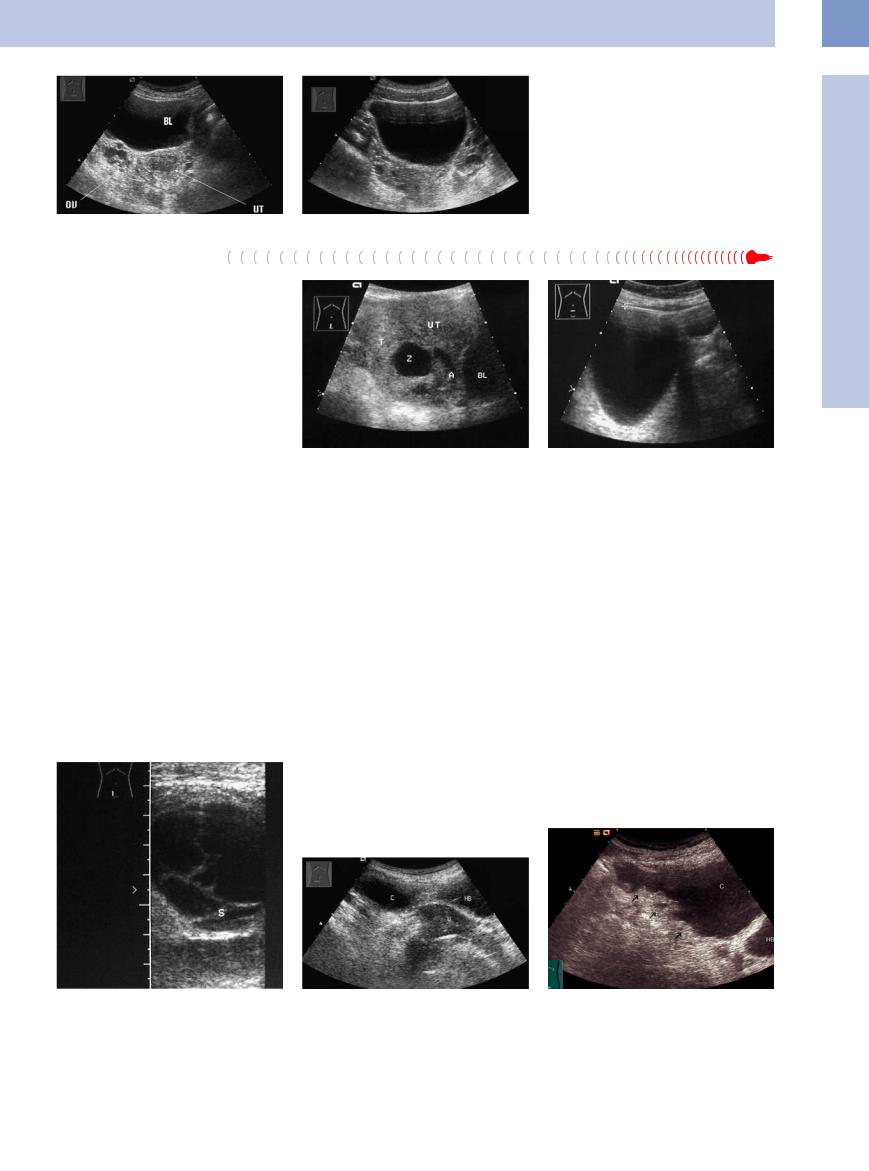
Fig. 13.89 Polycystic ovary (OV) on the right side. Thickened endometrium in the uterus (UT). BL = bladder.
f Fig. 13.90 Bilateral polycystic ovaries in menarche.
Cystic Ovarian Tumors
Tumors
These lesions include benign cystadenoma, mucinous cystoma (which has a high malignant potential), and cystadenocarcinoma.
Cystadenoma. Benign cystadenoma (serous cystadenoma) is the most common benign ovarian tumor. It corresponds histologically to a cystoma serosum simplex. The wall of the cystic epithelial tumor is thin, smooth, and lined with epithelium. Cystadenoma is usually unifocal and rarely multifocal. If bilateral cystic tumors are present, they are very likely to be
malignant (65–75% of carcinomas are bilat- |
Fig. 13.91 Cystadenoma: anechoic mass (Z) in the en- |
Fig. 13.92 Serous |
cystadenoma: ovarian |
cyst (approxi- |
eral). A cystadenoma may have serous contents |
larged ovary (T). A = ascites; UT = uterus; BL = bladder. |
mately anechoic mass) in menopause, 10 cm in diameter. |
||
(similar to the fallopian tubes) or mucinous |
|
|
|
|
contents (similar to the endocervix), depend- |
|
|
|
|
ing on the type of epithelium and the tumor |
nosed. Mucinous tumors are rarely bilateral |
Cystadenocarcinoma. Serous cystadenocarci- |
||
origin. The cysts are typically large and may fill |
(5% of benign tumors, 10% of borderline tu- |
noma is the most common malignant ovarian |
||
the entire abdomen. |
mors, and 20% of mucinous carcinomas are |
tumor, accounting for approximately one-third |
||
The sonographic criteria are as follows (Fig. |
bilateral). The older the patient, the greater |
of all ovarian cancers. The tumor is predomi- |
||
13.91, Fig.13.92): |
the likelihood that the tumor is malignant. |
nantly cystic, but unlike benign cystadenoma |
||
● Anechoic mass |
Mucinous cystomas are frequently septated, |
there are solid hyperechoic elements in the |
||
● Thin echogenic wall |
and their echogenicity can vary. The interior of |
cyst wall, which shows circumscribed thicken- |
||
● Thin septa, sometimes multilocular |
the septated areas may show a fine to coarse |
ing (Fig.13.94, Fig.13.95). Hyperechoic, some- |
||
● Smooth surface |
echogenic pattern. Needle aspiration is not ad- |
times polypoid structures may project into the |
||
Mucinous cystoma. Mucinous cystomas even- |
vised owing to the risk of tumor cell dissem- |
cyst lumen. |
Nonhomogeneous, |
hypoechoic |
ination. |
areas may also appear due to intratumoral |
|||
tually undergo malignant transformation and |
The sonographic criteria are as follows |
hemorrhage and necrosis. Stromal infiltration |
||
therefore should be removed without delay. |
(Fig.13.93): |
can be demonstrated histologically. |
||
They account for approximately 25% of all ovar- |
● Cystic |
|
|
|
ian tumors, and 10% are malignant when diag- |
● Multifocal, often septated |
|
|
|
|
● Thin-walled with no deposits |
|
|
|
13
Ovaries
Fig. 13.93 Mucinous ovarian cystoma appears as a cystic mass with multiple septa (S).
Fig. 13.94 Ovarian cyst (C), bladder (BL), uterus (U). The cyst wall should be scrutinized for local thickening (cystadenocarcinoma).
Fig. 13.95 Cystadenocarcinoma (C): circumscribed irregular hyperechoic thickening of the cyst wall (arrows).
463
