
- •Contents
- •Preface
- •Contributors
- •1 Vessels
- •1.1 Aorta, Vena Cava, and Peripheral Vessels
- •Aorta, Arteries
- •Anomalies and Variant Positions
- •Dilatation
- •Stenosis
- •Wall Thickening
- •Intraluminal Mass
- •Perivascular Mass
- •Vena Cava, Veins
- •Anomalies
- •Dilatation
- •Intraluminal Mass
- •Compression, Infiltration
- •1.2 Portal Vein and Its Tributaries
- •Enlarged Lumen Diameter
- •Portal Hypertension
- •Intraluminal Mass
- •Thrombosis
- •Tumor
- •2 Liver
- •Enlarged Liver
- •Small Liver
- •Homogeneous Hypoechoic Texture
- •Homogeneous Hyperechoic Texture
- •Regionally Inhomogeneous Texture
- •Diffuse Inhomogeneous Texture
- •Anechoic Masses
- •Hypoechoic Masses
- •Isoechoic Masses
- •Hyperechoic Masses
- •Echogenic Masses
- •Irregular Masses
- •Differential Diagnosis of Focal Lesions
- •Diagnostic Methods
- •Suspected Diagnosis
- •3 Biliary Tree and Gallbladder
- •3.1 Biliary Tree
- •Thickening of the Bile Duct Wall
- •Localized and Diffuse
- •Bile Duct Rarefaction
- •Localized and Diffuse
- •Bile Duct Dilatation and Intraductal Pressure
- •Intrahepatic
- •Hilar and Prepancreatic
- •Intrapancreatic
- •Papillary
- •Abnormal Intraluminal Bile Duct Findings
- •Foreign Body
- •The Seven Most Important Questions
- •3.2 Gallbladder
- •Changes in Size
- •Large Gallbladder
- •Small/Missing Gallbladder
- •Wall Changes
- •General Hypoechogenicity
- •General Hyperechogenicity
- •General Tumor
- •Focal Tumor
- •Intraluminal Changes
- •Hyperechoic
- •Hypoechoic
- •Nonvisualized Gallbladder
- •Missing Gallbladder
- •Obscured Gallbladder
- •4 Pancreas
- •Diffuse Pancreatic Change
- •Large Pancreas
- •Small Pancreas
- •Hypoechoic Texture
- •Hyperechoic Texture
- •Focal Changes
- •Anechoic Lesion
- •Hypoechoic Lesion
- •Isoechoic Lesion
- •Hyperechoic Lesion
- •Irregular (Complex Structured) Lesion
- •Dilatation of the Pancreatic Duct
- •Marginal/Mild Dilatation
- •Marked Dilatation
- •5 Spleen
- •Nonfocal Changes of the Spleen
- •Diffuse Parenchymal Changes
- •Large Spleen
- •Small Spleen
- •Focal Changes of the Spleen
- •Anechoic Mass
- •Hypoechoic Mass
- •Hyperechoic Mass
- •Splenic Calcification
- •6 Lymph Nodes
- •Peripheral Lymph Nodes
- •Head/Neck
- •Extremities (Axilla, Groin)
- •Abdominal Lymph Nodes
- •Porta Hepatis
- •Splenic Hilum
- •Mesentery (Celiac, Upper and Lower Mesenteric Station)
- •Stomach
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •Small/Large Intestine
- •Focal Wall Changes
- •Extended Wall Changes
- •Dilated Lumen
- •Narrowed Lumen
- •8 Peritoneal Cavity
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Anechoic Structure
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Wall Structures
- •Smooth Margin
- •Irregular Margin
- •Intragastric Processes
- •Intraintestinal Processes
- •9 Kidneys
- •Anomalies, Malformations
- •Aplasia, Hypoplasia
- •Cystic Malformation
- •Anomalies of Number, Position, or Rotation
- •Fusion Anomaly
- •Anomalies of the Renal Calices
- •Vascular Anomaly
- •Diffuse Changes
- •Large Kidneys
- •Small Kidneys
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Irregular Structure
- •Circumscribed Changes
- •Anechoic Structure
- •Hypoechoic or Isoechoic Structure
- •Complex Structure
- •Hyperechoic Structure
- •10 Adrenal Glands
- •Enlargement
- •Anechoic Structure
- •Hypoechoic Structure
- •Complex Echo Structure
- •Hyperechoic Structure
- •11 Urinary Tract
- •Malformations
- •Duplication Anomalies
- •Dilatations and Stenoses
- •Dilated Renal Pelvis and Ureter
- •Anechoic
- •Hypoechoic
- •Hypoechoic
- •Hyperechoic
- •Large Bladder
- •Small Bladder
- •Altered Bladder Shape
- •Intracavitary Mass
- •Hypoechoic
- •Hyperechoic
- •Echogenic
- •Wall Changes
- •Diffuse Wall Thickening
- •Circumscribed Wall Thickening
- •Concavities and Convexities
- •12.1 The Prostate
- •Enlarged Prostate
- •Regular
- •Irregular
- •Small Prostate
- •Regular
- •Echogenic
- •Circumscribed Lesion
- •Anechoic
- •Hypoechoic
- •Echogenic
- •12.2 Seminal Vesicles
- •Diffuse Change
- •Hypoechoic
- •Circumscribed Change
- •Anechoic
- •Echogenic
- •Irregular
- •12.3 Testis, Epididymis
- •Diffuse Change
- •Enlargement
- •Decreased Size
- •Circumscribed Lesion
- •Anechoic or Hypoechoic
- •Irregular/Echogenic
- •Epididymal Lesion
- •Anechoic
- •Hypoechoic
- •Intrascrotal Mass
- •Anechoic or Hypoechoic
- •Echogenic
- •13 Female Genital Tract
- •Masses
- •Abnormalities of Size or Shape
- •Uterus
- •Abnormalities of Size or Shape
- •Myometrial Changes
- •Intracavitary Changes
- •Endometrial Changes
- •Fallopian Tubes
- •Hypoechoic Mass
- •Anechoic Cystic Mass
- •Solid Echogenic or Nonhomogeneous Mass
- •14 Thyroid Gland
- •Diffuse Changes
- •Enlarged Thyroid Gland
- •Small Thyroid Gland
- •Hypoechoic Structure
- •Hyperechoic Structure
- •Circumscribed Changes
- •Anechoic
- •Hypoechoic
- •Isoechoic
- •Hyperechoic
- •Irregular
- •Differential Diagnosis of Hyperthyroidism
- •Types of Autonomy
- •15 Pleura and Chest Wall
- •Chest Wall
- •Masses
- •Parietal Pleura
- •Nodular Masses
- •Diffuse Pleural Thickening
- •Pleural Effusion
- •Anechoic Effusion
- •Echogenic Effusion
- •Complex Effusion
- •16 Lung
- •Masses
- •Anechoic Masses
- •Hypoechoic Masses
- •Complex Masses
- •Index
■ Aorta, Arteries
Anomalies and Variant Positions
Vessels

Aorta, Arteries
Anomalies and Variant Positions
Dilatation
Stenosis
Wall Thickening
Intraluminal Mass
Perivascular Mass
Vena Cava, Veins
Situs Inversus
Aplasia, Hypoplasia, and Duplication of the Aorta
Coarctation of the Aorta
Oblique/Transverse Course of the Aorta
Kinking of the Aorta
Variant and Duplicated Arteries
Situs Inversus 




















































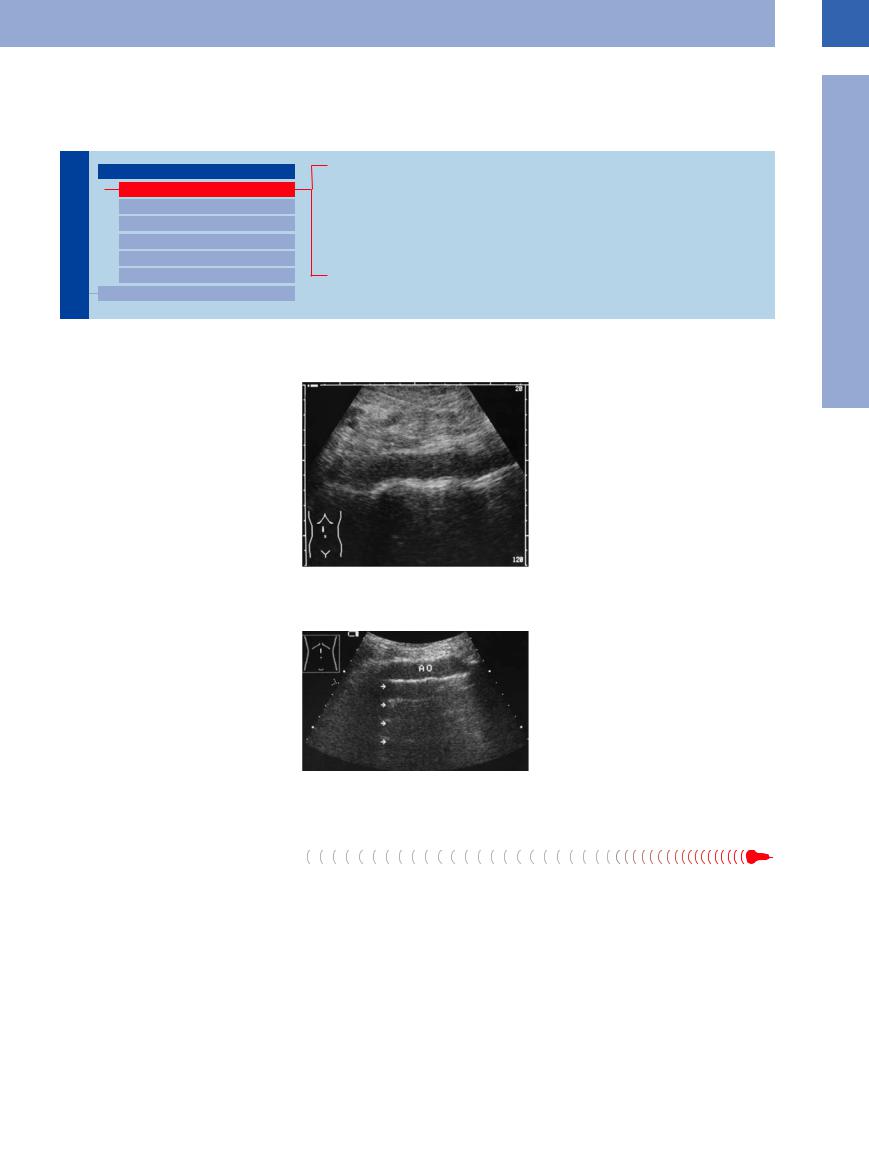
Anomalies of the aorta are quite rare; a situs |
Fig. 1.6 Situs inversus: the aorta is on the right and the |
|
inversus is the most likely reason for surprise, |
vena cava (not depicted) on the left; in most cases the |
|
but in most cases it is already known to the |
diagnosis is clear-cut if both subcostal views demonstrate |
|
malrotation of the liver, i. e., it is located on the left. |
||
patient (Fig.1.6). |
||
|
1
Aorta, Arteries
Aplasia, Hypoplasia, and Duplication of the
and Duplication of the Aorta
Aorta 
































Aplasia, hypoplasia, and duplication of the aorta are extremely rare and in most cases they will have been diagnosed during the perinatal period. Usually an aplastic aorta results in a fatal defect, and only two cases of aortic duplication have been described in the literature. Artifacts mimicking duplication may arise in the transverse epigastric plane from the prismlike effects of the fatty tissue between the rectus abdominis muscles, resulting in re-
fraction artifacts.4 There is also the possibility of a misdiagnosis due to physical ultrasound if echo reverberation produces an image of multiple aortas. The preferred location of this mirroring is found at especially hard interfaces, e. g., plaque incrustation of the aortic wall; here, a kind of ping-pong effect at the echointensive reflectors increases the transit time, which in turn generates multiple images of the aorta (Fig.1.7).
Fig. 1.7 Mirror imaging of the aorta (artifact) due to the parallel thin echogenic lines. AO = aorta.
Coarctation of the Aorta
the Aorta 




Congenital circumscribed circular narrowing of the aorta is known as aortic coarctation. It is found at several segments of the aorta: for the
abdomen these are segments IV (from the subphrenium to below the renal arteries) and V (all the way to the aortic bifurcation). Sonograph-
ically these stenoses are best studied by colorflow Doppler imaging and analysis of the flow parameters (see below).
5

1
Vessels
Oblique/Transverse Course  of the Aorta
of the Aorta






































In case of severe scoliosis or kyphosis of the spine, the aorta will follow this course; the usual longitudinal plane may result in a double lumen, while the transverse view would depict the aorta as a short bandlike structure (Fig.1.8).
Fig. 1.8 Oblique course of the aorta due to kinking.
a Longitudinal subxiphoid plane: because of its oblique course the aorta is visualized twice.
b Oblique subxiphoid plane: the aorta (AO) has been insonated in an almost exactly longitudinal direction. Calipers: aortic ectasia bordering on an aneurysm.
Kinking of  the
the Aorta
Aorta 

























Hypertension and arteriosclerosis can lead not only to an ectatic but also tortuous aorta. Since the spine does not yield, the aorta becomes displaced in a lateral and/or anterior direction and escapes detection by the standard view in the paramedian plane. If the vessel does not follow a normal straight course, the probe should be angled and shifted laterally in order to demonstrate any kinking (Fig.1.9).
Fig. 1.9 Kinking of the aorta (AO) induced by hypertension and arteriosclerosis (arrows: arteriosclerotic lesions).
Variant and Duplicated  Arteries
Arteries 



















The intraperitoneal and retroperitoneal vessels |
● Duplicated renal arteries (Fig.1.12) |
exhibit numerous variations in their branches |
● Renal artery coursing anterior to the vena |
and may course atypically or even become du- |
cava (Fig.1.13). |
plicated. Variants of clinical significance are |
Figure 1.10 is a schematic summary of the most |
found at the celiac axis and the renal arteries, |
|
as demonstrated by the following examples: |
important variants of the celiac axis. The most |
● Variants of the celiac axis (Fig.1.10, Fig.1.11) |
common variant anatomy is found in only 25% |
Fig. 1.10 Variant anatomy of the celiac axis. |
6 |
of cases: here, the left gastric, common hepatic, and splenic arteries arise from a common trunk. Quite often the left gastric artery branches off first, followed by the other two arteries.
Fig. 1.11 Variant anatomy of the celiac axis. AO = aorta; AL = splenic artery; AH = hepatic artery.
a Separate origins of the hepatic and splenic arteries from the aorta.
b Atypical origin of the superior mesenteric artery (AMS) from the celiac trunk (TR): celiomesenteric trunk. V = superior mesenteric vein.
1
Aorta, Arteries
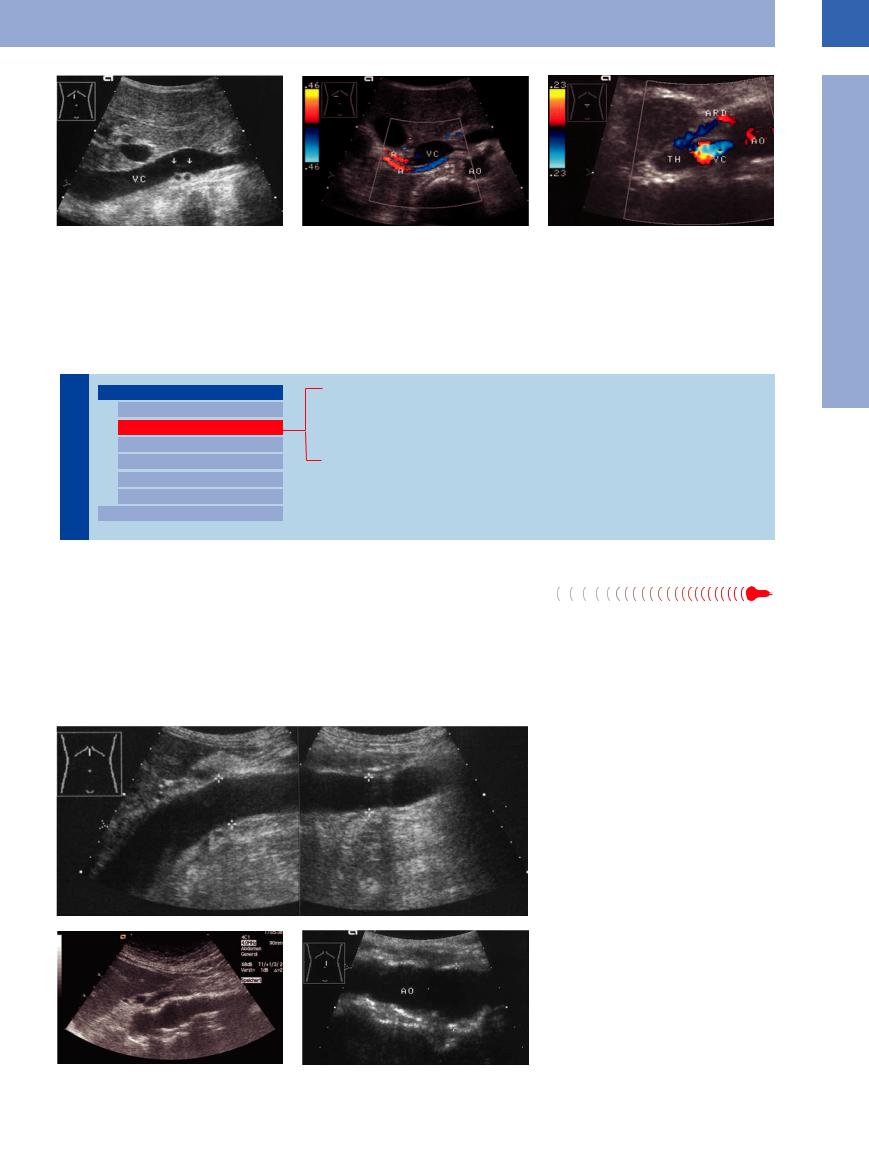
Fig. 1.12 Duplication of the renal artery.
a Longitudinal view: cross-section of two distinct arteries (arrows) posterior to the vena cava (VC).
b Transverse view of the right upper quadrant, color-flow Doppler study. Both renal arteries (A) are depicted; if stenosis of the renal artery is suspected, possible duplication should be ruled out. AO = aorta.
Fig. 1.13 Irregular course of the right renal artery (ARD) anterior to the vena cava (VC). Secondary finding: partial thrombosis (TH) of the vena cava. AO = aorta.
Dilatation
Vessels |
|
|
Aorta, Arteries |
|
|
|
|
||
|
|
|
|
Anomalies and Variant Positions |
|
|
|
|
|
|
|
|
|
Dilatation |
|
|
|
|
|
|
|
|
|
Stenosis |
|
|
|
|
Wall Thickening |
|
|
|
|
Intraluminal Mass |
|
|
|
|
Perivascular Mass |
|
|
|
Vena Cava, Veins |
|
|
|
|||
Aortic Ectasia
True Aneurysms
Dissecting Aneurysms
False Aneurysms
Aortic Ectasia 





























A condition where the lumen diameter of the |
Marfan syndrome is a rare genetic disease |
aorta expands to 25–30 mm is defined as ecta- |
with a high risk of a life-threatening acute |
sia. The primary trigger factors for this degen- |
aortic syndrome resulting from a dissecting |
eration are hypertension and arteriosclerosis. |
aneurysm of the thoracic aorta. The cause is a |
In most cases an ectatic aorta also displays a |
mutation in fibrillin, a connective tissue pro- |
tortuous course. |
tein. Other organs and symptoms are therefore |
also involved, such as joint hypermobility (found in all forms, in addition to the aortic aneurysm), and, in most cases, thoracic deformations, arachnodactylia, and mitral valve prolapse. Early diagnosis allows preventive or timely surgical treatment (Fig.1.14).
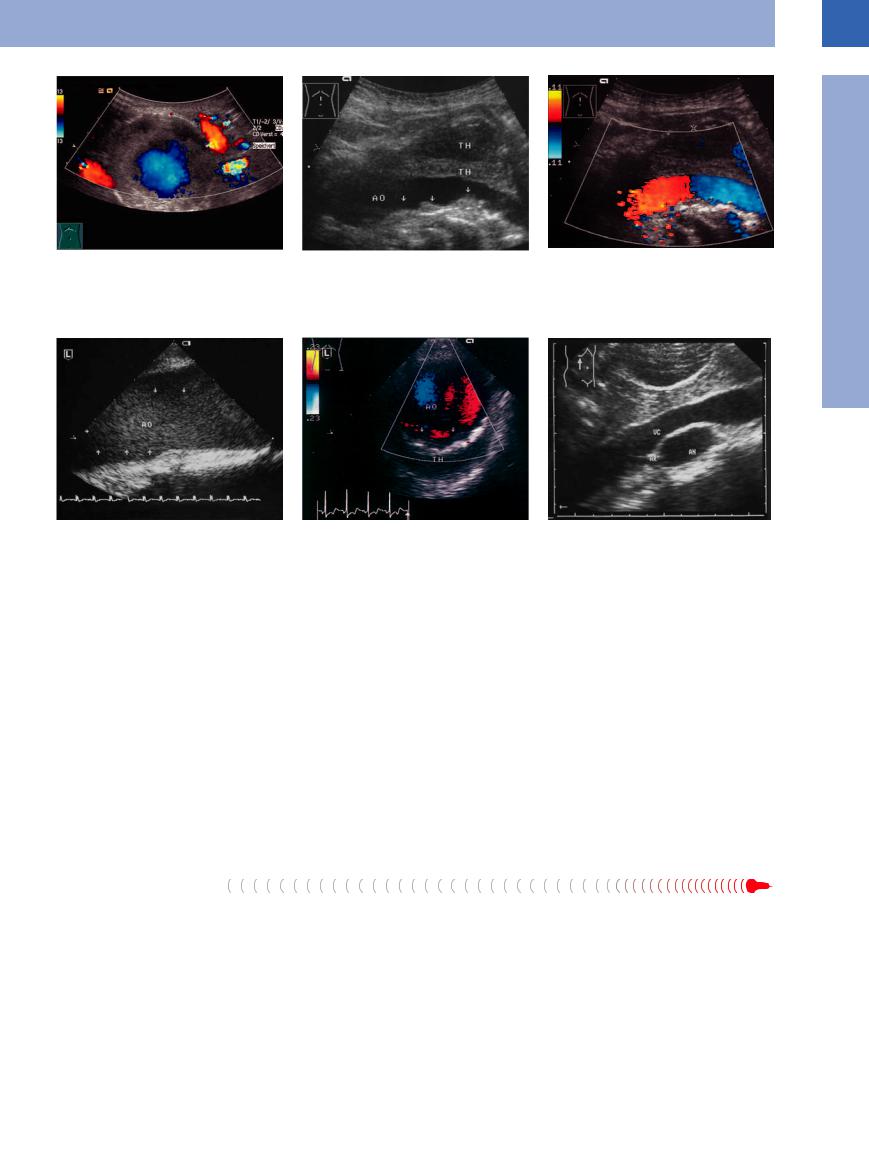
Fig. 1.14
a Marfan syndrome. Ectatic aorta with severe insufficiency of the aortic valve: tortuous ectatic aorta.
b Ectatic aorta. Localized expansion of the ectatic aorta (AO) to 2.7 cm.
c Borderline aortic aneurysm: localized expansion of the ectatic aorta (AO) to 29.5 mm just cephalad of the bifurcation.
7

1
Vessels
Fig. 1.15
a Types of arterial aneurysms. Left: True aneurysms: 1, fusiform; 2, saccular; 3, transition into the iliac arteries. Center: Dissecting aneurysm. Right: False aneurysm.
b Morphometric definition of abdominal aortic aneurysms to the calculated endoluminal aortic grafts (reproduced with permission from Schumacher et al21 © International Society of Endovascular Specialists).
True Aneurysms









An aneurysm is a localized dilatation of the arterial wall. Aneurysms may be classified as follows:
●Etiology
–Congenital
–Atherosclerotic
–Dissecting
–Inflammatory
–Traumatic
●Morphology
–Fusiform
–Pouch-shaped
–Tortuous
–Cylindrical
–Saccular
–Navicular
●Pathogenesis
–True
–False (pseudo-)
–Dissecting
Aortic aneurysms. A practical working definition of an abdominal aortic aneurysm is a transverse lumen diameter of at least 30 mm. Localized atheromatous lesions in the wall facilitate the dilatation of the vessel and impair its contractility, resulting in a weakened aortic wall with aneurysm formation. Small aneurysms do not usually have any thrombotic deposits. Larger aortic aneurysms are characterized by dilatation, arteriosclerosis, and white thrombi, each of these being identifiable by ultrasound. If all three signs can be demonstrated sonographically, the diagnosis of an aneurysm is confirmed (Fig.1.15, Fig.1.16).
Increased size. Aneurysms tend to expand in size over time. The larger the lumen becomes, the less pressure the wall will tolerate before rupturing. According to Laplace’s law the tension in the wall is directly proportional to the radius; thus, small aneurysms with a diameter
of less than 5 cm are not very likely to rupture, while for aneurysms with a diameter of more than 7 cm the probability of rupture within 1 year approaches 50%. The aneurysm expansion rate can be monitored sonographically: aneurysms more than 5 cm in diameter exhibit a mean annual increase in diameter of 0.6 cm, while for smaller aneurysms the corresponding value drops to 0.2 cm. Patients with diagnosed aneurysms should undergo follow-up scans every 3–6 months; if the jet flow direction is toward the wall and the quarterly growth rate is more than 0.3–0.4 cm, an operation should be discussed with the multidisciplinary team. Generally aneurysms greater than 5.5 cm in size (AP depth) should be surgically treated.
Aneurysm morphology. Ultrasound imaging shows the various pathologically familiar shapes possible in abdominal aortic aneurysms. Most often the aciniform, pouchshaped, navicular-type, and tortuous aneur- ysms—and some of the saccular ones as well— are true aneurysms. Other saccular aneurysms and the unilateral navicular type may be clas-
sified as dissecting aneurysms. Cylindrical aneurysms start and end rather abruptly (hence their cylindrical appearance). Asymmetric aneurysms should be surgically treated sooner than fusiform ones. Since the introduction of endovascular prosthethic therapy as an alternative to conventional surgery there is now a simplified classification (aciniform, saccular, asymmetric, extended to the iliac arteries), which has significance for the evaluation of further therapeutic procedures5 (Fig.1.15; see also intraluminal masses, endovascular prothesis, Fig.1.50).
The various shapes of abdominal aortic aneurysms are illustrated in  1.1.
1.1.
Diagnosis in abdominal aortic aneurysms.
Aortic aneurysms attract special interest because of their high propensity to rupture; saccular asymmetric dilatations and marginal
flow indicate |
a higher rate of rupture |
(Fig. 1.16b,c; |
1.1c,g,h). |
In ultrasound studies the rupture appears as an anechoic para-aortic band (“stable” rupture), and is confirmed when the leak with its
Formal and Causative Aneurysm Formation
In true aneurysms there is a localized dilatation of the entire wall, the most frequent cause being arteriosclerosis. Congenital aneurysms arise from defects in or fibromuscular dysplasia of the media. Most mycotic aneurysms are sequelae of systemic mycosis.
False aneurysms result from vascular trauma (nowadays most often because of diagnostic procedures, such as catheterization) leading to a perivascular hematoma that is walled off by the surrounding tissue and quite often ends up becoming thrombosed.
Dissecting aneurysms occur spontaneously in middle-aged and older adults, with hypertension being one of the most common factors. Congenital (Marfan syndrome, idiopathic cystic necrosis of the media) and acquired (syphilis) etiology plays a minor role. The trigger event is a tear in the intima (entry point) with blood now forcing itself into the media. This results in a “pseudolumen” which rejoins the true lumen at a more caudad location through another tear in the intima (re-entry point).
8
1
Aorta, Arteries
Fig. 1.16 Large aortic aneurysm.
a Large aortic aneurysm, centrally perfused, marginal thrombi.
Fig. 1.17
a Thoracic aortic aneurysm (AO). TEE: expansion of the aorta to 40 mm, floating thrombus within the lumen (red thrombus made up of fibrin and red blood cells; arrows). Massive thickening and sclerosis of the wall.
blood jet (“open” rupture) can be demonstrated ( 1.2a,b). In most cases this lifethreatening emergency, requiring emergency repair, does not leave much time for any detailed study.
1.2a,b). In most cases this lifethreatening emergency, requiring emergency repair, does not leave much time for any detailed study.
Other essential parameters to note during ultrasound imaging of abdominal aortic aneurysms are location, length, and the possible involvement of the renal and iliac arteries. With regard to operative possibilities and prognosis, especially when renal arteries are involved, endovascular prosthesis is not possible (
1.2c–i).
Thoracic aortic aneurysm. A true thoracic aneurysm is most often detected by chest ra-
Dissecting Aneurysms
In ultrasonography, dissecting aneurysms are characterized by an echogenic intima floating in synchronous motion with the pulse, or this echogenic intima may be thickened and rigid as a result of atherosclerosis. The vessel itself may be dilated and display all other signs of an aneurysm; however, quite often the artery appears otherwise normal, and the diagnosis is confirmed only by demonstrating the shearedoff intima within the lumen generating the color-flow Doppler image of two distinct lumina.
b All criteria of a true aneurysm are fulfilled in this B- mode image: widening of the vessel, sclerosis of the aorta (arrows), white thrombi (TH). AO = aorta.
b Dissecting aortic aneurysm, Stanford type A. TEE demonstrates the intraluminal hyperechoic intima (arrows) and the thrombosed, nonperfused lumen of the dissected area. (Patient declined surgery, died suddenly 2 weeks later.) AO = aorta.
diograph; it will be confirmed by transesophageal echocardiography (TEE) or CT. Investigation of acute thoracic pathology (often an acute coronary syndrome) should also involve a diagnosis or exclusion of aortic root dissection; sometimes an extension into the abdominal aorta may also be detected here (Fig.1.17).
Nonaortic aneurysms. Aneurysms are not limited to the aorta but may also be found in other intraand retroperitoneal or peripheral (subclavian artery) vessels. However, isolated iliac artery aneurysms without an associated abdominal aortic aneurysm are rare ( 1.1 d,g,h).
1.1 d,g,h).
In nonaortic aneurysms the actual diameters separating ectatic and aneurysmic arteries
In more than 90% of patients, dissection with its massive, sharp chest pain will commence as thoracic aneurysm and propagate to the abdominal aorta (Stanford classification type B: begins at the ascending aorta or the aortic arch with possible extension to the abdominal aorta). Thoracic aneurysms immediately distal to the aortic valve (Stanford classification type A: involving only the aortic arch and ascending aorta) are diagnosed sonographically by echocardiography or TEE (Fig.1.17b, Fig.1.19).
c Color-flow Doppler: the remaining perfused lumen is defined by the color-coded signal; the color change (from red through black to blue) signifies the change in the flow direction—the flow toward the probe is red, and away from the probe it is blue.
Fig. 1.18 Right subxiphoid view in the longitudinal plane: the vena cava (VC) is insonated through the oblique section of the aneurysm (AN). AR = renal artery.
from normal vessels have not been defined. Diffuse dilatation of the vessel is classified as ectatic, but aneurysms always consist of a localized dilatation of the artery with thinning and structural weakening of the wall. The most common factor in the pathogenesis is atherosclerosis (60%), congenital defects in the wall playing a minor (20%) but still more frequent role than in the aorta where atherosclerosis accounts for the overwhelming majority (95%) of cases. The most frequent location of visceral artery aneurysms is the splenic artery, followed by the hepatic and renal arteries (Fig. 1.18).
The causes of aortic dissection can include damage to the collagen elastic fibrous tissue of the media due to ischemia (arteriosclerosis), inflammation (syphilis), or genetics (Marfan syndrome) (Fig.1.14).
Marfan syndrome and similar diseases are very rare causes of an aortic disorder—ectasia or aneurysm, most often with a dissection. But because there is a possibility of acute dissection, long-term and rigorous follow-up is necessary for these patients.
9
1
Vessels
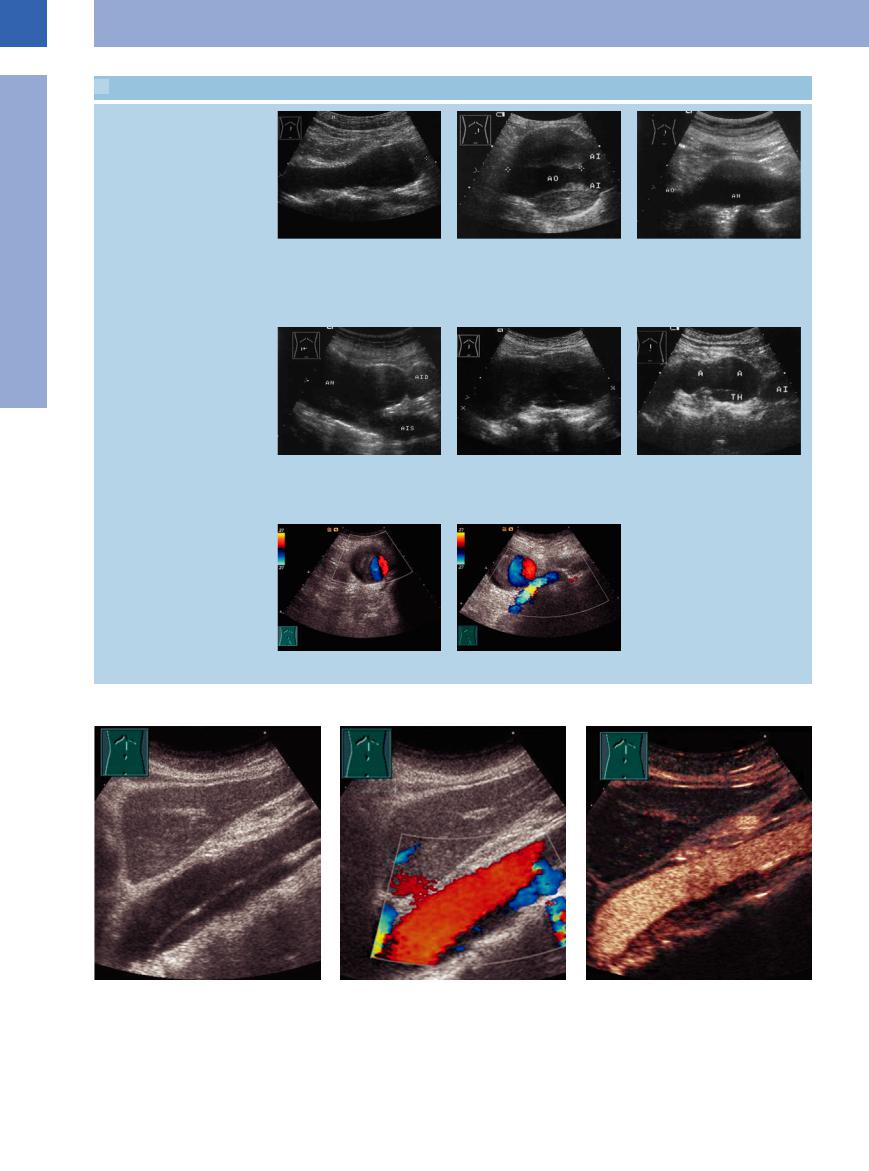
 1.1 The Shape of Aortic Aneurysms
1.1 The Shape of Aortic Aneurysms
Fusiform, saccular aneurysm
a Fusiform aneurysm (cursors). Symmetrical ovally dilated aorta. Longitudinal dimension 5.8 cm, depth 3.7 cm.22
Involvement of the iliac arteries, cylindrical aneurysm
d Aortic aneurysm (AN), junction with the aneurysmal right and left iliac arteries (AID, AIS). Longitudinal scan plane from right to left (to the origin of the inferior mesenteric artery). Type IIB.
Iliac aneurysm
g Iliac aneurysm, eccentric lumen.
b Saccular (old classification; from the operative point of view, fusiform) aneurysm > 10 cm: remaining lumen (AO) in the center surrounded by a concentric thrombosis; the aneurysm ends where the aorta bifurcates into the iliac artery (AI). Type IIB.
e Large cylindrical aortic aneurysm extending over 13.2 cm, i. e., including the renal arteries (distance from aortic bifurcation is > 9.5 cm). Type IIA.
h Additionally, the external iliac vein is seen.
c Boat-shaped (old classification; from the operative point of view, saccular) aneurysm (AN; cursors). Type I. The aneurysm projects anteriorly on one side.
f Berry-shaped double aneurysm (A), diameter > 30 cm including the marginal thrombosis (TH). AI = ectatic iliac artery.22
Fig. 1.19 Dissecting thoracic aneurysm. Dissection detect- |
b CDS. |
c Contrast-enhanced sonography (CEUS). |
able as far as the level of the renal arteries. Perfusion is |
|
|
reduced in the false lumen (Stanford type B). |
|
|
a B-mode. |
|
|
10
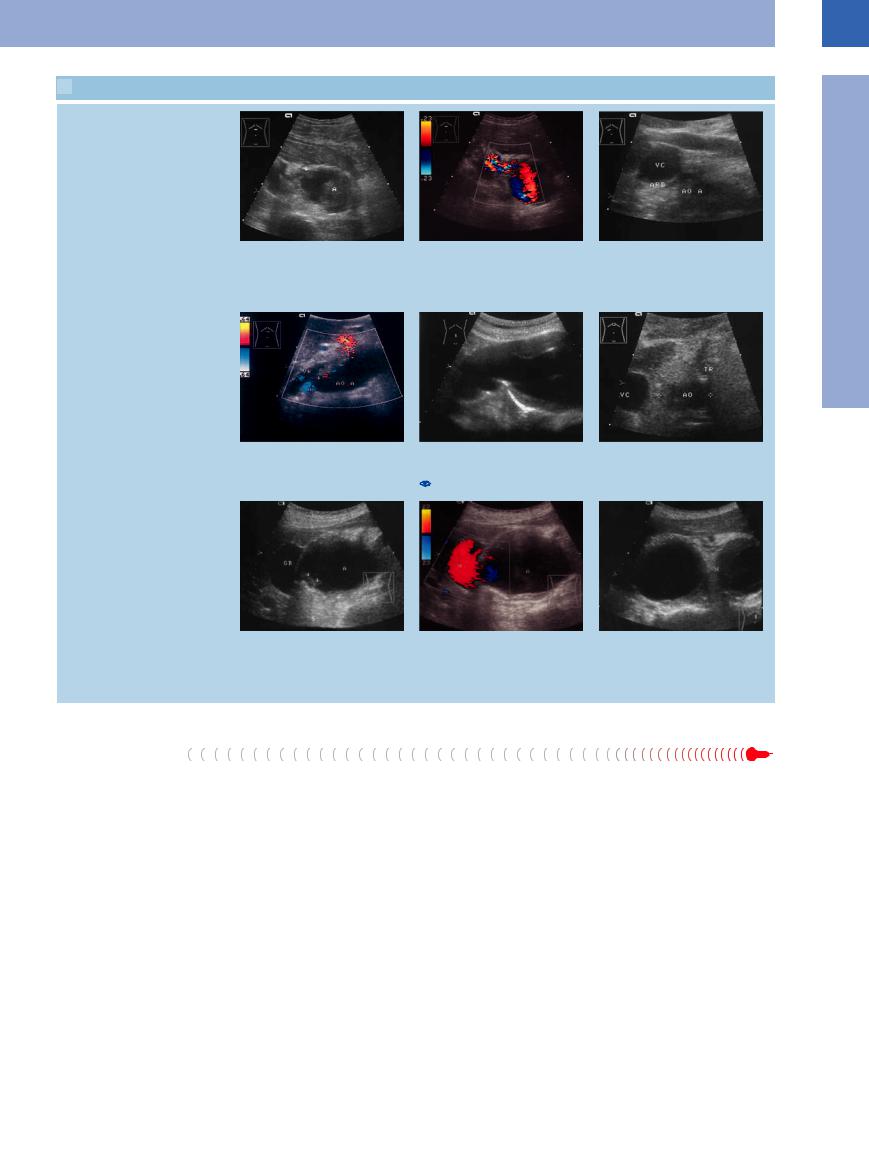
 1.2 Diagnosis in Aortic Aneurysm
1.2 Diagnosis in Aortic Aneurysm
Aneurysm extent, aneurysm complications
a and b Aortic aneurysm with open rupture, transverse subxiphoid scans.
a B-mode image: suprarenal aortic aneur- |
b Color-flow Doppler image: rupture leak- |
ysm (A) with partial thrombosis and an- |
ing blood; the turbulence produces this |
echoic band (arrow) extending to a |
“confetti-like” phenomenon. |
subhepatic fluid pool. |
|
d Tortuous aortic aneurysm (AO A). Origin |
e Tortuous aortic aneurysm. These forms |
of the renal artery (A, arrow) and celiac |
are di cult to define with ultrasound be- |
trunk (TR, cut by the scan) from the |
cause the loops usually run sideways (see |
aneurysm. VR = renal vein. |
1.2c). |
g Aneurysmic aorta (A): the entire vessel |
h Color-flow Doppler: the anechoic masses |
has become a succession of aneurysms. |
in d are part of the aortic aneurysmosis |
Right paramedian transverse subxiphoid |
(A); without the color-flow Doppler the |
view: crescent-shaped mass in the porta |
anechoic crescent would be misdiagnosed |
hepatis, initially misdiagnosed as gallblad- |
as gallbladder. |
der (GB). |
|
c Origin of the right renal artery (ARD) from the aortic aneurysm (AOA). VC = vena cava.
f Origin of the celiac trunk (TR) from the aneurysm (AO).
i Longitudinal subxiphoid view in the median plane: adjacent saccular aneurysms.
False Aneurysms
Aorta. Compared with the false aneurysms (pseudoaneurysms) most common today—i. e., after puncture of the femoral artery—traumatic aneurysms of the aorta are quite rare. Sometimes false aortic aneurysms are seen as a result of suture-line leakage, but these anastomotic pseudoaneurysms of the aorta are also rather infrequent. In ultrasound studies they
appear as an anechoic periaortic mass, usually situated at the anastomosis and coursing with the aorta.
Arteries. Femoral artery. Catheterization of the femoral artery is complicated by a fairly significant rate of pseudoaneurysm formation. The diagnosis is confirmed by color-flow Dop-
pler, which shows blood spurting into the cavity of the aneurysm. This differentiates the diagnosis from other perivascular masses (see below). Quite often the leak is pinpointed precisely by the blood jet and may then be treated by compression of this area with the probe, resulting in thrombosis of the leak and thus closure (Fig.1.20).
1
Aorta, Arteries
11
