
- •COMPUTATIONAL CHEMISTRY
- •CONTENTS
- •PREFACE
- •1.1 WHAT YOU CAN DO WITH COMPUTATIONAL CHEMISTRY
- •1.2 THE TOOLS OF COMPUTATIONAL CHEMISTRY
- •1.3 PUTTING IT ALL TOGETHER
- •1.4 THE PHILOSOPHY OF COMPUTATIONAL CHEMISTRY
- •1.5 SUMMARY OF CHAPTER 1
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •2.1 PERSPECTIVE
- •2.2 STATIONARY POINTS
- •2.3 THE BORN–OPPENHEIMER APPROXIMATION
- •2.4 GEOMETRY OPTIMIZATION
- •2.6 SYMMETRY
- •2.7 SUMMARY OF CHAPTER 2
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •3.1 PERSPECTIVE
- •3.2 THE BASIC PRINCIPLES OF MM
- •3.2.1 Developing a forcefield
- •3.2.2 Parameterizing a forcefield
- •3.2.3 A calculation using our forcefield
- •3.3 EXAMPLES OF THE USE OF MM
- •3.3.2 Geometries and energies of polymers
- •3.3.3 Geometries and energies of transition states
- •3.3.4 MM in organic synthesis
- •3.3.5 Molecular dynamics and Monte Carlo simulations
- •3.4 GEOMETRIES CALCULATED BY MM
- •3.5 FREQUENCIES CALCULATED BY MM
- •3.6 STRENGTHS AND WEAKNESSES OF MM
- •3.6.1 Strengths
- •3.6.2 Weaknesses
- •3.7 SUMMARY OF CHAPTER 3
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •4.1 PERSPECTIVE
- •4.2.1 The origins of quantum theory: blackbody radiation and the photoelectric effect
- •4.2.2 Radioactivity
- •4.2.3 Relativity
- •4.2.4 The nuclear atom
- •4.2.5 The Bohr atom
- •4.2.6 The wave mechanical atom and the Schrödinger equation
- •4.3.1 Introduction
- •4.3.2 Hybridization
- •4.3.3 Matrices and determinants
- •4.3.4 The simple Hückel method – theory
- •4.3.5 The simple Hückel method – applications
- •4.3.6 Strengths and weaknesses of the SHM
- •4.4.1 Theory
- •4.4.2 An illustration of the EHM: the protonated helium molecule
- •4.4.3 The extended Hückel method – applications
- •4.4.4 Strengths and weaknesses of the EHM
- •4.5 SUMMARY OF CHAPTER 4
- •REFERENCES
- •EASIER QUESTIONS
- •5.1 PERSPECTIVE
- •5.2.1 Preliminaries
- •5.2.2 The Hartree SCF method
- •5.2.3 The HF equations
- •5.2.3.1 Slater determinants
- •5.2.3.2 Calculating the atomic or molecular energy
- •5.2.3.3 The variation theorem (variation principle)
- •5.2.3.4 Minimizing the energy; the HF equations
- •5.2.3.5 The meaning of the HF equations
- •5.2.3.6a Deriving the Roothaan–Hall equations
- •5.3 BASIS SETS
- •5.3.1 Introduction
- •5.3.2 Gaussian functions; basis set preliminaries; direct SCF
- •5.3.3 Types of basis sets and their uses
- •5.4 POST-HF CALCULATIONS: ELECTRON CORRELATION
- •5.4.1 Electron correlation
- •5.4.3 The configuration interaction approach to electron correlation
- •5.5.1 Geometries
- •5.5.2 Energies
- •5.5.2.1 Energies: Preliminaries
- •5.5.2.2 Energies: calculating quantities relevant to thermodynamics and to kinetics
- •5.5.2.2a Thermodynamics; “direct” methods, isodesmic reactions
- •5.5.2.2b Thermodynamics; high-accuracy calculations
- •5.5.2.3 Thermodynamics; calculating heats of formation
- •5.5.2.3a Kinetics; calculating reaction rates
- •5.5.2.3b Energies: concluding remarks
- •5.5.3 Frequencies
- •Dipole moments
- •Charges and bond orders
- •Electrostatic potential
- •Atoms-in-molecules
- •5.5.5 Miscellaneous properties – UV and NMR spectra, ionization energies, and electron affinities
- •5.5.6 Visualization
- •5.6 STRENGTHS AND WEAKNESSES OF AB INITIO CALCULATIONS
- •5.7 SUMMARY OF CHAPTER 5
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •6.1 PERSPECTIVE
- •6.2 THE BASIC PRINCIPLES OF SCF SE METHODS
- •6.2.1 Preliminaries
- •6.2.2 The Pariser-Parr-Pople (PPP) method
- •6.2.3 The complete neglect of differential overlap (CNDO) method
- •6.2.4 The intermediate neglect of differential overlap (INDO) method
- •6.2.5 The neglect of diatomic differential overlap (NDDO) method
- •6.2.5.2 Heats of formation from SE electronic energies
- •6.2.5.3 MNDO
- •6.2.5.7 Inclusion of d orbitals: MNDO/d and PM3t; explicit electron correlation: MNDOC
- •6.3 APPLICATIONS OF SE METHODS
- •6.3.1 Geometries
- •6.3.2 Energies
- •6.3.2.1 Energies: preliminaries
- •6.3.2.2 Energies: calculating quantities relevant to thermodynamics and kinetics
- •6.3.3 Frequencies
- •6.3.4 Properties arising from electron distribution: dipole moments, charges, bond orders
- •6.3.5 Miscellaneous properties – UV spectra, ionization energies, and electron affinities
- •6.3.6 Visualization
- •6.3.7 Some general remarks
- •6.4 STRENGTHS AND WEAKNESSES OF SE METHODS
- •6.5 SUMMARY OF CHAPTER 6
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •7.1 PERSPECTIVE
- •7.2 THE BASIC PRINCIPLES OF DENSITY FUNCTIONAL THEORY
- •7.2.1 Preliminaries
- •7.2.2 Forerunners to current DFT methods
- •7.2.3.1 Functionals: The Hohenberg–Kohn theorems
- •7.2.3.2 The Kohn–Sham energy and the KS equations
- •7.2.3.3 Solving the KS equations
- •7.2.3.4a The local density approximation (LDA)
- •7.2.3.4b The local spin density approximation (LSDA)
- •7.2.3.4c Gradient-corrected functionals and hybrid functionals
- •7.3 APPLICATIONS OF DENSITY FUNCTIONAL THEORY
- •7.3.1 Geometries
- •7.3.2 Energies
- •7.3.2.1 Energies: preliminaries
- •7.3.2.2 Energies: calculating quantities relevant to thermodynamics and kinetics
- •7.3.2.2a Thermodynamics
- •7.3.2.2b Kinetics
- •7.3.3 Frequencies
- •7.3.6 Visualization
- •7.4 STRENGTHS AND WEAKNESSES OF DFT
- •7.5 SUMMARY OF CHAPTER 7
- •REFERENCES
- •EASIER QUESTIONS
- •HARDER QUESTIONS
- •8.1 FROM THE LITERATURE
- •8.1.1.1 Oxirene
- •8.1.1.2 Nitrogen pentafluoride
- •8.1.1.3 Pyramidane
- •8.1.1.4 Beyond dinitrogen
- •8.1.2 Mechanisms
- •8.1.2.1 The Diels–Alder reaction
- •8.1.2.2 Abstraction of H from amino acids by the OH radical
- •8.1.3 Concepts
- •8.1.3.1 Resonance vs. inductive effects
- •8.1.3.2 Homoaromaticity
- •8.2 TO THE LITERATURE
- •8.2.1 Books
- •8.2.2 The Worldwide Web
- •8.3 SOFTWARE AND HARDWARE
- •8.3.1 Software
- •8.3.2 Hardware
- •8.3.3 Postscript
- •REFERENCES
- •INDEX

368 Computational Chemistry
bonds, C/H and C/C, both of which are nonpolar. A problem with AM1 and PM3 IR spectra, then, is that a “missing” band may not be particularly weak, and may represent a prominent structural feature, e.g. the O–H stretch of methanol.
The wavenumbers (frequencies) of SE vibrations are more reliable than the intensities. All the normal modes are actually present in the results of an AM1 or PM3 frequency calculation, and animation of these will usually give, approximately, the frequencies of these vibrations. A very extensive compilation of experimental, MNDO and AM1 frequencies has been given by Healy and Holder, who conclude that the AM1 error of 10% can be reduced to 6% by an empirical correction, and that entropies and heat capacities are accurately calculated from the frequencies [72]. In this regard, Coolidge et al. conclude – surprisingly, in view of our results for the four molecules in Figs 6.5–6.8 – from a study of 61 molecules that (apart from problems with ringand heavy atom-stretch for AM1 and S–H, P–H and O–H stretch for PM3) “both AM1 and PM3 should provide results that are close to experimental gas phase spectra” [73].
6.3.4Properties arising from electron distribution: dipole moments, charges, bond orders
The discussion in section 5.5.4 on dipole moments, charges and bond orders applies in a general way to the calculation of these quantities by SE methods too. Electrostatic

Semiempirical Calculations 369
potentials, whether visualized as regions of space or mapped onto van der Waals surfaces, are usually qualitatively the same for AM1 and PM3 as for ab initio methods. Atoms-in-molecules calculations are not viable for SE methods, because the core orbitals, lacking in these methods, are important for AIM calculations.
Dipole moments
Hehre’s extensive survey of practical computational methods reports the results of ab initio and DFT single point dipole moment  calculations on AM1 geometries [74]. There does not appear to be much advantage to calculating HF/6-31G* dipole moments on HF/6-31G* geometries (HF/6-31G*//HF/6-31G* calculations) rather than on the much more quicklyobtained AM1 geometries (HF/6-31G*//AM1 calculations). Indeed, even the relatively time-consuming MP2/6-31G*//MP2/6-31G* calculations seem to offer little advantage over fast HF/6-31G*//AM1 calculations as far as dipole moments are concerned (Tables 2.19 and 2.21 in Ref. [74]). This is consistent with our finding that AM1 geometries are quite good (section 6.3.1). Table 6.5 compares calculated and experimental dipole moments for 10 molecules,
calculations on AM1 geometries [74]. There does not appear to be much advantage to calculating HF/6-31G* dipole moments on HF/6-31G* geometries (HF/6-31G*//HF/6-31G* calculations) rather than on the much more quicklyobtained AM1 geometries (HF/6-31G*//AM1 calculations). Indeed, even the relatively time-consuming MP2/6-31G*//MP2/6-31G* calculations seem to offer little advantage over fast HF/6-31G*//AM1 calculations as far as dipole moments are concerned (Tables 2.19 and 2.21 in Ref. [74]). This is consistent with our finding that AM1 geometries are quite good (section 6.3.1). Table 6.5 compares calculated and experimental dipole moments for 10 molecules,

370 Computational Chemistry
using these methods: AM1 (using the AM1 method to calculate 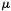 for the AM1 geometry, AM1//AM1), HF/6-31*//AM1, PM3 (PM3//PM3), HF/6-31G*//PM3, and MP2/6-31G* (MP2/6-31G*//MP2/6-31G*). For this set of molecules, the smallest deviation from experiment, as judged by the arithmetic mean of the absolute deviations from the experimental values, is shown by the AM1 calculation (0.21 Debyes), and the largest deviation is shown by the “highest” method, MP2/6-31G* (0.34 D). The other three methods give essentially the same errors (0.27–0.29 D). It is of course possible that AM1 gives the best results (for this set on molecules, at least) because errors in geometry and errors in the calculation of the electron distribution cancel. A study of 196 C, H, N, O, F, Cl, Br, I molecules gave these mean absolute errors: AM1, 0.35 D; PM3, 0.40D; SAM1, 0.32 D [50]. Another study with 125 H, C, N, O, F, A1, Si, P, S, Cl, Br, I molecules gave mean absolute errors of: AM1, 0.35 D and PM3, 0.38D [44]. So with these larger samples the AM1 errors were somewhat bigger. Nevertheless, all these results taken together do indicate that unless one is prepared to use the slower approach of large basis sets with density functional (chapter 7) methods (errors of ca. 0.1 D [75]; this paper also gives some results for ab initio calculations), AM1 dipole moments using AM1 geometries may be as good a way as any to calculate this quantity.
for the AM1 geometry, AM1//AM1), HF/6-31*//AM1, PM3 (PM3//PM3), HF/6-31G*//PM3, and MP2/6-31G* (MP2/6-31G*//MP2/6-31G*). For this set of molecules, the smallest deviation from experiment, as judged by the arithmetic mean of the absolute deviations from the experimental values, is shown by the AM1 calculation (0.21 Debyes), and the largest deviation is shown by the “highest” method, MP2/6-31G* (0.34 D). The other three methods give essentially the same errors (0.27–0.29 D). It is of course possible that AM1 gives the best results (for this set on molecules, at least) because errors in geometry and errors in the calculation of the electron distribution cancel. A study of 196 C, H, N, O, F, Cl, Br, I molecules gave these mean absolute errors: AM1, 0.35 D; PM3, 0.40D; SAM1, 0.32 D [50]. Another study with 125 H, C, N, O, F, A1, Si, P, S, Cl, Br, I molecules gave mean absolute errors of: AM1, 0.35 D and PM3, 0.38D [44]. So with these larger samples the AM1 errors were somewhat bigger. Nevertheless, all these results taken together do indicate that unless one is prepared to use the slower approach of large basis sets with density functional (chapter 7) methods (errors of ca. 0.1 D [75]; this paper also gives some results for ab initio calculations), AM1 dipole moments using AM1 geometries may be as good a way as any to calculate this quantity.

Semiempirical Calculations 371
This applies, of course, only to conventional molecules; molecules of exotic structure (note the remarks for the geometries of hypervalent molecules and molecules ofunusual structure in section 6.3.1) may defy accurate SE predictions.
Charges and bond orders
The conceptual and mathematical bases of these concepts were outlined in chapter 5 (section 5.5 4). We saw that unlike, say, frequencies and dipole moments, charges and bond orders cannot even in principle be measured experimentally; as physicists say, they are not observables. Thus there are no “right” values to calculate, and in fact no single, correct, definitions of these terms, since as with ab initio calculations, SE charges and bond orders can be defined in various ways. The concepts are nevertheless useful, and electrostatic potential charges and Löwdin bond orders are preferred nowadays to the Mulliken parameters.
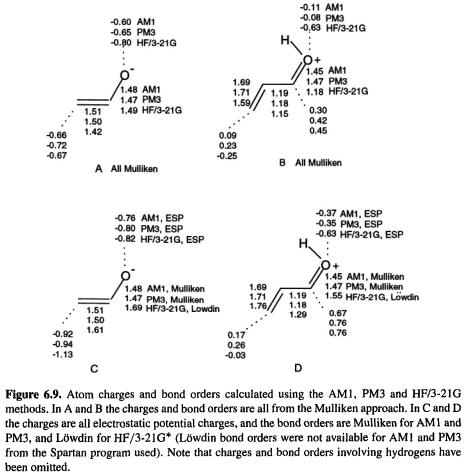
Figure 6.9 shows charges and bond orders calculated for an enolate (the conjugate base of ethenol or vinyl alcohol) and for a protonated enone system (protonated propenal). Consider first Mulliken charges and bond orders of the enolate (Fig. 6.9A). The AM1 and PM3 charges, which are essentially the same, are a bit surprising in that the carbon which shares charge with the oxygen in the alternative resonance structure is given a bigger charge than the oxygen; intuitively, one expects most of the negative charge to be on the more electronegative atom, oxygen (this “defect” of AM1 and PM3 has been noted by Anh et al. [76]). The HF/3-21G method gives the oxygen the bigger charge (–0.80 vs. –0.67). The two SE and the HF methods all give C/C and C/O bond orders of about 1.5; this, and the rough equality of O and C charges, suggests approximately equal contributions from the O-anion and C-anion resonance structures.
372 Computational Chemistry
The Mulliken charges of the protonated enone system (Fig. 6.9B) make the oxygen negative, which may seem surprising. However, this is normal for protonated oxygen and nitrogen (though not protonated sulfur and phosphorus): the hetero atom in 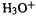 and in
and in  is calculated to be negative (i.e. the positive charge is on the hydrogens) and the hetero atom is also negative in
is calculated to be negative (i.e. the positive charge is on the hydrogens) and the hetero atom is also negative in 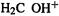 and
and  On the oxygen and the carbon furthest from the oxygen
On the oxygen and the carbon furthest from the oxygen  the HF/3-21G charges differ considerably from the SE ones: the HF calculations make the O much more negative, and make
the HF/3-21G charges differ considerably from the SE ones: the HF calculations make the O much more negative, and make 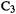 negative, suggesting that they place more positive charge on the hydrogens than do the semiempirical calculations (in all cases the charge on
negative, suggesting that they place more positive charge on the hydrogens than do the semiempirical calculations (in all cases the charge on  is 0.3–0.5). The three methods do not differ as greatly in their bond order results, although HF method makes the formal C/O double bond essentially a single bond (bond order 1.18).
is 0.3–0.5). The three methods do not differ as greatly in their bond order results, although HF method makes the formal C/O double bond essentially a single bond (bond order 1.18).
Finally, electrostatic potential (ESP) charges and, for the HF/3-21G calculations, Löwdin bond orders, are shown (Figs 6.9C and D). For the enolate, all three methods make the ESP charge on carbon more negative than that on oxygen, but the bond orders
