
- •Preface to the 3rd edition
- •General Pharmacology
- •Systems Pharmacology
- •Therapy of Selected Diseases
- •Subject Index
- •Abbreviations
- •General Pharmacology
- •History of Pharmacology
- •Drug and Active Principle
- •The Aims of Isolating Active Principles
- •European Plants as Sources of Effective Medicines
- •Drug Development
- •Congeneric Drugs and Name Diversity
- •Oral Dosage Forms
- •Drug Administration by Inhalation
- •Dermatological Agents
- •From Application to Distribution in the Body
- •Potential Targets of Drug Action
- •External Barriers of the Body
- •Blood–Tissue Barriers
- •Membrane Permeation
- •Binding to Plasma Proteins
- •The Liver as an Excretory Organ
- •Biotransformation of Drugs
- •Drug Metabolism by Cytochrome P450
- •The Kidney as an Excretory Organ
- •Presystemic Elimination
- •Drug Concentration in the Body as a Function of Time—First Order (Exponential) Rate Processes
- •Time Course of Drug Concentration in Plasma
- •Time Course of Drug Plasma Levels during Repeated Dosing (A)
- •Time Course of Drug Plasma Levels during Irregular Intake (B)
- •Accumulation: Dose, Dose Interval, and Plasma Level Fluctuation (A)
- •Dose–Response Relationship
- •Concentration–Effect Curves (B)
- •Concentration–Binding Curves
- •Types of Binding Forces
- •Agonists—Antagonists
- •Other Forms of Antagonism
- •Enantioselectivity of Drug Action
- •Receptor Types
- •Undesirable Drug Effects, Side Effects
- •Drug Allergy
- •Cutaneous Reactions
- •Drug Toxicity in Pregnancy and Lactation
- •Pharmacogenetics
- •Placebo (A)
- •Systems Pharmacology
- •Sympathetic Nervous System
- •Structure of the Sympathetic Nervous System
- •Adrenergic Synapse
- •Adrenoceptor Subtypes and Catecholamine Actions
- •Smooth Muscle Effects
- •Cardiostimulation
- •Metabolic Effects
- •Structure–Activity Relationships of Sympathomimetics
- •Indirect Sympathomimetics
- •Types of
- •Antiadrenergics
- •Parasympathetic Nervous System
- •Cholinergic Synapse
- •Parasympathomimetics
- •Parasympatholytics
- •Actions of Nicotine
- •Localization of Nicotinic ACh Receptors
- •Effects of Nicotine on Body Function
- •Aids for Smoking Cessation
- •Consequences of Tobacco Smoking
- •Dopamine
- •Histamine Effects and Their Pharmacological Properties
- •Serotonin
- •Vasodilators—Overview
- •Organic Nitrates
- •Calcium Antagonists
- •ACE Inhibitors
- •Drugs Used to Influence Smooth Muscle Organs
- •Cardiac Drugs
- •Cardiac Glycosides
- •Antiarrhythmic Drugs
- •Iron Compounds
- •Prophylaxis and Therapy of Thromboses
- •Possibilities for Interference (B)
- •Heparin (A)
- •Hirudin and Derivatives (B)
- •Fibrinolytics
- •Intra-arterial Thrombus Formation (A)
- •Formation, Activation, and Aggregation of Platelets (B)
- •Inhibitors of Platelet Aggregation (A)
- •Presystemic Effect of ASA
- •Plasma Volume Expanders
- •Lipid-lowering Agents
- •Diuretics—An Overview
- •NaCl Reabsorption in the Kidney (A)
- •Aquaporins (AQP)
- •Osmotic Diuretics (B)
- •Diuretics of the Sulfonamide Type
- •Potassium-sparing Diuretics (A)
- •Vasopressin and Derivatives (B)
- •Drugs for Gastric and Duodenal Ulcers
- •Laxatives
- •Antidiarrheal Agents
- •Drugs Affecting Motor Function
- •Muscle Relaxants
- •Nondepolarizing Muscle Relaxants
- •Depolarizing Muscle Relaxants
- •Antiparkinsonian Drugs
- •Antiepileptics
- •Pain Mechanisms and Pathways
- •Eicosanoids
- •Antipyretic Analgesics
- •Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
- •Cyclooxygenase (COX) Inhibitors
- •Local Anesthetics
- •Opioid Analgesics—Morphine Type
- •General Anesthesia and General Anesthetic Drugs
- •Inhalational Anesthetics
- •Injectable Anesthetics
- •Sedatives, Hypnotics
- •Benzodiazepines
- •Pharmacokinetics of Benzodiazepines
- •Therapy of Depressive Illness
- •Mania
- •Therapy of Schizophrenia
- •Psychotomimetics (Psychedelics, Hallucinogens)
- •Hypothalamic and Hypophyseal Hormones
- •Thyroid Hormone Therapy
- •Glucocorticoid Therapy
- •Follicular Growth and Ovulation, Estrogen and Progestin Production
- •Oral Contraceptives
- •Antiestrogen and Antiprogestin Active Principles
- •Aromatase Inhibitors
- •Insulin Formulations
- •Treatment of Insulin-dependent Diabetes Mellitus
- •Treatment of Maturity-Onset (Type II) Diabetes Mellitus
- •Oral Antidiabetics
- •Drugs for Maintaining Calcium Homeostasis
- •Drugs for Treating Bacterial Infections
- •Inhibitors of Cell Wall Synthesis
- •Inhibitors of Tetrahydrofolate Synthesis
- •Inhibitors of DNA Function
- •Inhibitors of Protein Synthesis
- •Drugs for Treating Mycobacterial Infections
- •Drugs Used in the Treatment of Fungal Infections
- •Chemotherapy of Viral Infections
- •Drugs for the Treatment of AIDS
- •Drugs for Treating Endoparasitic and Ectoparasitic Infestations
- •Antimalarials
- •Other Tropical Diseases
- •Chemotherapy of Malignant Tumors
- •Targeting of Antineoplastic Drug Action (A)
- •Mechanisms of Resistance to Cytostatics (B)
- •Inhibition of Immune Responses
- •Antidotes and Treatment of Poisonings
- •Therapy of Selected Diseases
- •Hypertension
- •Angina Pectoris
- •Antianginal Drugs
- •Acute Coronary Syndrome— Myocardial Infarction
- •Congestive Heart Failure
- •Hypotension
- •Gout
- •Obesity—Sequelae and Therapeutic Approaches
- •Osteoporosis
- •Rheumatoid Arthritis
- •Migraine
- •Common Cold
- •Bronchial Asthma
- •Emesis
- •Alcohol Abuse
- •Local Treatment of Glaucoma
- •Further Reading
- •Further Reading
- •Picture Credits
- •Drug Indexes

106 Drugs Acting on the Parasympathetic Nervous System
Parasympathomimetics
Acetylcholine (ACh) is too rapidly hydrolyzed and inactivated by acetylcholinesterase (AChE) to be of any therapeutic use; however, its action can be replicated by other substances, namely, direct or indirect parasympathomimetics.
Direct parasympathomimetics. The choline ester of carbamic acid, carbachol, activates M-cholinoceptors, but is not hydrolyzed by AChE. Carbachol can thus be effectively employed for local application to the eye (glaucoma) and systemic administration (bowel atonia, bladder atonia). The alkaloids pilocarpine (from Pilocarpus jaborandi) and arecoline (from Areca catechu; betel nut) also act as direct parasympathomimetics. As tertiary amines, they moreover exert central effects. The central effect of muscarine-like substances consists in an enlivening, mild stimulation that is probably the effect desired in betel chewing, a widespread habit in South Asia. Of this group, only pilocarpine enjoys therapeutic use, which is almost exclusively by local application to the eye in glaucoma (p.346).
Indirect parasympathomimetics inhibit local AChE and raise the concentration of ACh at receptors of cholinergic synapses. This action is evident at all synapses where ACh is the mediator. Chemically, these agents include esters of carbamic acid (carbamates such as physostigmine, neostigmine) and of phosphoric acid (organophosphates such as paraoxon = E600, and nitrostigmine = parathion = E605, its prodrug).
Members of both groups react like ACh with AChE. The esters are hydrolyzed upon formation of a complex with the enzyme. The rate-limiting step in ACh hydrolysis is deacetylation of the enzyme, which takes only milliseconds, thus permitting a high turnover rate and activity of AChE. Decarbaminoylation following hydrolysis of a carbamate takes hours to days, the enzyme re-
maining inhibited as long as it is carbaminoylated. Cleavage of the phosphate residue, i.e., dephosphorylation, is practically impossible; enzyme inhibition is irreversible.
Uses. The quaternary carbamate neostigmine is employed as an indirect parasympathomimetic in postoperative atonia of the bowel or bladder. Applied topically to the eye, neostigmine is used in the treatment of glaucoma. Furthermore, it is needed to overcome the relative ACh-deficiency at the motor end plate in myasthenia gravis or to reverse the neuromuscular blockade (p.184) caused by nondepolarizing muscle relaxants (decurarization before discontinuation of anaesthesia). Pyridostigmine has a similar use. The tertiary carbamate physostigmine can be used as an antidote in poisoning with parasympatholytic drugs, because it has access to AChE in the brain. Carbamates and organophosphates also serve as insecticides. Although they possess high acute toxicity in humans, they are more rapidly degraded than is DDT following their release into the environment.
In the early stages of Alzheimer disease, administration of centrally acting AChE inhibitors can bring about transient improvement in cognitive function or slow down deterioration in some patients. Suitable drugs include rivastigmine, donezepil, and galantamine, which require slowly increasing dosage. Peripheral side effects (inhibition of ACh breakdown) limit therapy. Donezepil and galantamine are not esters of carbamic acid and act by a different molecular action. Galantamine is also thought to promote the action of ACh at nicotinic cholinoceptors by an allosteric mechanism.
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.

|
|
|
|
|
|
|
Parasympathomimetics |
|
107 |
||||
A. Direct and indirect parasympathomimetics |
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
O |
|
|
|
|
CH3 |
|
|
|
H CO |
C |
|
|
|
|
|
|
|
|
|
|
|
|
3 |
|
|
|
H N |
C |
O |
CH |
CH |
+N |
CH |
|
|
|
|
|
|
|
2 |
|
|
2 |
2 |
|
|
3 |
|
|
|
|
N |
CH3 |
Carbachol |
|
|
|
CH3 |
|
|
Arecoline |
|
|||||
|
|
|
Direct parasympatho- |
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
mimetics |
|
Arecoline= |
|
|
|
|
|
O |
|
|
|
|
CH3 |
|
|
|
|
|
||
|
|
|
|
|
|
|
ingredient of |
|
|
||||
H3C C O CH2 |
CH2 |
+ |
N CH3 |
|
betel nut: |
|
|
|
|||||
|
|
betel |
|
|
|
||||||||
Acetylcholine |
|
|
CH3 |
|
|
|
|
|
|||||
|
|
|
|
chewing |
|
|
|
||||||
|
|
|
|
|
|
|
AChE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H3C |
|
|
|
|
|
|
|
|
|
|
|
ACh |
|
N |
CH CH3 |
|
|
||
|
|
|
|
|
|
|
organ |
|
|
|
|||
|
|
|
|
|
|
|
H3C |
|
|
CH3 |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Effector |
|
|
O |
C |
N |
|
|
|
|
|
|
|
|
Rivastigmine |
O |
CH2 |
CH3 |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3 |
|
|
H |
|
|
|
|
|
|
|
|
|
|
O |
C |
N |
|
|
|
|
|
|
|
|
|
N |
|
||||
|
|
|
|
|
|
|
|
|
|
O |
CH3 |
||
|
|
|
|
|
|
|
H3C |
|
N |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
H3C |
Physostigmine |
|
||
|
|
|
|
|
|
|
AChE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H3C |
O |
|
H3C |
|
|
Inhibitors of |
|
OC2H5 |
|
|
|
||
|
CH3 |
acetylcholinesterase |
|
|
|
|
|
|
|||||
|
|
|
|
+ |
O |
P |
O |
|
NO2 |
|
|||
N |
C |
O |
|
N |
|
|
(AChE) |
|
|
||||
H C |
|
|
|
|
CH |
Indirect |
|
OC2H5 |
|
|
|
||
3 |
Neostigmine |
|
|
3 |
|
|
Paraoxon (E 600) |
|
|||||
|
|
|
|
parasympathomimetics |
|
|
|
||||||
Acetylcholine |
|
|
|
|
|
Nitrostigmine = |
|
|
|||||
|
|
|
|
Acetyl |
Parathion = |
|
|
|
|||||
|
|
+ |
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
E 605 |
|
|
|
|
||
|
AChE |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
ms |
|
|
|
|
|
|
|
|
Choline |
|
|
|
Deacetylation |
|
|
|
|
|
|
|
Neostigmine |
|
|
|
|
Carbaminoyl |
|
|
|
|
|
|
||
|
|
+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
AChE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Hours |
|
|
|
|
|
|
|
|
|
|
|
|
|
Decarbaminoylation |
|
|
|
|
|
|
Paraoxon |
|
|
|
|
Phosphoryl |
|
|
|
|
|
|
||
|
|
+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
AChE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dephosphorylation impossible |
|
|
|
|||
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.

108 Drugs Acting on the Parasympathetic Nervous System
Parasympatholytics
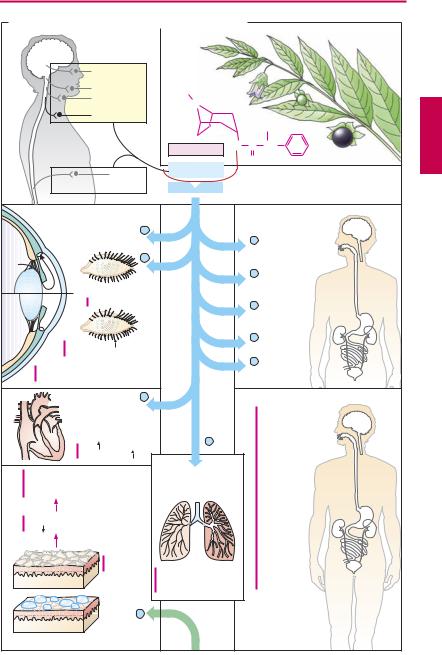
Excitation of the parasympathetic division causes release of acetylcholine at neuroeffector junctions in different target organs. The major effects are summarized in (A) (blue arrows). Some of these effects have therapeutic applications, as indicated by the clinical uses of parasympathomimetics (p.106).
Substances acting antagonistically at the M-cholinoceptor are designated parasympatholytics (prototype: the alkaloid atropine; actions marked red in the panels). Therapeutic use of these agents is complicated by their low organ selectivity. Possibilities for a targeted action include:
Local application
Selection of drugs with favorable membrane penetrability
Administration of drugs possessing receptor subtype selectivity.
Parasympatholytics are employed for the following purposes:
1. Inhibition of glandular secretion.
Bronchial secretion. Premedication with atropine before inhalation anesthesia prevents a possible hypersecretion of bronchial mucus, which cannot be expectorated by coughing during anesthesia.
Gastric secretion. Atropine displays about equally high af nity for all muscarinic cholinoceptor subtypes and thus lacks organ specificity. Pirenzepine has preferential af nity for the M1 subtype and was used to inhibit production of HCl in the gastric mucosa, because vagally mediated stimulation of acid production involves M1 receptors. This approach has proved inadequate because the required dosage of pirenzepine produced too many atropine-like side effects. Also, more effective pharmacological means are available to lower HCl production in a graded fashion (H2-antihistaminics, proton pump inhibitors).
2.Relaxation of smooth musculature. As a rule, administration of a parasympatholytic agent by inhalation is quite effective in chronic obstructive pulmonary disease. Ipratropium has a relatively short lasting effect; four aerosol puffs usually being required per day. The newly introduced substance tiotropium needs to be applied only once daily because of its “adhesiveness.”Tiotropium is effective in chronic obstructive lung disease; however, it is not indicated in the treatment of bronchial asthma.
Spasmolysis by N-butylscopolamine in biliary or renal colic (p.130). Because of its quaternarynitrogenatom,thisdrug doesnot enter the brain and requires parenteral administration. Its spasmolytic action is especially marked because of additional ganglionic blocking and direct muscle-relaxant actions.
Lowering of pupillary sphincter tonus and pupillary dilation by local administration of homatropine or tropicamide (mydriatics) allows observation of the ocular fundus. For diagnostic uses, only short-term pupillary dilation is needed. The effect of both agents subsides quickly in comparison with that of atropine (duration of several days).
3.Cardioacceleration. Ipratropium is used in bradycardia and AV-block, respectively, to raise heart rate and to facilitate cardiac impulse conduction. As a quaternary substance, it does not penetrate into the brain, which greatly reduces the risk of CNS disturbances (see below). However, it is also poorly absorbed from the gut (absorption rate < 30%). To achieve adequate levels in the blood, it must be given in significantly higher dosage than needed parenterally.
Atropine may be given to prevent cardiac arrest resulting from vagal reflex activation, incidental to anaesthetic induction, gastric lavage, or endoscopic procedures.
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.
|
|
|
|
Parasympatholytics |
109 |
|
A. Effects of parasympathetic stimulation and blockade |
|
|
|
|||
|
|
Deadly |
|
|
|
|
|
|
nightshade |
|
|
|
|
N. oculo- |
|
Atropa |
|
|
|
|
|
belladonna |
|
|
|
|
|
motorius |
|
|
|
|
|
|
N. facialis |
|
H3C |
|
|
|
|
N. glosso- |
|
|
|
|
|
|
|
N |
|
|
|
|
|
pharyngeus |
|
|
|
|
|
|
N. vagus |
|
|
|
|
|
|
|
|
|
|
|
CH2OH |
|
|
|
Atropine |
O |
C |
CH |
|
|
|
|
|
|
|
|
|
|
Acetylcholine |
|
O |
|
|
|
|
|
|
|
|
|
Nn. sacrales |
|
|
Muscarinic acetylcholine receptor |
|
||
|
|
|
|
|||
SchlemmÕs |
|
|
|
|
|
|
canal wide |
+ |
|
|
|
|
|
Ciliary muscle |
|
|
+ |
|
|
|
contracted |
|
|
|
Salivary secretion |
|
|
+ |
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
+ |
Gastric |
|
Pupil narrow |
|
|
|
|
|
|
|
|
|
|
acid |
|
|
Pupil wide |
|
|
|
+ |
|
|
|
|
|
|
Pancreatic juice |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
production |
|
Photophobia |
|
|
|
+ |
Bowel peristalsis |
|
|
|
|
|
|
||
Near vision impossible |
|
|
+ |
|
|
|
Drainage of aqueous |
|
|
|
Bladder tone |
|
|
|
|
|
|
|
||
humor impaired |
|
|
|
|
|
|
|
|
|
|
|
|
|
Rate |
- |
|
|
|
|
|
|
|
|
|
Restlessness |
|
|
AV conduction |
|
|
|
|
|
|
|
|
|
|
Irritability |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Hallucinations |
|
|
|
+ |
|
|
Antiparkinsonian |
|
Rate |
|
|
|
effect |
|
|
|
|
|
|
|
||
AV conduction |
|
|
|
|
Antiemetic effect |
|
Increased blood flow |
|
|
|
|
Dry mouth |
|
for increasing |
|
Bronchial secretion |
|
|
|
|
|
|
|
|
|
||
heat dissipation |
|
Bronchoconstriction |
|
|
Acid production |
|
|
|
|
|
|
decreased |
|
Evaporative heat |
|
|
|
|
Pancreatic |
|
loss |
|
|
|
|
secretory activity |
|
|
|
|
|
|
decreased |
|
ÒFlushed |
|
|
|
|
Bowel peristalsis |
|
|
|
|
|
decreased |
|
|
dry skinÒ |
|
Bronchial secretion |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
Bladder tone |
|
|
|
|
decreased |
|
|
|
|
|
|
Bronchodilation |
|
|
decreased |
|
+ |
Sympathetic |
|
|
|
|
|
|
|
nerves |
|
|
|
|
Sweat production |
|
|
|
|
|
|
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.

110 Drugs Acting on the Parasympathetic Nervous System
4. CNS damping effects. Scopolamine is effective in the prophylaxis of kinetosis (motion sickness, sea sickness, see p.342); it is mostly applied by a transdermal patch. Scopolamine (pKa = 7.2) penetrates the blood– brain barrier faster than does atropine (pKa = 9), because at physiological pH a larger proportion is present in the neutral, membranepermeant form.
In psychotic excitement (agitation), sedation can be achieved with scopolamine. Unlike atropine, scopolamine exerts a calming and amnesiogenic action that can also be used to advantage in anesthetic premedication.
Symptomatic treatment in parkinsonism for the purpose of restoring a dopaminer- gic-cholinergic balance in the corpus striatum. Antiparkinsonian agents, such as benztropine (p.188) readily penetrate the blood–brain barrier. At centrally equieffective dosages, their peripheral effects are less marked than those of atropine.
Contraindications for parasympatholytics.
Closed angle glaucoma. Since drainage of aqueous humor is impeded during relaxation of the pupillary sphincter, intraocular pressure rises.
Prostatic hyperplasia with impaired micturition: loss of parasympathetic control of the detrusor muscle exacerbates dif culties in voiding urine.
Atropine poisoning. Parasympatholytics have a wide therapeutic margin. Rarely lifethreatening, poisoning with atropine is characterized by the following peripheral and central effects.
Peripheral. Tachycardia; dry mouth; hyperthermia secondary to the inhibition of sweating. Although sweat glands are innervated by sympathetic fibers, these are cholinergic in nature. When sweat secretion is inhibited, the body loses the ability to dissipate metabolic heat by evaporation of sweat. There is a compensatory vasodilation in the skin, allowing increased heat ex-
change through increased cutaneous blood flow. Decreased peristaltic activity of the intestines leads to constipation.
Central. Motor restlessness, progressing to maniacal agitation, psychic disturbances, disorientation and hallucinations. It may be noted that scopolamine-containing herbal preparations (especially from Datura stramonium) served as hallucinogenic intoxicants in the Middle Ages. Accounts of witches’ rides to satanic gatherings and similar excesses are likely the products of CNS poisoning. Recently, Western youths have been reported to make “recreational” use of Angel’s Trumpet flowers (several Brugmansia species grown as ornamental shrubs). Plants of this genus are a source of scopolamine used by South American natives since pre-Columbian times.
Elderly subjects have an enhanced sensitivity, particularly toward the CNS toxic manifestations. In this context, the diversity of drugs producing atropine-like side effects should be borne in mind: e.g., tricyclic antidepressants, neuroleptics, antihistaminics, antiarrhythmics, antiparkinsonian agents.
Apart from symptomatic, general measures (gastric lavage, cooling with ice water), therapy of severe atropine intoxication includes the administration of the indirect parasympathomimetic physostigmine (p.106). The most common instances of “atropine”-intoxication are observed after ingestion of the berrylike fruits of belladonna (in children). A similar picture may be seen after intentional overdosage with tricyclic antidepressants in attempted suicide.
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.

|
|
|
|
|
|
|
|
|
Parasympatholytics |
|
111 |
||||
A. Parasympatholytics |
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
H3C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
C |
C |
OH |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Homatropine |
|
|
|
|||
|
|
|
|
|
|
|
|
|
M1 |
M1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M3 |
M1 M1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M3 |
|
|
|
|
|
|
|
H3C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
C |
H |
|
|
|
|
|
|
|
|
|
|
|
Benzatropine |
|
|
|
|
|
|
M2 |
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
H3C |
CH3 |
|
|
|
|
|
|
|
|
M1 |
M1 |
|
|
|
|
Tiotropium |
|
|
|
|
|
M |
|
|
|
|
|||||
+ |
|
|
|
|
|
|
|
|
|
||||||
N |
|
|
|
|
|
|
|
|
M1 |
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M1 |
|
|
|
|
O |
|
HO |
S |
|
|
|
|
|
|
|
M1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
O |
C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O S |
|
|
|
|
|
|
M |
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
+N |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H9C4 |
|
|
|
|
|
|
|
|
|
|
|
H3C |
|
“Patch” |
O |
|
|
O |
H |
|
||
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
N |
|
|
|
|
|
|
O |
C |
C |
CH2OH |
|
|
|
|
|
|
|
|
|
|
N-Butyl- |
|
|
|
||
|
|
|
|
|
O |
|
|
|
CH2OH |
scopolamine |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Scopolamine |
O |
C |
CH |
|
|
|
|
|
|
|
|
|
|
|
|
0.2–2 mg |
|
|
O |
|
|
|
|
|
|
|
H3C |
CH3 |
|
ED |
|
|
|
|
|
|
|
|
|
|
|
|
+N |
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
0.5–1 mg |
|
|
|
|
|
|
|
|
|
|
|||
|
CH |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H3C |
|
|
O |
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
O C C CH2OH |
|
|
|
|
|
|
|
|
|
|
|||
|
Ipratropium |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
ED 10 mg |
|
|
|
|
|
|
|
|
+ ganglioplegic |
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
+ direct muscle relaxant |
||||
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.
