
- •Preface to the 3rd edition
- •General Pharmacology
- •Systems Pharmacology
- •Therapy of Selected Diseases
- •Subject Index
- •Abbreviations
- •General Pharmacology
- •History of Pharmacology
- •Drug and Active Principle
- •The Aims of Isolating Active Principles
- •European Plants as Sources of Effective Medicines
- •Drug Development
- •Congeneric Drugs and Name Diversity
- •Oral Dosage Forms
- •Drug Administration by Inhalation
- •Dermatological Agents
- •From Application to Distribution in the Body
- •Potential Targets of Drug Action
- •External Barriers of the Body
- •Blood–Tissue Barriers
- •Membrane Permeation
- •Binding to Plasma Proteins
- •The Liver as an Excretory Organ
- •Biotransformation of Drugs
- •Drug Metabolism by Cytochrome P450
- •The Kidney as an Excretory Organ
- •Presystemic Elimination
- •Drug Concentration in the Body as a Function of Time—First Order (Exponential) Rate Processes
- •Time Course of Drug Concentration in Plasma
- •Time Course of Drug Plasma Levels during Repeated Dosing (A)
- •Time Course of Drug Plasma Levels during Irregular Intake (B)
- •Accumulation: Dose, Dose Interval, and Plasma Level Fluctuation (A)
- •Dose–Response Relationship
- •Concentration–Effect Curves (B)
- •Concentration–Binding Curves
- •Types of Binding Forces
- •Agonists—Antagonists
- •Other Forms of Antagonism
- •Enantioselectivity of Drug Action
- •Receptor Types
- •Undesirable Drug Effects, Side Effects
- •Drug Allergy
- •Cutaneous Reactions
- •Drug Toxicity in Pregnancy and Lactation
- •Pharmacogenetics
- •Placebo (A)
- •Systems Pharmacology
- •Sympathetic Nervous System
- •Structure of the Sympathetic Nervous System
- •Adrenergic Synapse
- •Adrenoceptor Subtypes and Catecholamine Actions
- •Smooth Muscle Effects
- •Cardiostimulation
- •Metabolic Effects
- •Structure–Activity Relationships of Sympathomimetics
- •Indirect Sympathomimetics
- •Types of
- •Antiadrenergics
- •Parasympathetic Nervous System
- •Cholinergic Synapse
- •Parasympathomimetics
- •Parasympatholytics
- •Actions of Nicotine
- •Localization of Nicotinic ACh Receptors
- •Effects of Nicotine on Body Function
- •Aids for Smoking Cessation
- •Consequences of Tobacco Smoking
- •Dopamine
- •Histamine Effects and Their Pharmacological Properties
- •Serotonin
- •Vasodilators—Overview
- •Organic Nitrates
- •Calcium Antagonists
- •ACE Inhibitors
- •Drugs Used to Influence Smooth Muscle Organs
- •Cardiac Drugs
- •Cardiac Glycosides
- •Antiarrhythmic Drugs
- •Iron Compounds
- •Prophylaxis and Therapy of Thromboses
- •Possibilities for Interference (B)
- •Heparin (A)
- •Hirudin and Derivatives (B)
- •Fibrinolytics
- •Intra-arterial Thrombus Formation (A)
- •Formation, Activation, and Aggregation of Platelets (B)
- •Inhibitors of Platelet Aggregation (A)
- •Presystemic Effect of ASA
- •Plasma Volume Expanders
- •Lipid-lowering Agents
- •Diuretics—An Overview
- •NaCl Reabsorption in the Kidney (A)
- •Aquaporins (AQP)
- •Osmotic Diuretics (B)
- •Diuretics of the Sulfonamide Type
- •Potassium-sparing Diuretics (A)
- •Vasopressin and Derivatives (B)
- •Drugs for Gastric and Duodenal Ulcers
- •Laxatives
- •Antidiarrheal Agents
- •Drugs Affecting Motor Function
- •Muscle Relaxants
- •Nondepolarizing Muscle Relaxants
- •Depolarizing Muscle Relaxants
- •Antiparkinsonian Drugs
- •Antiepileptics
- •Pain Mechanisms and Pathways
- •Eicosanoids
- •Antipyretic Analgesics
- •Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
- •Cyclooxygenase (COX) Inhibitors
- •Local Anesthetics
- •Opioid Analgesics—Morphine Type
- •General Anesthesia and General Anesthetic Drugs
- •Inhalational Anesthetics
- •Injectable Anesthetics
- •Sedatives, Hypnotics
- •Benzodiazepines
- •Pharmacokinetics of Benzodiazepines
- •Therapy of Depressive Illness
- •Mania
- •Therapy of Schizophrenia
- •Psychotomimetics (Psychedelics, Hallucinogens)
- •Hypothalamic and Hypophyseal Hormones
- •Thyroid Hormone Therapy
- •Glucocorticoid Therapy
- •Follicular Growth and Ovulation, Estrogen and Progestin Production
- •Oral Contraceptives
- •Antiestrogen and Antiprogestin Active Principles
- •Aromatase Inhibitors
- •Insulin Formulations
- •Treatment of Insulin-dependent Diabetes Mellitus
- •Treatment of Maturity-Onset (Type II) Diabetes Mellitus
- •Oral Antidiabetics
- •Drugs for Maintaining Calcium Homeostasis
- •Drugs for Treating Bacterial Infections
- •Inhibitors of Cell Wall Synthesis
- •Inhibitors of Tetrahydrofolate Synthesis
- •Inhibitors of DNA Function
- •Inhibitors of Protein Synthesis
- •Drugs for Treating Mycobacterial Infections
- •Drugs Used in the Treatment of Fungal Infections
- •Chemotherapy of Viral Infections
- •Drugs for the Treatment of AIDS
- •Drugs for Treating Endoparasitic and Ectoparasitic Infestations
- •Antimalarials
- •Other Tropical Diseases
- •Chemotherapy of Malignant Tumors
- •Targeting of Antineoplastic Drug Action (A)
- •Mechanisms of Resistance to Cytostatics (B)
- •Inhibition of Immune Responses
- •Antidotes and Treatment of Poisonings
- •Therapy of Selected Diseases
- •Hypertension
- •Angina Pectoris
- •Antianginal Drugs
- •Acute Coronary Syndrome— Myocardial Infarction
- •Congestive Heart Failure
- •Hypotension
- •Gout
- •Obesity—Sequelae and Therapeutic Approaches
- •Osteoporosis
- •Rheumatoid Arthritis
- •Migraine
- •Common Cold
- •Bronchial Asthma
- •Emesis
- •Alcohol Abuse
- •Local Treatment of Glaucoma
- •Further Reading
- •Further Reading
- •Picture Credits
- •Drug Indexes

146 Antithrombotics
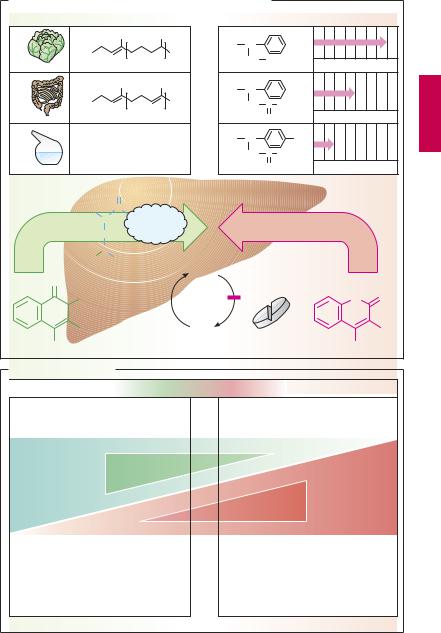
VitaminKAntagonistsand VitaminK
Vitamin K promotes the hepatic γ-carboxy- lation of glutamate residues on the precursors of factors II, VII, IX, and X. Carboxyl groups are required for Ca2+-mediated binding to phospholipid surfaces (p.144). There are several vitamin K derivatives of different origins: K1 (phytomenadione) from chlorophyllous plants; K2 from gut bacteria; and K3 (menadione) synthesized chemically. All are hydrophobic and require bile acids for absorption.
Oral anticoagulants. Structurally related to vitamin K, 4-hydroxycoumarins act as ”false” vitamin K and prevent regeneration of reduced (active) vitamin K from vitamin K epoxide, hence the synthesis of vitamin K- dependent clotting factors
Coumarins are well absorbed after oral administration. Their duration of action varies considerably. Synthesis of clotting factors depends on the intrahepatocytic concentration ratio between coumarins and vitamin K. The dose required for an adequate anticoagulant effect must be determined individually for each patient (monitoring of the International Normalized Ratio, INR).
Indications. Hydroxycoumarins are used for the prophylaxis of thromboembolism as, for instance, in atrial fibrillation or after heart valve replacement.
The most important adverse effect is bleeding. With coumarins, this can be counteracted by giving vitamin K1. However, coagulability of blood returns to normal only after hours or days, when the liver has resumed synthesis and restored suf cient blood levels of carboxylated clotting factors. In urgent cases, deficient factors must be replenished directly (e.g., by transfusion of whole bloodor ofprothrombin concentrate).
Other notable adverse effects include: at the start of therapy, hemorrhagic skin necroses and alopecia; with exposure in utero, disturbances of fetal cartilage and bone for-
mation and CNS injury (due to bleeding); enhanced risk of retroplacental bleeding.
Possibilities for Interference (B)
Adjusting the dosage of a hydroxycoumarin calls for a delicate balance between the opposing risks of bleeding (effect too strong) and of thrombosis (effect too weak). After the dosage has been titrated successfully, loss of control may occur if certain interfering factorsare ignored. If the patientchanges dietary habits and consumes more vegetables, vitamin K may predominate over the vitamin K antagonist. If vitamin K-producing gut flora is damaged in the course of antibiotic therapy, the antagonist may prevail. Drugs that increase hepatic biotransformation via enzyme induction (p.38) may accelerate elimination of a hydroxycoumarin and thus lower its blood level. Inhibitors of hepatic biotransformation (e.g., the H2 blocker cimetidine) augment the action of hydroxycoumarins. Apart from pharmacokinetic alterations, pharmacodynamic interactions must be taken into account. Thus, acetylsalicylic acid is contraindicated because (a) it retards hemostasis by inhibiting platelet aggregation and (b) itmaycause damage tothe gastric mucosawith erosion of blood vessels.
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.
Vitamin K Antagonists and Vitamin K |
147 |
A. Vitamin K-antagonists of the coumarin type and vitamin K |
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
Duration of action/days |
||
|
|
|
CH3 |
|
CH3 |
R = |
CH |
|
|
|
|
|
|
R = |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
CH3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH2 |
CH3 |
|
|
|
||
Vit. K1 |
Phytomenadione |
3 |
|
Phenprocoumon |
||||||||
|
|
|
|
|
||||||||
|
|
|
CH3 |
|
CH3 |
R = |
CH |
|
|
|
|
|
|
R = |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
CH3 |
|
CH2 |
C |
CH3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Vit. K2 |
|
|
|
|
1 – 12 |
|
|
O |
|
Warfarin |
|
|
|
|
|
|
|
|
|
|
|
||||
|
R = H |
|
|
|
|
R = |
CH |
|
|
NO2 |
|
|
|
|
|
|
|
|
CH2 C CH3 |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Vit. K3 |
Menadione |
|
|
|
|
O |
|
Acenocoumarol |
||||
|
|
|
O |
Carboxylation of glutamine residues |
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H2N |
CH |
C |
|
|
|
|
|
|
|
|
|
|
|
II, VII, IX, X |
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
CH2 |
|
|
|
|
|
|
|
|
|
|
|
HOOC |
CH |
|
|
|
|
|
|
|
|
|
|
|
|
COOH |
|
Vit. K- |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
Epoxid |
|
|
|
|
|
|
|
CH3 |
|
|
|
|
|
|
|
|
|
O |
O |
|
R |
|
|
|
|
Vit. K |
4-Hydroxy- |
|
|
R |
||
OH |
|
|
|
|
|
|
|
OH |
|
|||
Vit. K derivates |
|
|
coumarin derivatives |
|
||||||||
B. Possible interactions
Risk of thrombosis |
Optimal adjustment |
Risk of bleeding |
|
|
|
Increased intake of vitamin K-rich food
Increase
Vitamin K effect
Decrease
Inhibition of enteral coumarin absorption by adsorbents, e.g., antacids, medicinal charcoal
Acceleration of hepatic coumarin metabolism: enzyme induction, e.g., by carbamazepine, rifampicin
Damage to vitamin K-producing intestinal bacteria by antibiotics
Decrease
Hydroxycoumarin effect
Increase
Inhibition of hepatic coumarin metabolism, e.g., by cimetidine, metronidazole
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.

148 Antithrombotics
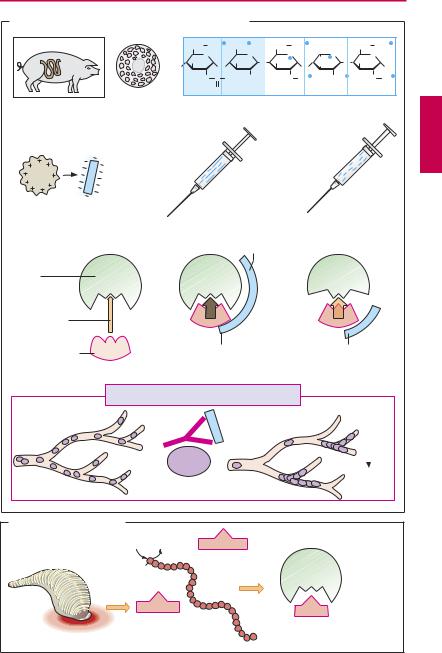
Heparin (A)
Occurrence and structure. Heparin can be obtained from porcine gut, where it is present (together with histamine) in storage vesicles of mast cells. Heparin molecules are chains of amino sugars bearing –COO– and –SO3– groups. Chain length is not constant and anticoagulant ef cacy varies with chain length. The potency of a preparation is standardized in international units of activity (IU) by bioassay and comparison with a reference preparation. The molecular weight (MW)for unfractionatedheparinrangesfrom 4000 to 40 000, with a peak around 15 000. Low-molecular-weight fractionated heparin can be produced by cleavage of native heparin; molecular size is less heterogeneous, with a mean MW of 5000 (e.g., certoparin, dalteparin, enoxaparin).The synthetic fondaparinux (MW 1728) resembles the basic pentasaccharide subunit of heparin, essential for activity. The numerous negative charges are significant in several respects: (1) they contribute to complex formation with antithrombin III that underlies the anticoagulant effect; (2) they permit binding of heparin to its antidote, protamine (a polycationic protein from salmon sperm); (3) they confer poor membrane penetrability, necessitating administration of heparin by injection.
Mechanism of action. Antithrombin III (AT III) is a circulating glycoprotein capable of inhibiting activated clotting factors by occupation andirreversibleblockade ofthe active center. Heparin acts to inhibit clotting by accelerating formation of this complex more than 1000-fold. Activated clotting factors have differing requirements for optimal chain length of heparin. For instance, to inactivate thrombin, the heparin molecule must simultaneously contact the factor and AT III. With factor Xa, however, contact between heparin and AT III is suf cient for speeding up inactivation.
Indications. Heparin is used for the prophylaxis and therapy of thrombosis. For the former, low dosages, given subcutaneously, are suf cient. Unfractionated heparin must be injected about three time daily, fractionated heparins and fondaparinux can be administered once daily. For treatment of thrombosis, heparin must be infused intravenously in an increased daily dose.
Adverse effects. When bleeding is induced by heparin, the heparin action can be instantly reversed by protamine. Against fractionated heparins and fondaparinux, protamine is less or not effective. Heparin-in- duced thrombocytopenia type II (HIT II) is a dangerous complication. It results from formation of antibodies that precipitate with bound heparin on platelets. The platelets aggregate and give rise to vascular occlusions. Because of the thrombocytopenia, hemorrhages may occur. Fondaparinux is also contraindicated in HIT II.
The drug danaparoid consists mostly of the heparinoid heparan sulfate. Its chains are composed of a part of the heparin molecule (indicated by blue color underlay). Its effect is mediated by AT III.
Hirudin and Derivatives (B)
The polypeptide hirudin from the saliva of the European medicinal leech inhibits clotting of the leech’s blood meal by blockade of the active center of thrombin. This action is independent of AT III and thus also occurs in patients with AT III deficiency. Lepirudin and desrudin are yeast-derivedrecombinant analogues. They can be used in patients with HIT II.
Ximelagatran is a modified thrombin antagonist suitable for oral use.
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.
Heparin 149
A. Heparins: origin, structure, and mechanism of action
CH2 OSO3– |
COO– |
CH2 OSO3– |
|
|
CH2 OSO3– |
||||
O |
|
|
O |
O |
|
COO |
–O |
O |
|
OH |
|
OH |
O |
– |
|
O |
OH |
|
|
O |
OSO3 |
O |
OH |
O |
|||||
O |
|
|
|
|
|
|
|||
HN |
C |
CH3 |
OH |
HN |
SO3– |
|
OSO3– |
HN |
SO3– |
|
O |
|
|
|
|
|
|
|
|
Mast cell |
Pentasaccharide basic unit |
Antidote protamine
Activated clotting factor
Inactivation
Antithrombin III
Standard heparin |
Low-MW heparin |
unfractionated |
fractionated |
mean |
mean MW ~5000 |
MW ~15 000 |
|
~3x daily s.c. |
~once daily s.c. |
|
Acceleration |
|
of inactivation |
IIa |
Xa |
Thrombin |
|
Standard heparin |
Low-MW heparin |
required |
sufficient |
Heparin-induced thrombocytopenia type II |
|
|
Platelet |
Antibody |
aggregation |
Heparin |
|
|
Thrombo- |
Platelet |
embolism |
|
|
B. Hirudin and derivatives |
|
|
|
Leucine |
Isoleucine: |
Lepirudin |
|
H2N |
|
IIa |
Direct |
|
Thrombin |
||
|
|
selective |
|
|
|
|
inhibition |
Hirudin |
|
|
|
Hirudo medicinalis |
|
|
|
Luellmann, Color Atlas of Pharmacology © 2005 Thieme
All rights reserved. Usage subject to terms and conditions of license.
