
- •ICU Protocols
- •Preface
- •Acknowledgments
- •Contents
- •Contributors
- •1: Airway Management
- •Suggested Reading
- •2: Acute Respiratory Failure
- •Suggested Reading
- •Suggested Reading
- •Website
- •4: Basic Mechanical Ventilation
- •Suggested Reading
- •Suggested Reading
- •Websites
- •Suggested Reading
- •Websites
- •7: Weaning
- •Suggested Reading
- •8: Massive Hemoptysis
- •Suggested Reading
- •9: Pulmonary Thromboembolism
- •Suggested Reading
- •Suggested Reading
- •Websites
- •11: Ventilator-Associated Pneumonia
- •Suggested Readings
- •12: Pleural Diseases
- •Suggested Reading
- •Websites
- •13: Sleep-Disordered Breathing
- •Suggested Reading
- •Websites
- •14: Oxygen Therapy
- •Suggested Reading
- •15: Pulse Oximetry and Capnography
- •Conclusion
- •Suggested Reading
- •Websites
- •16: Hemodynamic Monitoring
- •Suggested Reading
- •Websites
- •17: Echocardiography
- •Suggested Readings
- •Websites
- •Suggested Reading
- •Websites
- •19: Cardiorespiratory Arrest
- •Suggested Reading
- •Websites
- •20: Cardiogenic Shock
- •Suggested Reading
- •21: Acute Heart Failure
- •Suggested Reading
- •22: Cardiac Arrhythmias
- •Suggested Reading
- •Website
- •23: Acute Coronary Syndromes
- •Suggested Reading
- •Website
- •Suggested Reading
- •25: Aortic Dissection
- •Suggested Reading
- •26: Cerebrovascular Accident
- •Suggested Reading
- •Websites
- •27: Subarachnoid Hemorrhage
- •Suggested Reading
- •Websites
- •28: Status Epilepticus
- •Suggested Reading
- •29: Acute Flaccid Paralysis
- •Suggested Readings
- •30: Coma
- •Suggested Reading
- •Suggested Reading
- •Websites
- •32: Acute Febrile Encephalopathy
- •Suggested Reading
- •33: Sedation and Analgesia
- •Suggested Reading
- •Websites
- •34: Brain Death
- •Suggested Reading
- •Websites
- •35: Upper Gastrointestinal Bleeding
- •Suggested Reading
- •36: Lower Gastrointestinal Bleeding
- •Suggested Reading
- •37: Acute Diarrhea
- •Suggested Reading
- •38: Acute Abdominal Distension
- •Suggested Reading
- •39: Intra-abdominal Hypertension
- •Suggested Reading
- •Website
- •40: Acute Pancreatitis
- •Suggested Reading
- •Website
- •41: Acute Liver Failure
- •Suggested Reading
- •Suggested Reading
- •Websites
- •43: Nutrition Support
- •Suggested Reading
- •44: Acute Renal Failure
- •Suggested Reading
- •Websites
- •45: Renal Replacement Therapy
- •Suggested Reading
- •Website
- •46: Managing a Patient on Dialysis
- •Suggested Reading
- •Websites
- •47: Drug Dosing
- •Suggested Reading
- •Websites
- •48: General Measures of Infection Control
- •Suggested Reading
- •Websites
- •49: Antibiotic Stewardship
- •Suggested Reading
- •Website
- •50: Septic Shock
- •Suggested Reading
- •51: Severe Tropical Infections
- •Suggested Reading
- •Websites
- •52: New-Onset Fever
- •Suggested Reading
- •Websites
- •53: Fungal Infections
- •Suggested Reading
- •Suggested Reading
- •Website
- •55: Hyponatremia
- •Suggested Reading
- •56: Hypernatremia
- •Suggested Reading
- •57: Hypokalemia and Hyperkalemia
- •57.1 Hyperkalemia
- •Suggested Reading
- •Website
- •58: Arterial Blood Gases
- •Suggested Reading
- •Websites
- •59: Diabetic Emergencies
- •59.1 Hyperglycemic Emergencies
- •59.2 Hypoglycemia
- •Suggested Reading
- •60: Glycemic Control in the ICU
- •Suggested Reading
- •61: Transfusion Practices and Complications
- •Suggested Reading
- •Websites
- •Suggested Reading
- •Website
- •63: Onco-emergencies
- •63.1 Hypercalcemia
- •63.2 ECG Changes in Hypercalcemia
- •63.3 Superior Vena Cava Syndrome
- •63.4 Malignant Spinal Cord Compression
- •Suggested Reading
- •64: General Management of Trauma
- •Suggested Reading
- •65: Severe Head and Spinal Cord Injury
- •Suggested Reading
- •Websites
- •66: Torso Trauma
- •Suggested Reading
- •Websites
- •67: Burn Management
- •Suggested Reading
- •68: General Poisoning Management
- •Suggested Reading
- •69: Syndromic Approach to Poisoning
- •Suggested Reading
- •Websites
- •70: Drug Abuse
- •Suggested Reading
- •71: Snakebite
- •Suggested Reading
- •72: Heat Stroke and Hypothermia
- •72.1 Heat Stroke
- •72.2 Hypothermia
- •Suggested Reading
- •73: Jaundice in Pregnancy
- •Suggested Reading
- •Suggested Reading
- •75: Severe Preeclampsia
- •Suggested Reading
- •76: General Issues in Perioperative Care
- •Suggested Reading
- •Web Site
- •77.1 Cardiac Surgery
- •77.2 Thoracic Surgery
- •77.3 Neurosurgery
- •Suggested Reading
- •78: Initial Assessment and Resuscitation
- •Suggested Reading
- •79: Comprehensive ICU Care
- •Suggested Reading
- •Website
- •80: Quality Control
- •Suggested Reading
- •Websites
- •81: Ethical Principles in End-of-Life Care
- •Suggested Reading
- •82: ICU Organization and Training
- •Suggested Reading
- •Website
- •83: Transportation of Critically Ill Patients
- •83.1 Intrahospital Transport
- •83.2 Interhospital Transport
- •Suggested Reading
- •84: Scoring Systems
- •Suggested Reading
- •Websites
- •85: Mechanical Ventilation
- •Suggested Reading
- •86: Acute Severe Asthma
- •Suggested Reading
- •87: Status Epilepticus
- •Suggested Reading
- •88: Severe Sepsis and Septic Shock
- •Suggested Reading
- •89: Acute Intracranial Hypertension
- •Suggested Reading
- •90: Multiorgan Failure
- •90.1 Concurrent Management of Hepatic Dysfunction
- •Suggested Readings
- •91: Central Line Placement
- •Suggested Reading
- •92: Arterial Catheterization
- •Suggested Reading
- •93: Pulmonary Artery Catheterization
- •Suggested Reading
- •Website
- •Suggested Reading
- •95: Temporary Pacemaker Insertion
- •Suggested Reading
- •96: Percutaneous Tracheostomy
- •Suggested Reading
- •97: Thoracentesis
- •Suggested Reading
- •98: Chest Tube Placement
- •Suggested Reading
- •99: Pericardiocentesis
- •Suggested Reading
- •100: Lumbar Puncture
- •Suggested Reading
- •Website
- •101: Intra-aortic Balloon Pump
- •Suggested Reading
- •Appendices
- •Appendix A
- •Appendix B
- •Common ICU Formulae
- •Appendix C
- •Appendix D: Syllabus for ICU Training
- •Index

Onco-emergencies |
63 |
|
|
Atul Kulkarni and Vandana Agarwal |
|
A 58-year-old male patient with metastatic renal cell carcinoma presented with lethargy, confusion, anorexia, nausea, and constipation. He was polyuric and polydipsic over the past few days.
63.1Hypercalcemia
Oncological emergencies such as hypercalcemia, tumor lysis syndrome, superior vena cava syndrome, and spinal cord compression are occasionally seen as an intercurrent problem or presenting manifestation in certain cancers.
Step 1: Resuscitate
•Hydration is of utmost importance in these patients, and intravenous saline should be given rapidly once hypercalcemia is confirmed (refer to Chap. 78).
Step 2: Send investigations
•Measure ionized serum calcium (arterial or venous).
•If total serum calcium is measured, correct for the albumin level. Corrected calcium = measured total calcium + [0.8 × (4.0−albumin)].
•Also, check serum creatinine, electrolytes, and alkaline phosphatase.
•A low serum chloride level (<100 mEq/L) suggests hypercalcemia of malignancy.
•ECG abnormalities reflect altered transmembrane potentials, affecting conduction such as QT interval shortening (common) and QRS interval lengthening (high levels). T waves may flatten or invert, and a variable degree of heart block may develop.
A. Kulkarni, M.D. (*) • V. Agarwal, M.D., F.R.C.A.
Department of Anaesthesia, Critical Care and Pain, Tata Memorial Hospital, Mumbai, India
e-mail: kaivalyaak@yahoo.co.in
R. Chawla and S. Todi (eds.), ICU Protocols: A stepwise approach, |
499 |
DOI 10.1007/978-81-322-0535-7_63, © Springer India 2012 |
|
500 |
A. Kulkarni and V. Agarwal |
|
|
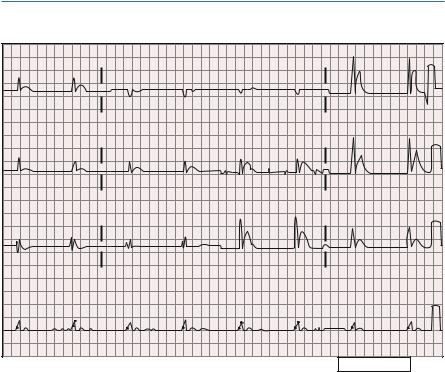
63.2ECG Changes in Hypercalcemia
I |
|
aVR |
|
V1 |
|
V4 |
|
II |
|
aVL |
|
V2 |
|
V5 |
|
III |
|
aVF |
|
V3 |
|
V6 |
|
II |
|
|
|
|
|
|
|
Device |
Speed |
25 mm / sec |
L1 ab |
10 aa / aV |
Chest |
10 aa / aV 40-0.5-4.0 W1 V |
1w0701 |
The above ECG shows a short QT interval with hardly any ST segment, characteristic of hypercalcemia. In this tracing, the QRS complexes are wide, indicative of an intraventricular conduction defect. No P waves are evident, and this is most likely a junctional escape rhythm.
Step 3: Infuse fluid
•Severe hypercalcemia is usually associated with marked hypovolemia.
•Give 500–1,000 mL of normal saline in the first hour and continue at a lower rate until volume repletion is achieved and urine output is established.
•In patients with impaired cardiorespiratory and renal function, aggressive fluid resuscitation should be done with close hemodynamic monitoring.
Step 4: Start diuretics after fluid repletion
•Consider loop diuretics only when euvolemia is achieved, as hypovolemia causes renal hypoperfusion hampering calcium excretion.
•Diuretics are specifically useful if features of hypervolemia, secondary to aggressive fluid resuscitation, develop.

63 Onco-emergencies |
501 |
|
|
|
|
Table 63.1 Treatment for hypercalcemia |
|
|
Intervention |
Dosage |
Comment |
Normal saline |
250–500 mL/h until euvolemic, thereafter |
Caution in patients with |
|
100–150 mL/h IV, may require 3–4 L |
congestive heart failure |
|
Keep urine output 100–150 mL/h |
|
Furosemide |
20–40 mg IV |
After volume correction |
Bisphosphonates |
Pamidronate: 60–90 mg IV over 2–24 hours |
Caution in renal impairment |
|
Zoledronic acid: 4 mg IV over 15 min |
|
Glucocorticoids |
Prednisolone: 60 mg/day PO |
Hyperglycemia, |
|
Hydrocortisone: 100 mg IV every 6 h |
immunosuppression |
Calcitonin |
4–8 IU/kg SC or IM every 12 h |
Rapid onset but short lived |
Step 5: Start specific therapy
•Bisphosphonates block osteoclastic bone resorption.
•Use zoledronic acid with caution in patients with renal impairment, and adjust the dose according to creatinine clearance (Table 63.1).
•Subcutaneous or intramuscular calcitonin lowers calcium levels quickly, but the effect is short lived.
•In some patients with lymphomas, particularly Hodgkin’s disease, hypercalce-
mia is caused by elevated levels of vitamin D (1,25(OH)2D); glucocorticoids are particularly effective.
Step 6: Decrease intake
•Eliminate dietary sources of calcium.
•Discontinue medications such as thiazide diuretics (increase the reabsorption of calcium) and vitamin D that increase the calcium level.
Step 7: Consider dialysis
•Dialysis should be considered for patients with renal failure and/or congestive heart failure when aggressive hydration and bisphosphonates cannot be used safely.
Step 8: Treat the cause
•Treat the malignancy with chemotherapy and radiation to control the hypercalcemia if possible.
Step 9: Evaluate prognosis
•In patients with advanced malignancy, hypercalcemia may commonly occur.
•In such circumstances, it may be appropriate, ethical, and humane to institute only comfort measures if no effective treatment for malignancy exists (Fig. 63.1).

502 |
A. Kulkarni and V. Agarwal |
|
|
High index of suspicion
Measure serum calcium
Mild hypercalcemia 11 to 12 mg/dL
Asymptomatic
Hydration with saline Eliminate dietary sources of calcium and thiazide diuretics
Moderate hypercalcemia |
Severe hypercalcemia |
|
12 to 18 mg/dL |
||
> 18 mg/dL |
||
|
||
Symptomatic |
Hydration with saline |
|
|
||
|
Loop diuretics |
|
|
Calcitonin |
|
|
Bisphosphonates |
|
|
Steriods if tumour responsive |
|
|
Hemodialysis |
|
Hydration with saline |
Eliminate dietary sources of |
|
calcium and thiazide diuretics |
||
Loop diuretics when |
||
|
||
euvolaemic |
|
|
Bisphosphonate |
|
|
Eliminate dietary |
|
|
sources of calcium |
Measure serum calcium and |
|
and thiazide diuretics |
||
other electrolytes following |
||
|
||
|
treatment particularly serum |
|
|
potassium at least twice a day till |
|
|
calcium levels start decreasing |
Fig. 63.1 Management of hypercalcemia
Tumor Lysis Syndrome
A 26-year-old patient with Burkitt’s lymphoma recently started on chemotherapy presented with anorexia, lethargy, disorientation, vomiting, muscle cramps, tachypnea, and decreased urine output.
Step 1: Resuscitate
•These patients are usually dehydrated and will benefit with intravenous fluids (refer to Chap. 78).
Step 2: Make a diagnosis
•Investigations may show hyperuricemia, hyperkalemia, hyperphosphatemia, hypocalcemia, uremia, and raised lactate dehydrogenase levels.
63 Onco-emergencies |
503 |
|
|
•Obtain electrocardiogram to rule out serious arrhythmias and conduction abnormalities.
•Tumor lysis is associated with malignancies like acute lymphoblastic leukemia or Burkitt’s lymphoma with high tumor burden and responds rapidly to chemotherapy.
•It usually follows chemotherapy, but may occur after radiation, corticosteroid therapy, or chemoembolization and rarely spontaneously.
•Check urine output and renal function.
Step 3: Start hydration
•A high infusion rate of fluids is appropriate.
•Patients with high risk of tumor lysis syndrome should have aggressive volume replacement as a preventive measure prior to chemotherapy.
•Infuse isotonic fluid at a rate of 200–300 mL/h.
•Volume should be adapted for the patient’s age, cardiac function, and urine output.
•Increasing the urinary flow rate is the most effective strategy for preventing urate-induced obstructive uropathy.
•Urine output should be maintained within a range of 80–100 mL/m2/h (4–6 mL/ kg/h if <10 kg).
•Urine-specific gravity should be maintained at £1.010.
Step 4: Use diuretics cautiously
•Maintain adequate urine output.
•It is contraindicated if hypovolemia or obstructive uropathy exists.
Step 5: Alkalinization of the urine
•This was recommended earlier but is controversial as it can lead to the formation of urinary xanthine crystals.
•This may cause obstruction of renal tubules if allopurinol is used concurrently.
•It is not recommended with recombinant urate oxidase (rasburicase).
Step 6: Start allopurinol
•Purine catabolism results in the production of hypoxanthine and xanthine, which are metabolized to uric acid via the enzymatic action of xanthine oxidase.
•This pathway can be blocked by the use of allopurinol, a hypoxanthine analog that competitively inhibits xanthine oxidase.
•Start allopurinol PO at 600 mg/day if uric acid is less than 8 mg/dL.
•Allopurinol should be started prior to chemotherapy.
•After about 2–3 days, allopurinol therapy results in increased excretion of both hypoxanthine, which is more soluble than uric acid, and xanthine, which is less soluble than uric acid.
•A marked increase in xanthine excretion can occur when allopurinol is given for prevention of tumor lysis syndrome and may lead to acute renal failure or xanthine stones.
