
- •ICU Protocols
- •Preface
- •Acknowledgments
- •Contents
- •Contributors
- •1: Airway Management
- •Suggested Reading
- •2: Acute Respiratory Failure
- •Suggested Reading
- •Suggested Reading
- •Website
- •4: Basic Mechanical Ventilation
- •Suggested Reading
- •Suggested Reading
- •Websites
- •Suggested Reading
- •Websites
- •7: Weaning
- •Suggested Reading
- •8: Massive Hemoptysis
- •Suggested Reading
- •9: Pulmonary Thromboembolism
- •Suggested Reading
- •Suggested Reading
- •Websites
- •11: Ventilator-Associated Pneumonia
- •Suggested Readings
- •12: Pleural Diseases
- •Suggested Reading
- •Websites
- •13: Sleep-Disordered Breathing
- •Suggested Reading
- •Websites
- •14: Oxygen Therapy
- •Suggested Reading
- •15: Pulse Oximetry and Capnography
- •Conclusion
- •Suggested Reading
- •Websites
- •16: Hemodynamic Monitoring
- •Suggested Reading
- •Websites
- •17: Echocardiography
- •Suggested Readings
- •Websites
- •Suggested Reading
- •Websites
- •19: Cardiorespiratory Arrest
- •Suggested Reading
- •Websites
- •20: Cardiogenic Shock
- •Suggested Reading
- •21: Acute Heart Failure
- •Suggested Reading
- •22: Cardiac Arrhythmias
- •Suggested Reading
- •Website
- •23: Acute Coronary Syndromes
- •Suggested Reading
- •Website
- •Suggested Reading
- •25: Aortic Dissection
- •Suggested Reading
- •26: Cerebrovascular Accident
- •Suggested Reading
- •Websites
- •27: Subarachnoid Hemorrhage
- •Suggested Reading
- •Websites
- •28: Status Epilepticus
- •Suggested Reading
- •29: Acute Flaccid Paralysis
- •Suggested Readings
- •30: Coma
- •Suggested Reading
- •Suggested Reading
- •Websites
- •32: Acute Febrile Encephalopathy
- •Suggested Reading
- •33: Sedation and Analgesia
- •Suggested Reading
- •Websites
- •34: Brain Death
- •Suggested Reading
- •Websites
- •35: Upper Gastrointestinal Bleeding
- •Suggested Reading
- •36: Lower Gastrointestinal Bleeding
- •Suggested Reading
- •37: Acute Diarrhea
- •Suggested Reading
- •38: Acute Abdominal Distension
- •Suggested Reading
- •39: Intra-abdominal Hypertension
- •Suggested Reading
- •Website
- •40: Acute Pancreatitis
- •Suggested Reading
- •Website
- •41: Acute Liver Failure
- •Suggested Reading
- •Suggested Reading
- •Websites
- •43: Nutrition Support
- •Suggested Reading
- •44: Acute Renal Failure
- •Suggested Reading
- •Websites
- •45: Renal Replacement Therapy
- •Suggested Reading
- •Website
- •46: Managing a Patient on Dialysis
- •Suggested Reading
- •Websites
- •47: Drug Dosing
- •Suggested Reading
- •Websites
- •48: General Measures of Infection Control
- •Suggested Reading
- •Websites
- •49: Antibiotic Stewardship
- •Suggested Reading
- •Website
- •50: Septic Shock
- •Suggested Reading
- •51: Severe Tropical Infections
- •Suggested Reading
- •Websites
- •52: New-Onset Fever
- •Suggested Reading
- •Websites
- •53: Fungal Infections
- •Suggested Reading
- •Suggested Reading
- •Website
- •55: Hyponatremia
- •Suggested Reading
- •56: Hypernatremia
- •Suggested Reading
- •57: Hypokalemia and Hyperkalemia
- •57.1 Hyperkalemia
- •Suggested Reading
- •Website
- •58: Arterial Blood Gases
- •Suggested Reading
- •Websites
- •59: Diabetic Emergencies
- •59.1 Hyperglycemic Emergencies
- •59.2 Hypoglycemia
- •Suggested Reading
- •60: Glycemic Control in the ICU
- •Suggested Reading
- •61: Transfusion Practices and Complications
- •Suggested Reading
- •Websites
- •Suggested Reading
- •Website
- •63: Onco-emergencies
- •63.1 Hypercalcemia
- •63.2 ECG Changes in Hypercalcemia
- •63.3 Superior Vena Cava Syndrome
- •63.4 Malignant Spinal Cord Compression
- •Suggested Reading
- •64: General Management of Trauma
- •Suggested Reading
- •65: Severe Head and Spinal Cord Injury
- •Suggested Reading
- •Websites
- •66: Torso Trauma
- •Suggested Reading
- •Websites
- •67: Burn Management
- •Suggested Reading
- •68: General Poisoning Management
- •Suggested Reading
- •69: Syndromic Approach to Poisoning
- •Suggested Reading
- •Websites
- •70: Drug Abuse
- •Suggested Reading
- •71: Snakebite
- •Suggested Reading
- •72: Heat Stroke and Hypothermia
- •72.1 Heat Stroke
- •72.2 Hypothermia
- •Suggested Reading
- •73: Jaundice in Pregnancy
- •Suggested Reading
- •Suggested Reading
- •75: Severe Preeclampsia
- •Suggested Reading
- •76: General Issues in Perioperative Care
- •Suggested Reading
- •Web Site
- •77.1 Cardiac Surgery
- •77.2 Thoracic Surgery
- •77.3 Neurosurgery
- •Suggested Reading
- •78: Initial Assessment and Resuscitation
- •Suggested Reading
- •79: Comprehensive ICU Care
- •Suggested Reading
- •Website
- •80: Quality Control
- •Suggested Reading
- •Websites
- •81: Ethical Principles in End-of-Life Care
- •Suggested Reading
- •82: ICU Organization and Training
- •Suggested Reading
- •Website
- •83: Transportation of Critically Ill Patients
- •83.1 Intrahospital Transport
- •83.2 Interhospital Transport
- •Suggested Reading
- •84: Scoring Systems
- •Suggested Reading
- •Websites
- •85: Mechanical Ventilation
- •Suggested Reading
- •86: Acute Severe Asthma
- •Suggested Reading
- •87: Status Epilepticus
- •Suggested Reading
- •88: Severe Sepsis and Septic Shock
- •Suggested Reading
- •89: Acute Intracranial Hypertension
- •Suggested Reading
- •90: Multiorgan Failure
- •90.1 Concurrent Management of Hepatic Dysfunction
- •Suggested Readings
- •91: Central Line Placement
- •Suggested Reading
- •92: Arterial Catheterization
- •Suggested Reading
- •93: Pulmonary Artery Catheterization
- •Suggested Reading
- •Website
- •Suggested Reading
- •95: Temporary Pacemaker Insertion
- •Suggested Reading
- •96: Percutaneous Tracheostomy
- •Suggested Reading
- •97: Thoracentesis
- •Suggested Reading
- •98: Chest Tube Placement
- •Suggested Reading
- •99: Pericardiocentesis
- •Suggested Reading
- •100: Lumbar Puncture
- •Suggested Reading
- •Website
- •101: Intra-aortic Balloon Pump
- •Suggested Reading
- •Appendices
- •Appendix A
- •Appendix B
- •Common ICU Formulae
- •Appendix C
- •Appendix D: Syllabus for ICU Training
- •Index

488 |
N. Amin and V. Patil |
|
|
Step 8: Use less allergenic blood products
•In patients with multiple blood transfusion and transfusion-related complications, alternatively processed blood products should be considered (Table 61.1).
Step 9: Consider threshold for blood transfusion
•If the bleeding has stopped and serum lactate is normal, do not transfuse any more blood or blood products.
•In the absence of active bleeding, keep a transfusion threshold of less than 7.0 g% and keep 7–9 g/dL Hb in critically ill patients who are hemodynamically stable.
•RBC transfusion may be beneficial in anemic patients with acute coronary syndrome (keep Hb >10 g/dL).
Step 10: Use blood products judiciously
•In the absence of bleeding, do not correct high INR with FFP.
•Patients having inadequate intake or on anticoagulants and broad-spectrum antibiotics are likely to have vitamin K deficiency, which can cause deranged INR.
•They will benefit from intravenous vitamin K supplementation.
Suggested Reading
1.Napolitano LM, Kurek S, American College of Critical Care Medicine of the Society of Critical Care Medicine, Eastern Association for the Surgery of Trauma Practice Management Workgroup. Clinical practice guideline: red blood cell transfusion in adult trauma and critical care. Crit Care Med. 2009;37:3124–57.
These are evidence-based guidelines on the use of RBC transfusions in adult trauma and critical care endorsed by SCCM.
2.Klein HG, Spahn DR. Series on transfusion medicine. Lancet. 2007;370:415–48.
An excellent review on red cell transfusion, platelet transfusion, and coagulation factor concentrates.
3.BCSH. Guidelines for management of massive blood loss. Br J Haematol. 2006;135:634–41.
It is an evidence-based guideline on management of massive blood loss.
Websites
1.www.transfusionguidelines.org.uk
2.www.asahq.org/publicationsAndServices/transfusion.pdf
3.www.bcshguidelines.com

Disseminated Intravascular Coagulation |
62 |
and Thrombocytopenia |
Vijaya Patil, Nayana Amin, Reshma Ambulkar,
and Atul Kulkarni
A 40-year-old male patient was admitted with acute pancreatitis. He developed fever, tachycardia, hypotension, and respiratory distress on the third day of admission. His abdomen was severely tender and distended. Next morning the nurse noticed excessive oozing from arterial and central line insertion site, and his abdomen was further distended.
Bleeding manifestation due to disseminated intravascular coagulation (DIC) occurs in 1% of hospital admission. Assessing and managing these patients require a systematic approach as DIC is a reflection of underlying systemic disease affecting the coagulation system, resulting in procoagulant activation, fibrinolytic activation, consumption coagulopathy, and end organ damage, which needs to be recognized and treated.
Step 1: Initial resuscitation
•Special emphasis should be placed on stabilizing hemodynamics, and if needed, blood and blood product transfusion should be started.
•Care should be taken in establishing venous access in actively bleeding patients who may be coagulopathic.
•Peripheral access is preferable to central.
•Use ultrasound-guided venous cannulation if possible and preferably choose compressible sites like internal jugular or femoral vein.
•Avoid arterial punctures.
V. Patil, M.D. (*) • N. Amin, M.D. • R. Ambulkar, M.D., F.R.C.A. • A. Kulkarni, M.D. Department of Anaesthesia, Critical Care & Pain, Tata Memorial Hospital,
Mumbai, India
e-mail: vijayappatil@yahoo.com
R. Chawla and S. Todi (eds.), ICU Protocols: A stepwise approach, |
489 |
DOI 10.1007/978-81-322-0535-7_62, © Springer India 2012 |
|

490 |
V. Patil et al. |
|
|
Table 62.1 Conditions associated with DIC |
|
Infections |
Bacterial—Gram-negative and Gram-positive sepsis |
|
Viral—cytomegalovirus, HIV, hepatitis, dengue |
|
Fungal |
|
Parasitic—malaria, leptospirosis |
Malignancy |
Solid tumors |
|
Hematological—acute promyelocytic leukemia is |
|
commonly associated with DIC |
Obstetric |
Amniotic fluid embolism |
|
Placenta abruption |
|
Preeclampsia |
|
Intrauterine fetal death/retained products of conception |
Toxic and immunological insults |
Viper snake bites |
|
Massive transfusion |
|
ABO transfusion incompatibility |
|
Transplant rejection |
Massive inflammation |
Severe trauma |
|
Crush injuries |
|
Massive burns |
|
Fulminant liver failure |
|
Severe hypo-/hyperthermia |
|
Severe pancreatitis |
Vascular disorders |
Aortic aneurysms |
|
Giant hemangiomas |
Step 2: Take relevant history and perform focused physical examination
•Take history of known systemic conditions associated with DIC and coagulation disorders (Table 62.1).
•Review the drug history, particularly the use of heparin and warfarin, and consumption of antiplatelet agents including nonsteroidal anti-inflammatory drugs.
•Look for bleeding manifestation, superficial like skin and mucosal (petechiae, purpura) or visceral and deep seated (gastrointestinal bleeding).
•Look for thrombotic manifestations like deep vein thrombosis (DVT) of lower limbs or venous or arterial thrombosis at any other site (e.g., cerebral).
Step 3: Investigate to ascertain the type and cause of bleeding (Table 62.2)
•Complete blood count, including platelet count and peripheral smear, for the presence of fragmented RBCs.
•Prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT).
•Fibrinogen level, fibrin degradation product (FDP), D-dimer.
•Renal and liver function tests.
•The commonest laboratory abnormality is thrombocytopenia followed by elevated FDPs, prolonged PT, prolonged APTT, and a low fibrinogen.
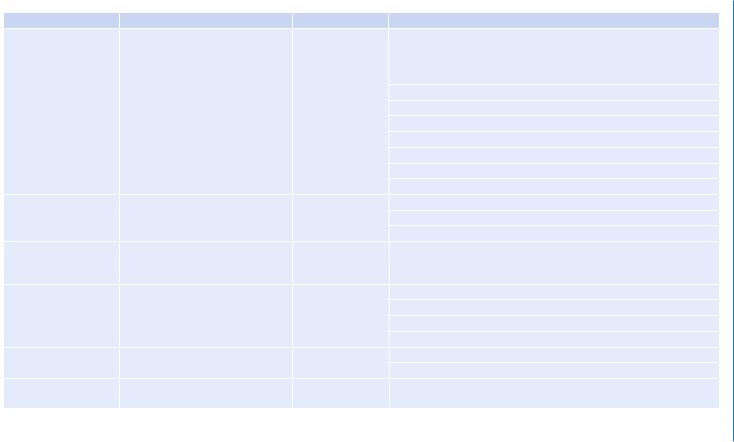
Table 62.2 Coagulation profile |
|
|
|
Test |
What does it monitor |
Normal value |
Inference |
Prothrombin time |
Factors that are in the extrinsic |
11–13 s |
Prolongation of the PT is most often a result of deficiencies in factor |
|
pathway and common pathway: |
|
VII but can also be caused by any of the extrinsic and common |
|
factors VII, X, V, and II |
|
pathway factors. Decreased fibrinogen, levels less than 100 mg/dL, |
|
|
|
will also prolong the PT |
|
|
|
Cholestatic jaundice |
|
|
|
Acute or chronic liver failure |
|
|
|
DIC |
|
|
|
Malabsorption |
|
|
|
Vitamin K deficiency |
|
|
|
Coumadin (warfarin) therapy |
|
|
|
Factors I, II,V, VII, X deficiency |
Activated partial |
Factors that are designated in the |
28–34 s |
Heparin therapy |
thromboplastin time |
intrinsic pathway: factors XII, XI, |
|
Factor deficiency |
|
IX, VIII, X, V, II, and fibrinogen |
|
Presence of an inhibitor like lupus anticoagulants |
|
|
|
|
Platelet count |
Quantifies platelet number |
130–400 × 109/L |
Decreased production (bone marrow disorder), increased destruction, |
|
|
|
idiopathic thrombocytopenic purpura (ITP), thrombotic thrombocy- |
|
|
|
topenic purpura, sequestration (hypersplenism) |
Thrombin time |
Evaluates the last step of |
13–15 s |
Heparin therapy |
|
coagulation (conversion of |
|
DIC |
|
fibrinogen to fibrin) |
|
Qualitative fibrinogen abnormalities or hypofibrinogenemia |
|
|
|
|
|
|
|
Elevated FDPs (fibrin degradation products) |
Fibrinogen level |
|
200–500 mg/dL |
Congenital and acquired hypofibrinogenemia |
|
|
|
DIC |
D-dimer |
Cross-linked D fragments of the |
500 ng/mL |
Deep venous thrombosis, DIC, pulmonary embolism, thrombolytic |
|
protein fibrinogen |
|
treatment, postoperative |
Thrombocytopenia and Coagulation Intravascular Disseminated 62
491
492 |
V. Patil et al. |
||
|
|
|
|
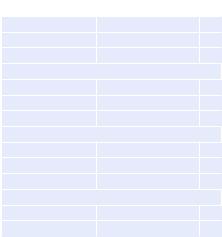
Table 62.3 ISTH diagnostic |
|
|
|
Platelet count |
|
|
|
scoring system for DIC |
>100 × 109/L |
0 |
|
|
<100 × 109/L |
1 |
|
|
<50 × 109/L |
2 |
|
|
Fibrin marker (e.g., D-dimer, FDP) |
|
|
|
No increase |
0 |
|
|
Moderate increase |
2 |
|
|
Strong increase |
3 |
|
|
Prolonged PT |
|
|
|
<3 s |
0 |
|
|
>3 but <6 s |
1 |
|
|
>6 s |
2 |
|
|
Fibrinogen level |
|
|
|
>1 g/dL |
0 |
|
|
<1 g/dL |
1 |
|
•D-dimer, FDP, and antithrombin levels can be used for rapid and specific diagnosis of DIC, with antithrombin providing an indicator for severity and prognosis.
•Diagnosis of DIC is essentially confirmed by demonstrating increased thrombin generation (decreased fibrinogen) and increased fibrinolysis (elevated D-dimer or FDP).
Step 4: Ascertain severity and prognosticate outcome
•Calculate the DIC score (Table 62.3) with the ISTH (International Society of Thrombosis and Haemostasis) scoring system which provides objective measurement of DIC and correlates with outcome.
Step 5: Continue resuscitation
•Continue resuscitation and maintain hemodynamic stability using crystalloids and/or colloids.
•In colloids, preferably use gelatins as they do not interfere with clotting.
•If you are using starches, use tetrastarch preferably, as they have less effect on the coagulation profile, but do not exceed maximum dose (50 mL/kg/day).
Step 6: Correct coagulopathy (see Table 61.2 in Chap. 61)
•Repeat the coagulation profile and complete blood count frequently and replace blood and blood products.
•In the presence of ongoing blood loss, try to normalize prothrombin time and APTT and aim to maintain platelet count of more than 50,000.
•Do not use antifibrinolytic agents as they may aggravate thrombosis.
•Patients who have DIC with a primary hyperfibrinolytic state and who have severe bleeding can be treated with lysine analogues, such as tranexamic acid (e.g., 1 g every 8 h).
•There is no role of heparin in actively bleeding patients.

62 Disseminated Intravascular Coagulation and Thrombocytopenia |
493 |
|
|
•It should be considered only where thrombosis predominates such as arterial or venous thromboembolism or severe purpura fulminans associated with vascular skin infarction.
Step 7: Treat the underlying disorder
•Repeat the tests to monitor the dynamically changing scenario and continue treatment based on clinical observation and laboratory results.
•Once patient stops bleeding, do not try to correct laboratory abnormalities as transfusion of blood and blood products should be based on clinical condition and bleeding rather than laboratory values only.
Calculate score
•More than 5 overt DIC: repeat score daily.
•Less than 5 suggestive for nonovert DIC: repeat for the next 1–2 days.
Thrombocytopenia
A 50-year-old male patient was admitted with acute pancreatitis. His blood investigations showed Hb 10.7 g%, WBC 12,000/mm3, and platelets 110,000/ mm3. On the third day, he worsened clinically. His WBC count was 20,000/mm3 and platelets were 70,000/mm3. However, the next day, he further deteriorated requiring inotropes and ventilatory support. His Hb dropped to 6.4 g%, WBC count rose to 28,000 mm3, and platelets further dropped to 40,000/mm3.
Step 1: Resuscitate
•Resuscitate, monitor, and stabilize in the ICU (refer to Chap. 78). In patients with low platelets and coagulopathy, ultrasound-guided jugular venous catheter insertion for fluid resuscitation should be performed.
•Send blood for peripheral blood smear, grouping, cross-matching, coagulation profile, and biochemistry.
Step 2: Assess severity of thrombocytopenia
• |
Thrombocytopenia is defined as a platelet count less than 150 × 109/L. |
• |
In critically ill patients, a threshold of less than100 × 109/L may be taken. |
•The ability to form a hemostatic plug is retained until the platelet count drops to less than 100 × 109/L
Step 3: Assess cause of thrombocytopenia (Table 62.4)
•Careful history, physical examination, previous medical records, and current chart review usually reveal the cause of low platelet count.
•Ask about bleeding from other sites in past, for example, frequent nosebleeds, gum bleeds, melena, hemoptysis, and blood in stool or urine.

494 |
V. Patil et al. |
|
|
Table 62.4 Causes of thrombocytopenia |
|
Pseudothrombocytopenia seen in |
EDTA causes in vitro clumping of platelets. Presence of |
asymptomatic patients |
platelet clumps in the peripheral smear and a normal repeat |
|
platelet count in citrated blood confirm pseudothrombocy- |
|
topenia. In some patients, automated blood reports show |
|
thrombocytopenia due to presence of giant platelets that are |
|
counted as RBCs in automated machines; however, manual |
|
platelet count is normal |
Dilutional thrombocytopenia |
Massive blood transfusion |
Ambulatory patients |
ITP |
|
Drug-induced—chemotherapy, miscellaneous drugs |
|
Infections—Epstein–Barr virus (EBV), HIV, others |
|
Connective tissue disorders—rheumatoid arthritis, systemic |
|
lupus erythematosus (SLE), antiphospholipid antibody |
|
syndrome |
|
Hypersplenism |
|
Primary marrow disorder |
Acutely ill patients |
Infection/sepsis |
|
DIC |
|
TTP-HUS |
|
Posttransfusion purpura |
Pregnant patient |
Gestational (platelet count >70 resolves after pregnancy) |
|
ITP |
|
HELLP—hemolysis, elevated liver enzymes, low platelets |
Cardiac patients |
HIT |
|
Cardiac bypass |
|
Dilutional |
|
Gp IIb/IIIa inhibitor-related |
|
TTP related to clopidogrel or ticlopidine |
Patient with thrombosis |
HIT |
|
Antiphospholipid antibody syndrome |
|
Paroxysmal nocturnal hemoglobinuria |
•History of previous platelet counts.
•History of previous blood or platelet transfusion.
•Medication history and review medication chart—particularly, use of heparin, warfarin, and antiplatelet agents including nonsteroidal anti-inflammatory drugs (Table 62.5).
•Heparin-induced thrombocytopenia (HIT) should be considered if the platelet count decreases by 50% and/or thrombosis occurs 5–14 days after starting heparin.
•History of known systemic conditions associated with defects in platelets like alcoholism, cirrhosis, HIV infection, systemic lupus erythematosus (SLE), and uremia.
•Family history of excessive bleeding.

62 Disseminated Intravascular Coagulation and Thrombocytopenia |
495 |
||||
|
|
|
|||
Table 62.5 Drugs associated with thrombocytopenia |
|
|
|||
Mechanism |
|
Drugs |
|
|
|
Drug-specific antibody |
|
H2 receptor blockers |
Ranitidine, cimetidine |
|
|
|
|
Gp IIb/IIIa inhibitors |
Abciximab |
|
|
Drug-dependent antibody |
Antibiotics |
Vancomycin, rifampicin, |
|
||
|
|
|
|
chloroquine, amphotericin B, |
|
|
|
|
|
sulfonamides |
|
|
|
Salicylates/NSAIDs |
Aspirin, diclofenac, ibuprofen |
|
|
|
|
Antiepileptics |
Valproate, carbamazepine, |
|
|
|
|
|
|
phenytoin |
|
|
|
Antiarrhythmics |
Amiodarone |
|
|
|
|
Miscellaneous |
Quinine, furosemide, thiazide, |
|
|
|
|
|
|
morphine |
|
Hapten-dependent antibody |
Antibiotic |
|
Penicillin, some cephalosporins |
|
|
Induction of autoantibodies |
Antiarrhythmics |
Procainamide |
|
||
|
|
Miscellaneous |
Gold salts |
|
|
Myelosuppression |
|
Antibiotics |
Linezolid |
|
|
|
|
Chemotherapeutic agents |
|
|
|
Unknown |
|
Antibiotics |
Fluconazole, daptomycin, |
|
|
|
|
|
|
ganciclovir, nitrofurantoin, |
|
|
|
|
|
piperacillin |
|
|
|
Miscellaneous |
Digoxin, haloperidol |
|
|
|
|
Gp IIb/IIIa inhibitors |
Eptifibatide |
|
|
Immune complex with PF4 |
Heparins |
|
Unfractionated and low-molecular- |
||
|
|
|
|
weight heparin |
|
Interference with folate |
Antibiotic |
|
Meropenem |
|
|
metabolism |
|
|
|
|
|
Thrombotic microangiopathy |
|
|
Clopidogrel, ticlopidine |
|
|
Preexisting antibodies |
|
|
|
Abciximab |
|
Table 62.6 Factors associated with platelet refractoriness |
|
|
|||
Nonimmune factors |
Clinical factors |
Splenomegaly, fever, infection, bleeding, dissemi- |
|
||
|
|
|
nated intravascular coagulation |
|
|
|
Drugs |
|
Amphotericin B, vancomycin, ciprofloxacin, heparin |
||
|
Patient factors |
previous pregnancies, previous transfusions |
|
||
Immune factors |
Antibodies |
HLA, platelet specific, erythrocyte |
|
||
|
Others |
|
Length of time the platelets are stored |
|
|
•Perform physical examination to look for:
–Evidence of bleeding in skin, mucous membrane, joints, soft tissue
–Lymphadenopathy
–Splenomegaly
Step 4: Transfuse platelets (Table 62.6)
•Three types of platelet products are commonly used in clinical practice:
– Random-donor platelets (RDP)
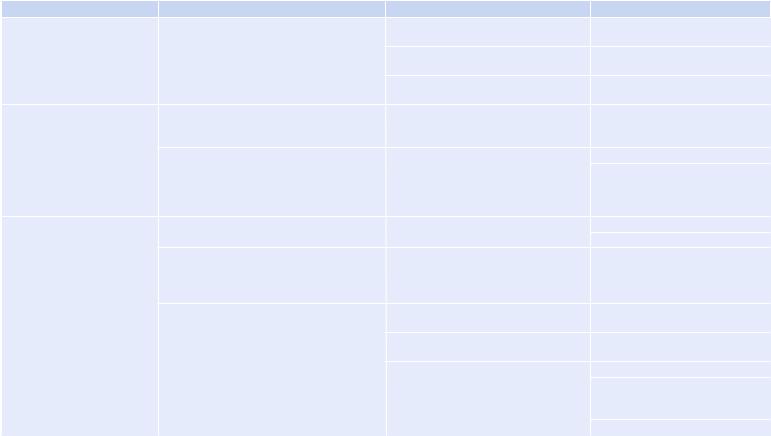
Table 62.7 Approach for management of thrombocytopenia |
|
|
|
Etiology |
Mechanism |
Presentation |
Treatment |
ITP, after viral illness, may be |
IgG antibodies against platelet antigens, platelet |
All ages, common in young adult females |
Steroids, prednisolone 1 mg/kg/day |
associated with antiphospho |
clearance by spleen, inadequate platelet |
|
for 1–2 weeks, taper |
lipid antibody syndrome, may |
production response |
Severe thrombocytopenia with normal |
IVIG infusion 1g/kg/day for 2 days |
be initial presentation of |
|
RBC and WBC morphology and number |
|
connective tissue disease, |
|
Diagnosis by exclusion |
Anti RhD antibodies 50–75 m/kg IV |
lymphoploriferative malignancy |
|
||
|
|
(Rh + Ve patients with intact spleen) |
|
|
|
|
|
TTP-HUS |
-Inherited or acquired deficiency of von |
Microangiopathic hemolytic anemia, |
FFP transfusions until the patient is |
|
Willebrand factor cleaving protease |
thrombocytopenia, renal insufficiency, |
ready for plasma exchanges |
|
(ADAMTS13) |
fever, and mental status changes |
|
|
Idiopathic or secondary to Escherichia coli |
Schistiocytes in peripheral smear, raised |
Plasma exchanges |
|
diarrhea, HIV infection, certain drugs |
LDH, normal coagulation profile |
Platelet transfusions only in |
|
(ticlopidine, clopidogrel, quinine, cyclosporine |
|
life-threatening bleeding |
|
A, mitomycin A, cisplatin, etc.), pregnancy, bone |
|
|
|
marrow transplant, and metastatic carcinomas |
|
|
Drug-induced thrombocytopenia |
Antiplatelet agents’ and other drugs’ immune |
History—no other blood or coagulation |
Stop the offending drug |
|
mechanism |
abnormalities |
Supportive care |
|
Chemotherapy and alcohol—directly inhibit |
Most chemotherapeutic drugs—nadir of |
Supportive care |
|
megakaryocytes |
blood counts in 7–10 days, recovers over |
|
|
|
2–3 weeks Nitrosureas and mitomycin |
|
|
|
cause prolonged myelosuppression |
|
|
Heparin—antibodies against heparin–platelet |
Type I—modest transient thrombocytopenia |
Spontaneous recovery |
|
factor 4 complex |
in 2–3 days after heparin therapy |
|
|
|
Type II—less common, occurs 4–14 days |
Stop heparin |
|
|
after heparin therapy |
|
|
|
ELISA assay for anti-PF 4 antibody, |
Doppler to rule out thrombosis |
|
|
serotonin release assay, platelet aggregation |
Use direct thrombin inhibitors |
|
|
studies |
(argatroban, lepirudin) Fondaparinux |
|
|
|
should be used with caution |
|
|
|
LMWH and UFH should not be used |
