
- •Preface to the 3rd edition
- •General Pharmacology
- •Systems Pharmacology
- •Therapy of Selected Diseases
- •Subject Index
- •Abbreviations
- •General Pharmacology
- •History of Pharmacology
- •Drug and Active Principle
- •The Aims of Isolating Active Principles
- •European Plants as Sources of Effective Medicines
- •Drug Development
- •Congeneric Drugs and Name Diversity
- •Oral Dosage Forms
- •Drug Administration by Inhalation
- •Dermatological Agents
- •From Application to Distribution in the Body
- •Potential Targets of Drug Action
- •External Barriers of the Body
- •Blood–Tissue Barriers
- •Membrane Permeation
- •Binding to Plasma Proteins
- •The Liver as an Excretory Organ
- •Biotransformation of Drugs
- •Drug Metabolism by Cytochrome P450
- •The Kidney as an Excretory Organ
- •Presystemic Elimination
- •Drug Concentration in the Body as a Function of Time—First Order (Exponential) Rate Processes
- •Time Course of Drug Concentration in Plasma
- •Time Course of Drug Plasma Levels during Repeated Dosing (A)
- •Time Course of Drug Plasma Levels during Irregular Intake (B)
- •Accumulation: Dose, Dose Interval, and Plasma Level Fluctuation (A)
- •Dose–Response Relationship
- •Concentration–Effect Curves (B)
- •Concentration–Binding Curves
- •Types of Binding Forces
- •Agonists—Antagonists
- •Other Forms of Antagonism
- •Enantioselectivity of Drug Action
- •Receptor Types
- •Undesirable Drug Effects, Side Effects
- •Drug Allergy
- •Cutaneous Reactions
- •Drug Toxicity in Pregnancy and Lactation
- •Pharmacogenetics
- •Placebo (A)
- •Systems Pharmacology
- •Sympathetic Nervous System
- •Structure of the Sympathetic Nervous System
- •Adrenergic Synapse
- •Adrenoceptor Subtypes and Catecholamine Actions
- •Smooth Muscle Effects
- •Cardiostimulation
- •Metabolic Effects
- •Structure–Activity Relationships of Sympathomimetics
- •Indirect Sympathomimetics
- •Types of
- •Antiadrenergics
- •Parasympathetic Nervous System
- •Cholinergic Synapse
- •Parasympathomimetics
- •Parasympatholytics
- •Actions of Nicotine
- •Localization of Nicotinic ACh Receptors
- •Effects of Nicotine on Body Function
- •Aids for Smoking Cessation
- •Consequences of Tobacco Smoking
- •Dopamine
- •Histamine Effects and Their Pharmacological Properties
- •Serotonin
- •Vasodilators—Overview
- •Organic Nitrates
- •Calcium Antagonists
- •ACE Inhibitors
- •Drugs Used to Influence Smooth Muscle Organs
- •Cardiac Drugs
- •Cardiac Glycosides
- •Antiarrhythmic Drugs
- •Drugs for the Treatment of Anemias
- •Iron Compounds
- •Prophylaxis and Therapy of Thromboses
- •Possibilities for Interference (B)
- •Heparin (A)
- •Hirudin and Derivatives (B)
- •Fibrinolytics
- •Intra-arterial Thrombus Formation (A)
- •Formation, Activation, and Aggregation of Platelets (B)
- •Inhibitors of Platelet Aggregation (A)
- •Presystemic Effect of ASA
- •Plasma Volume Expanders
- •Lipid-lowering Agents
- •Diuretics—An Overview
- •NaCl Reabsorption in the Kidney (A)
- •Aquaporins (AQP)
- •Osmotic Diuretics (B)
- •Diuretics of the Sulfonamide Type
- •Potassium-sparing Diuretics (A)
- •Vasopressin and Derivatives (B)
- •Drugs for Gastric and Duodenal Ulcers
- •Laxatives
- •Antidiarrheal Agents
- •Drugs Affecting Motor Function
- •Muscle Relaxants
- •Nondepolarizing Muscle Relaxants
- •Depolarizing Muscle Relaxants
- •Antiparkinsonian Drugs
- •Antiepileptics
- •Pain Mechanisms and Pathways
- •Eicosanoids
- •Antipyretic Analgesics
- •Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
- •Cyclooxygenase (COX) Inhibitors
- •Local Anesthetics
- •Opioid Analgesics—Morphine Type
- •General Anesthesia and General Anesthetic Drugs
- •Inhalational Anesthetics
- •Injectable Anesthetics
- •Sedatives, Hypnotics
- •Benzodiazepines
- •Pharmacokinetics of Benzodiazepines
- •Therapy of Depressive Illness
- •Mania
- •Therapy of Schizophrenia
- •Psychotomimetics (Psychedelics, Hallucinogens)
- •Hypothalamic and Hypophyseal Hormones
- •Thyroid Hormone Therapy
- •Glucocorticoid Therapy
- •Follicular Growth and Ovulation, Estrogen and Progestin Production
- •Oral Contraceptives
- •Antiestrogen and Antiprogestin Active Principles
- •Aromatase Inhibitors
- •Insulin Formulations
- •Treatment of Insulin-dependent Diabetes Mellitus
- •Treatment of Maturity-Onset (Type II) Diabetes Mellitus
- •Oral Antidiabetics
- •Drugs for Maintaining Calcium Homeostasis
- •Drugs for Treating Bacterial Infections
- •Inhibitors of Cell Wall Synthesis
- •Inhibitors of Tetrahydrofolate Synthesis
- •Inhibitors of DNA Function
- •Inhibitors of Protein Synthesis
- •Drugs for Treating Mycobacterial Infections
- •Drugs Used in the Treatment of Fungal Infections
- •Chemotherapy of Viral Infections
- •Drugs for the Treatment of AIDS
- •Drugs for Treating Endoparasitic and Ectoparasitic Infestations
- •Antimalarials
- •Other Tropical Diseases
- •Chemotherapy of Malignant Tumors
- •Targeting of Antineoplastic Drug Action (A)
- •Mechanisms of Resistance to Cytostatics (B)
- •Inhibition of Immune Responses
- •Antidotes and Treatment of Poisonings
- •Therapy of Selected Diseases
- •Hypertension
- •Angina Pectoris
- •Antianginal Drugs
- •Acute Coronary Syndrome— Myocardial Infarction
- •Congestive Heart Failure
- •Hypotension
- •Gout
- •Obesity—Sequelae and Therapeutic Approaches
- •Osteoporosis
- •Rheumatoid Arthritis
- •Migraine
- •Common Cold
- •Atopy and Antiallergic Therapy
- •Bronchial Asthma
- •Emesis
- •Alcohol Abuse
- •Local Treatment of Glaucoma
- •Further Reading
- •Further Reading
- •Picture Credits
- •Drug Indexes

298 Anticancer Drugs
Chemotherapy of Malignant Tumors
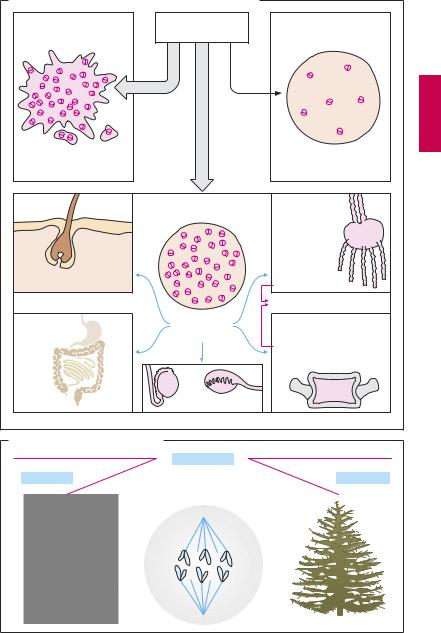
A tumor (neoplasm) consists of cells that proliferate independently of the body’s inherent “building plan.” A malignant tumor (cancer) is present when the tumor tissue destructively invades healthy surrounding tissue or when dislodged tumor cells form secondary tumors (metastases) in other organs. A cure requires the elimination of all malignant cells (curative therapy). When this is not possible, attempts can be made to slow tumor growth and thereby prolong the patient’s life or improve quality of life (palliative therapy). Chemotherapy is faced with the problem that the malignant cells are endogenous and almost lacking in specific metabolic properties.
Cytostatics (A) are cytotoxic substances that particularly affect proliferating or dividing (mitotic) cells. Rapidly dividing malignant cells are preferentially injured. Damage to mitotic processes not only retards tumor growth but also may initiate apoptosis (programmed cell death). Tissues with a low mitosis rate are largely unaffected; likewise, most healthy tissues. This, however, also applies to malignant tumors consisting of slowly dividing differentiated cells.
Tissues that have a physiologically high mitosis rate are bound to be affected by cytostatic therapy. Thus, typical adverse effects occur. Loss of hair results from injury to hair follicles; gastrointestinal disturbances, such as diarrhea, from inadequate replacement of enterocytes whose lifespan is limited to a few days; nausea and vomiting from stimulation of area postrema chemoreceptors (p.342); and lowered resistance to infection from weakening of the immune system (p.304). In addition, cytostatics cause bone marrow depression. Resupply of blood cells depends on the mitotic activity of bone marrow stem and daughter cells. When myeloid proliferation is arrested, the short-lived granulocytes are the first to be affected (neu-
tropenia), then blood platelets (thrombopenia) and, finally, the more long-lived erythrocytes (anemia). Infertility is caused by suppression of spermatogenesis or follicle maturation. Most cytostatics disrupt DNA metabolism. This entails the risk of a potential genomic alteration in healthy cells (mutagenic effect). Conceivably, the latter accounts for the occurrence of leukemias several years after cytostatic therapy (carcinogenic effect). Furthermore, congenital malformations are to be expected when cytostatics must be used during pregnancy (teratogenic effect).
Cytostatics possess different mechanisms of action.
Damage to the mitotic spindle (B). The contractile proteins of the spindle apparatus must draw apart the replicated chromosomes before the cell can divide. This process is prevented by the so-called spindle poisons (see also colchicine, p.326) that arrest mitosis at metaphase by disrupting the assembly into spindle threads of microtubuli. These consist of the proteins α-and β-tubulin. Surplus tubuli are broken down, enabling the tubulin subunits to be recycled.
The vinca alkaloids, vincristine and vinblastine (from the periwinkle plant, Vinca rosea), inhibit the polymerization of tubulin subunits into microtubuli. Damage to the nervous system is a predicted adverse effect arising from injury to microtubule-operated axonal transport mechanisms.
Paclitaxel, from the bark of the pacific yew (Taxus brevifolia), inhibits disassembly of microtubules and induces formation of atypical ones, and thus impedes the reassemblage of tubulins into properly functioning microtubules. Docetaxel is a semisynthetic derivative.
Chemotherapy of Malignant Tumors |
299 |
A. Chemotherapy of tumors: principal and adverse effects |
|
||
Malignant tissue |
Cytostatics inhibit |
Healthy tissue |
|
with numerous mitoses |
with few mitoses |
||
cell division |
|||
|
|
||
Wanted effect: |
|
Little effect |
|
inhibition of |
|
|
|
tumor growth |
|
|
|
|
Healthy tissue with |
Lymph node |
|
|
numerous mitoses |
|
|
|
|
Inhibition of |
|
|
|
lymphocyte |
|
Damage to hair follicle |
|
multiplication: |
|
|
immune |
||
Hair loss |
|
weakness |
|
|
|
Lowered resistance |
|
|
|
to infection |
|
|
Unwanted effects |
Bone marrow |
|
|
|
||
|
|
Inhibition of |
|
|
|
granulo-, |
|
|
|
thrombocyto-, |
|
Inhibition of |
|
and erythropoiesis |
|
ephithelial renewal |
|
|
|
Diarrhea |
Germinal cell damage |
|
|
B. Cytostatics: inhibition of mitosis |
|
|
Inhibition of |
Microtubules |
Inhibition of |
of mitotic spindle |
||
formation |
|
degradation |
Vinca alkaloids, |
|
Taxoids, |
e.g., vinblastine |
|
e.g., paclitaxel |
Vinca rosea |
|
Western yew tree |

300 Anticancer Drugs
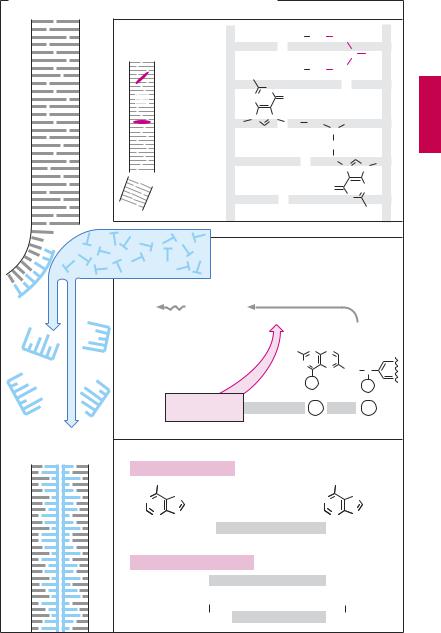
Inhibition of DNA and RNA synthesis (A).
Mitosis is preceded by replication of chromosomes (DNA synthesis) and increased protein synthesis (RNA synthesis). Existing DNA (gray) serves as a template for the synthesis of new (blue) DNA or RNA. De-novo synthesis may be inhibited by the following mechanisms.
Damage to the template (1). Alkylating cytostatics are reactive compounds that transfer alkyl residues into a covalent bond with DNA. For instance, mechlorethamine (nitrogen mustard) is able to cross-link doublestranded DNA on giving off its chlorine atoms. Correct reading of genetic information is thereby rendered impossible. Other alkylating agents are chlorambucil, melphalan, thio-TEPA, cyclophosphamide, ifosfamide, lomustine, and busulfan. Specific adverse reactions include irreversible pulmonary fibrosis due to busulfan and hemorrhagic cystitis caused by the cyclophosphamide metabolite acrolein (preventable by the uro-protectant mesna = sodium 2-mer- captoethanesulfonate). The platinum-con- taining compounds cisplatin and carboplatin release platinum, which binds to DNA.
Cystostatic antibiotics insert themselves into the DNA double strand; this may lead to strand breakage (e.g., with bleomycin). The anthracycline antibiotics daunorubicin and adriamycin (doxorubicin) may induce cardiomyopathy. Bleomycin can also cause pulmonary fibrosis.
Induction of strand breakage may result from inhibition of topoisomerase. The epipodophyllotoxins etoposide and tenoposide interact with topoisomerase II, which functions to split, transpose, and reseal DNA strands (p.276); these agents cause strand breakage by inhibiting resealing. The “tecans” topotecan and irinotecan are derivatives of camptothecin from the fruits of a Chinese tree (Camptotheca acuminata). They inhibit topoisomerase I, which induces breaks in single-strand DNA.
Inhibition of nucleobase synthesis (2). Tetrahydrofolic acid (THF) is required for the synthesis of both purine bases and thymidine. Formation of THF from folic acid involves dihydrofolate reductase (p. 274). The folate analogues aminopterin and methotrexate (amethopterin) inhibit enzyme activity. Cellular stores of THFare depleted. The effect of these antimetabolites can be reversed by administration of folinic acid (5-formyl-THF, leucovorin, citrovorum factor). Hydroxyurea (hydroxycarbamide) inhibits ribonucleotide reductase that normally converts ribonucleotides into deoxyribonucleotides subsequently used as DNA building blocks.
Incorporation of false building blocks (3).
Unnatural nucleobases (6-mercaptopurine; 5-fluorouracil) or nucleosides with incorrect sugars (cytarabine) act as antimetabolites. They inhibit DNA/RNA synthesis or lead to synthesis of missense nucleic acids.
6-Mercaptopurine results from biotransformation of the inactive precursor azathioprine (p.37). The uricostatic allopurinol (p.327) inhibits the degradation of 6-mer- captopurine such that coadministration of the two drugs requires dose reduction of the latter.
Combination therapy. Cytostatics are frequently administered in complex therapeutic regimens designed to improve ef cacy and tolerability of treatment.
Supportive therapy. Cancer chemotherapy can be supported by adjunctive medications. Thus, 5-HT3 serotonin receptor antagonists (e.g., ondansetron, p.342) afford effective protection against vomiting induced by highly emetogenic drugs such as cisplatin. Bone marrow depression can be counteracted by granulocyte and granulocyte/macrophage colony-stimulating factors (filgrastim and lenograstim and molgramostim, respectively).
Chemotherapy of Malignant Tumors |
301 |
A.Cytostatics: alkylating agents and cytostatic antibiotics (1), inhibitors of tetrahydrofolate synthesis (2), antimetabolites (3)
DNA |
Damage |
|
|
|
Cl |
CH2 |
CH2 |
|
|
|
to template |
|
|
|
|||||
|
|
|
|
|
|
|
|
||
|
|
Alkylation, |
|
|
|
|
|
N |
CH3 |
|
|
|
|
Cl |
CH2 |
C |
|
|
|
|
|
e.g., by |
H2N |
|
2 |
|
|||
|
|
mechlorethamine |
|
|
|
H |
|
|
|
|
|
|
NH |
Mechlorethamine |
|
||||
|
|
|
|
|
|
|
|
|
|
|
Pt |
Binding |
N |
|
O |
|
|
|
|
|
|
of platinum |
|
|
|
|
|
|
|
|
|
Insertion |
N |
N+ |
CH2 |
CH2 |
|
CH3 |
|
|
|
|
|
N |
|
||||
|
|
into DNA, e.g., |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
doxorubicin |
|
|
|
H2C |
|
|
|
|
|
|
|
|
|
|
|
||
|
|
Induction of |
|
|
|
H2C |
+ |
N |
|
|
|
strand breaks |
|
|
|
|
|
N |
|
|
|
Topoisomerase |
|
|
|
|
O |
|
N |
|
|
inhibitors: |
|
|
|
|
HN |
||
|
|
epipodophyllo- |
|
|
|
|
|
|
|
|
1 |
toxins, tecans |
|
|
|
|
|
|
NH2 |
|
|
|
|
|
|
|
|
||
|
|
|
Inhibition of nucleotide synthesis |
|
||||
|
Building blocks |
|
|
|
|
|
||
|
Purines |
|
Tetrahydro- |
Dihydrofolate |
|
|
|
|
|
|
|
|
|
|
|||
|
Thymine |
|
folate |
reductase |
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
nucleotide |
|
|
|
|
Folic acid |
||
|
|
|
|
H2N |
N |
N |
|
|
RNA |
|
Inhibition by |
|
N |
N |
|
|
|
|
|
|
CH2 |
N |
||||
|
|
|
|
|||||
|
|
|
|
|
OH |
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
Methotrexate |
|
NH2 |
|
CH3 |
||
|
2 |
|
|
|
|
|
|
|
DNA |
DNA |
|
Insertion of incorrect building blocks |
|||||
|
Purine antimetabolite |
|
|
|
|
|
||
|
SH |
H |
|
|
|
NH2 |
|
|
|
|
|
|
|
|
H |
|
|
|
N |
N |
|
|
N |
N |
|
|
|
|
|
|
|
|
|||
|
N |
N |
|
|
|
N |
N |
|
|
6-Mercaptopurine |
instead of |
Adenine |
|
||||
|
from azathioprine |
|
|
|
|
|
||
|
Pyrimidine antimetabolite |
|
|
|
|
|
||
|
5-Fluorouracil |
instead of |
Uracil |
|
||||
|
Cytarabine |
Cytosine |
|
|
Cytosine |
|
||
|
3 |
|
Arabinose |
instead of |
Desoxyribose |
|||
|
|
|
|
|
|
|
|
|
