
- •Preface to the 3rd edition
- •General Pharmacology
- •Systems Pharmacology
- •Therapy of Selected Diseases
- •Subject Index
- •Abbreviations
- •General Pharmacology
- •History of Pharmacology
- •Drug and Active Principle
- •The Aims of Isolating Active Principles
- •European Plants as Sources of Effective Medicines
- •Drug Development
- •Congeneric Drugs and Name Diversity
- •Oral Dosage Forms
- •Drug Administration by Inhalation
- •Dermatological Agents
- •From Application to Distribution in the Body
- •Potential Targets of Drug Action
- •External Barriers of the Body
- •Blood–Tissue Barriers
- •Membrane Permeation
- •Binding to Plasma Proteins
- •The Liver as an Excretory Organ
- •Biotransformation of Drugs
- •Drug Metabolism by Cytochrome P450
- •The Kidney as an Excretory Organ
- •Presystemic Elimination
- •Drug Concentration in the Body as a Function of Time—First Order (Exponential) Rate Processes
- •Time Course of Drug Concentration in Plasma
- •Time Course of Drug Plasma Levels during Repeated Dosing (A)
- •Time Course of Drug Plasma Levels during Irregular Intake (B)
- •Accumulation: Dose, Dose Interval, and Plasma Level Fluctuation (A)
- •Dose–Response Relationship
- •Concentration–Effect Curves (B)
- •Concentration–Binding Curves
- •Types of Binding Forces
- •Agonists—Antagonists
- •Other Forms of Antagonism
- •Enantioselectivity of Drug Action
- •Receptor Types
- •Undesirable Drug Effects, Side Effects
- •Drug Allergy
- •Cutaneous Reactions
- •Drug Toxicity in Pregnancy and Lactation
- •Pharmacogenetics
- •Placebo (A)
- •Systems Pharmacology
- •Sympathetic Nervous System
- •Structure of the Sympathetic Nervous System
- •Adrenergic Synapse
- •Adrenoceptor Subtypes and Catecholamine Actions
- •Smooth Muscle Effects
- •Cardiostimulation
- •Metabolic Effects
- •Structure–Activity Relationships of Sympathomimetics
- •Indirect Sympathomimetics
- •Types of
- •Antiadrenergics
- •Parasympathetic Nervous System
- •Cholinergic Synapse
- •Parasympathomimetics
- •Parasympatholytics
- •Actions of Nicotine
- •Localization of Nicotinic ACh Receptors
- •Effects of Nicotine on Body Function
- •Aids for Smoking Cessation
- •Consequences of Tobacco Smoking
- •Dopamine
- •Histamine Effects and Their Pharmacological Properties
- •Serotonin
- •Vasodilators—Overview
- •Organic Nitrates
- •Calcium Antagonists
- •ACE Inhibitors
- •Drugs Used to Influence Smooth Muscle Organs
- •Cardiac Drugs
- •Cardiac Glycosides
- •Antiarrhythmic Drugs
- •Drugs for the Treatment of Anemias
- •Iron Compounds
- •Prophylaxis and Therapy of Thromboses
- •Possibilities for Interference (B)
- •Heparin (A)
- •Hirudin and Derivatives (B)
- •Fibrinolytics
- •Intra-arterial Thrombus Formation (A)
- •Formation, Activation, and Aggregation of Platelets (B)
- •Inhibitors of Platelet Aggregation (A)
- •Presystemic Effect of ASA
- •Plasma Volume Expanders
- •Lipid-lowering Agents
- •Diuretics—An Overview
- •NaCl Reabsorption in the Kidney (A)
- •Aquaporins (AQP)
- •Osmotic Diuretics (B)
- •Diuretics of the Sulfonamide Type
- •Potassium-sparing Diuretics (A)
- •Vasopressin and Derivatives (B)
- •Drugs for Gastric and Duodenal Ulcers
- •Laxatives
- •Antidiarrheal Agents
- •Drugs Affecting Motor Function
- •Muscle Relaxants
- •Nondepolarizing Muscle Relaxants
- •Depolarizing Muscle Relaxants
- •Antiparkinsonian Drugs
- •Antiepileptics
- •Pain Mechanisms and Pathways
- •Eicosanoids
- •Antipyretic Analgesics
- •Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
- •Cyclooxygenase (COX) Inhibitors
- •Local Anesthetics
- •Opioid Analgesics—Morphine Type
- •General Anesthesia and General Anesthetic Drugs
- •Inhalational Anesthetics
- •Injectable Anesthetics
- •Sedatives, Hypnotics
- •Benzodiazepines
- •Pharmacokinetics of Benzodiazepines
- •Therapy of Depressive Illness
- •Mania
- •Therapy of Schizophrenia
- •Psychotomimetics (Psychedelics, Hallucinogens)
- •Hypothalamic and Hypophyseal Hormones
- •Thyroid Hormone Therapy
- •Glucocorticoid Therapy
- •Follicular Growth and Ovulation, Estrogen and Progestin Production
- •Oral Contraceptives
- •Antiestrogen and Antiprogestin Active Principles
- •Aromatase Inhibitors
- •Insulin Formulations
- •Treatment of Insulin-dependent Diabetes Mellitus
- •Treatment of Maturity-Onset (Type II) Diabetes Mellitus
- •Oral Antidiabetics
- •Drugs for Maintaining Calcium Homeostasis
- •Drugs for Treating Bacterial Infections
- •Inhibitors of Cell Wall Synthesis
- •Inhibitors of Tetrahydrofolate Synthesis
- •Inhibitors of DNA Function
- •Inhibitors of Protein Synthesis
- •Drugs for Treating Mycobacterial Infections
- •Drugs Used in the Treatment of Fungal Infections
- •Chemotherapy of Viral Infections
- •Drugs for the Treatment of AIDS
- •Drugs for Treating Endoparasitic and Ectoparasitic Infestations
- •Antimalarials
- •Other Tropical Diseases
- •Chemotherapy of Malignant Tumors
- •Targeting of Antineoplastic Drug Action (A)
- •Mechanisms of Resistance to Cytostatics (B)
- •Inhibition of Immune Responses
- •Antidotes and Treatment of Poisonings
- •Therapy of Selected Diseases
- •Hypertension
- •Angina Pectoris
- •Antianginal Drugs
- •Acute Coronary Syndrome— Myocardial Infarction
- •Congestive Heart Failure
- •Hypotension
- •Gout
- •Obesity—Sequelae and Therapeutic Approaches
- •Osteoporosis
- •Rheumatoid Arthritis
- •Migraine
- •Common Cold
- •Atopy and Antiallergic Therapy
- •Bronchial Asthma
- •Emesis
- •Alcohol Abuse
- •Local Treatment of Glaucoma
- •Further Reading
- •Further Reading
- •Picture Credits
- •Drug Indexes

46 Pharmacokinetics
Time Course of Drug Concentration in Plasma
(A). Drugs are taken up into and eliminated from the body by various routes. The body thus represents an open system wherein the actual drug concentration reflects the interplay of intake (ingestion) and egress (elimination). When orally administered drug is absorbed from the stomach and intestine, speed of uptake depends on many factors, including the speed of drug dissolution (in the case of solid dosage forms) and of gastrointestinal transit; the membrane penetrability of the drug; its concentration gradient across the mucosa–blood barrier; and mucosal blood flow. Absorption from the intestine causes the drug concentration in blood to increase. Transport in blood conveys the drug to different organs (distribution), into which it is taken up to a degree compatible with its chemical properties and rate of blood flow through the organ. For instance, well-perfused organs such as the brain receive a greater proportion than do less wellperfused ones. Uptake into tissue causes the blood concentration to fall. Absorption from the gut diminishes as the mucosa–blood gradient decreases. Plasma concentration reaches a peak when the amount of drug leaving the blood per unit of time equals that being absorbed.
Drug entry into hepatic and renal tissue constitutes movement into the organs of elimination. The characteristic phasic time course of drug concentration in plasma represents the sum of the constituent processes of absorption, distribution, and elimination, which overlap in time. When distribution takes place significantly faster than elimination, there is an initial rapid and then a greatly retarded fall in the plasma level, the former being designated the α-phase (distribution phase), the latter the β-phase (elimination phase). When the drug is distributed faster than it is absorbed, the time course of the plasma level can be described in mathematically simplified form by the Bateman
function (k1 and k2 represent the rate constants for absorption and elimination, respectively).
(B). The velocity of absorption depends on the route of administration. The more rapid the absorption, the shorter will be the time (tmax) required to reach the peak plasma level (cmax), the higher will be the cmax, and the earlier will the plasma level begin to fall again.
The area under the plasma level–time curve
(AUC) is independent of the route of administration, provided the doses and bioavailability are the same (law of corresponding areas). The AUC can thus be used to determine the bioavailability F of a drug. The ratio of AUC values determined after oral and intravenous administrations of a given dose of a particular drug corresponds to the proportion of drug entering the systemic circulation after oral administration. Thus,
F = |
AUCoral administration |
AUCiv administration |
The determination of plasma levels affords a comparison of different proprietary preparations containing the same drug in the same dosage. Identical plasma level–time curves of different manufacturers’ products with reference to a standard preparation indicate bioequivalence with the standard of the preparation under investigation.

Drug Concentration in Plasma |
47 |
A. Time course of drug concentration |
|
|
|
|
|
|
|
|
|||||
Absorption |
|
|
|
Distribution |
|
Elimination |
|||||||
Uptake from |
|
|
into body |
|
|
|
from body by |
||||||
stomach and |
|
|
tissues: |
|
|
|
|
|
|
biotransformation |
|||
intestines |
|
|
|
|
α -phase |
|
|
|
|
|
(chemical alteration) |
||
into blood |
|
|
|
|
|
|
|
|
|
|
|
excretion via kidney: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
β -phase |
in blood (c) |
|
|
|
|
|
|
|
|
|
|
|
|
|
concentration |
Bateman function |
|
|
|
|
|
|
|
|
|
|||
|
c = |
Dose |
k1 |
x (e |
-k |
1 |
t |
-e |
-k |
2 |
t |
) |
|
Drug |
|
Vapp |
x |
|
|
|
|
||||||
|
|
k2 |
- k1 |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
Time (t) |
B. Mode of application and time course of drug concentration |
|
|
(c) |
|
|
in blood |
Intravenous |
|
Intramuscular |
|
|
Drug concentration |
|
|
Subcutaneous |
|
|
|
Oral |
|
|
Time (t) |
|
|
|
|

48 Pharmacokinetics
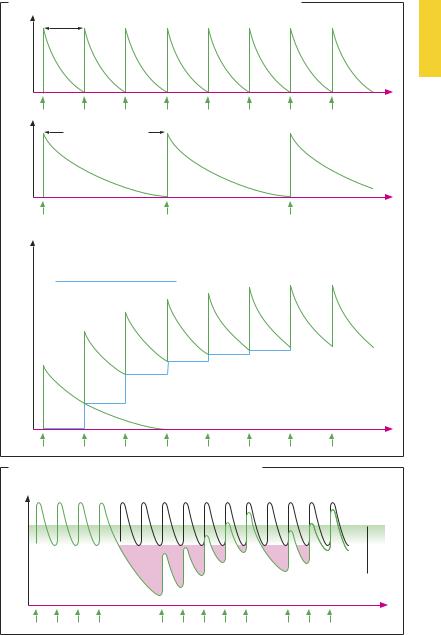
Time Course of Drug Plasma Levels during Repeated Dosing (A)
When a drug is administered at regular intervals over a prolonged period, the rise and fall of drug concentration in blood will be determined by the relationship between the half-life of elimination and the time interval between doses. If the amount of drug administered in each dose has been eliminated before the next dose is applied, repeated intake at constant intervals will result in similar plasma levels. If intake occurs before the preceding dose is eliminated completely, the next dose will add to the residual amount still present in the body—i.e., the drug accumulates. The shorter the dosing interval relative to the elimination half-life, the larger will be the residual amount of drug to which the next dose is added and the more extensively will the drug accumulate in the body. However, at a given dosing frequency, the drug does not accumulate infinitely and a steady state (concentration
Css) or accumulation equilibrium is eventually reached. This is so because the activity of elimination processes is concentrationdependent. The higher the drug concentration rises, the greater is the amount eliminated per unit time. After several doses, the concentration will have climbed to a level at which the amounts eliminated and taken in per unit of time become equal, i.e., a steadystate is reached. Within this concentration range, the plasma level will continue to rise (peak) and fall (trough) as dosing is continued at regular intervals. The height of the steady state (Css) depends upon the amount (D) administered per dosing interval (τ) and the clearance (Cl):
Css = τ DCl
The speed at which the steady state is reached corresponds to the speed of elimination of the drug. The time needed to reach
90% of the concentration plateau is about 3 times the t½ of elimination.
Time Course of Drug Plasma Levels during Irregular Intake (B)
In practice, it proves dif cult to achieve a plasma level that undulates evenly around the desired effective concentration. For instance, if two successive doses are omitted (“?” in B), the plasma level will drop below the therapeutic range and a longer period will berequiredto regain the desired plasma level. In everyday life, patients will be apt to neglect to take drugs at the scheduled time. Patient compliance means strict adherence to the prescribed regimen. Apart from poor compliance, the same problem may occur when the total daily dose is divided into three individual doses (t.i.d.) and the first dose is taken at breakfast, the second at lunch, and the third at supper. Under these conditions,thenocturnaldosingintervalwill betwicethe diurnalone. Consequently,plasma levels during the early morning hours may have fallen far below the desired, or possibly urgently needed, range.
Plasma Levels during Repeated/Irregular Dosing |
49 |
A. Time course of drug concentration in blood during regular intake |
||
|
Dosing interval |
|
concentration |
|
Time (t) |
|
|
|
Drug |
Dosing interval |
|
|
|
|
|
|
Time (t) |
|
Accumulation: |
Steady state: |
|
administered drug is |
drug intake equals |
|
not completely eliminated |
elimination during |
|
during interval |
dosing interval |
Drug concentration |
|
|
|
|
Time (t) |
B. Time course of drug concentration with irregular intake |
|
|
|
concentration |
|
|
Desired |
Drug |
|
|
|
|
|
therapeutic |
|
|
|
level |
|
|
|
|
|
? |
? |
? |
Time (t) |
