
- •VOLUME 3
- •CONTRIBUTOR LIST
- •PREFACE
- •LIST OF ARTICLES
- •ABBREVIATIONS AND ACRONYMS
- •CONVERSION FACTORS AND UNIT SYMBOLS
- •EDUCATION, COMPUTERS IN.
- •ELECTROANALGESIA, SYSTEMIC
- •ELECTROCARDIOGRAPHY, COMPUTERS IN
- •ELECTROCONVULSIVE THERAPHY
- •ELECTRODES.
- •ELECTROENCEPHALOGRAPHY
- •ELECTROGASTROGRAM
- •ELECTROMAGNETIC FLOWMETER.
- •ELECTROMYOGRAPHY
- •ELECTRON MICROSCOPY.
- •ELECTRONEUROGRAPHY
- •ELECTROPHORESIS
- •ELECTROPHYSIOLOGY
- •ELECTRORETINOGRAPHY
- •ELECTROSHOCK THERAPY.
- •ELECTROSTIMULATION OF SPINAL CORD.
- •ELECTROSURGICAL UNIT (ESU)
- •EMERGENCY MEDICAL CARE.
- •ENDOSCOPES
- •ENGINEERED TISSUE
- •ENVIRONMENTAL CONTROL
- •EQUIPMENT ACQUISITION
- •EQUIPMENT MAINTENANCE, BIOMEDICAL
- •ERGONOMICS.
- •ESOPHAGEAL MANOMETRY
- •EVENT-RELATED POTENTIALS.
- •EVOKED POTENTIALS
- •EXERCISE FITNESS, BIOMECHANICS OF.
- •EXERCISE, THERAPEUTIC.
- •EXERCISE STRESS TESTING
- •EYE MOVEMENT, MEASUREMENT TECHNIQUES FOR
- •FETAL MONITORING
- •FETAL SURGERY.
- •FEVER THERAPY.
- •FIBER OPTICS IN MEDICINE
- •FICK TECHNIQUE.
- •FITNESS TECHNOLOGY.
- •FIXATION OF ORTHOPEDIC PROSTHESES.
- •FLAME ATOMIC EMISSON SPECTROMETRY AND ATOMIC ABSORPTION SPECTROMETRY
- •FLAME PHOTOMETRY.
- •FLOWMETERS
- •FLOWMETERS, RESPIRATORY.
- •FLUORESCENCE MEASUREMENTS
- •FLUORESCENCE MICROSCOPY.
- •FLUORESCENCE SPECTROSCOPY.
- •FLUORIMETRY.
- •FRACTURE, ELECTRICAL TREATMENT OF.
- •FUNCTIONAL ELECTRICAL STIMULATION
- •GAMMA CAMERA.
- •GAMMA KNIFE
- •GAS AND VACUUM SYSTEMS, CENTRALLY PIPED MEDICAL
- •GAS EXCHANGE.
- •GASTROINTESTINAL HEMORRHAGE
- •GEL FILTRATION CHROMATOGRAPHY.
- •GLUCOSE SENSORS
- •HBO THERAPY.
- •HEARING IMPAIRMENT.
- •HEART RATE, FETAL, MONITORING OF.
- •HEART VALVE PROSTHESES
- •HEART VALVE PROSTHESES, IN VITRO FLOW DYNAMICS OF
- •HEART VALVES, PROSTHETIC
- •HEART VIBRATION.
- •HEART, ARTIFICIAL
- •HEART–LUNG MACHINES
- •HEAT AND COLD, THERAPEUTIC
- •HEAVY ION RADIOTHERAPY.
- •HEMODYNAMICS
- •HEMODYNAMIC MONITORING.
- •HIGH FREQUENCY VENTILATION
- •HIP JOINTS, ARTIFICIAL
- •HIP REPLACEMENT, TOTAL.
- •HOLTER MONITORING.
- •HOME HEALTH CARE DEVICES
- •HOSPITAL SAFETY PROGRAM.
- •HUMAN FACTORS IN MEDICAL DEVICES
- •HUMAN SPINE, BIOMECHANICS OF
Part II: the aging heart in health: links to heart disease. Circulation 2003;107:346–354.
118.Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a ‘‘set up’’ for vascular disease. Circulation 2003;107:139–146.
119.O’Rourke M. Arterial stiffness, systolic blood pressure, and logical treatment of arterial hypertension. Hypertension 1990;15:339–347.
120.Stergiopulos N, Westerhof N. Role of total arterial compliance and peripheral resistance in the determination of systolic and diastolic aortic pressure. Pathol Biol (Paris) 1999;47:641–647.
121.Stergiopulos N, Westerhof N. Determinants of pulse pressure. Hypertension 1998;32:556–559.
122.Sunagawa K, Maughan WL, Burkhoff D, Sagawa K. Left ventricular interaction with arterial load studied in isolated canine ventricle. Am J Physiol 1983;245:H773–H780.
123.Sunagawa K, Maughan WL, Sagawa K. Optimal arterial resistance for the maximal stroke work studied in isolate canine left ventricle. Circ Res 1985;56:586–595.
124.Burkhoff D, Sagawa K. Ventricular efficiency predicted by an analytical model. Am J Physiol 1986;250:R1021–R1027.
125.Kelly R, Ting C, Yang T, Liu C, Lowell W, Chang M, Kass D. Effective arterial elastance as index of arterial vascular load in humans. Circulation 1992;86:513–521.
126.Segers P, Stergiopulos N, Westerhof N. Relation of effective arterial elastance to arterial system properties. Am J Physiol Heart Circ Physiol 2002;282:H1041–H1046.
127.De Tombe PP, Jones S, Burkhoff D, Hunter WC, Kass DA. Ventricular stroke work and efficiency both remain nearly optimal despite altered vascular loading. Am J Physiol 1993;264:H1817–H1824.
128.Asanoi H, Sasayama S, Kameyama T. Ventriculoarterial coupling in normal and failing heart in humans. Circ Res 1989;65:483–493.
129.Sasayama S, Asanoi H. Coupling between the heart and arterial system in heart failure. Am J Med 1991;90: 14S–18S.
130.Stergiopulos N, Meister JJ, Westerhof N. Determinants of stroke volume and systolic and diastolic pressure. Am J Physiol 1996;270:H2050–H2059.
131.Segers P, Steendijk P, Stergiopulos N, Westerhof N. Predicting systolic and diastolic aortic blood pressure and stroke volume in the intact sheep. J Biomech 2001;34:41–50.
See also BIOIMPEDANCE IN CARDIOVASCULAR MEDICINE; BLOOD PRESSURE MEASUREMENT; FLOWMETERS, ELECTROMAGNETIC; MONITORING, HEMODYNAMIC.
HEMODYNAMIC MONITORING. See MONITORING,
HEMODYNAMIC.
HIGH FREQUENCY VENTILATION
J. BERT BUNNELL
Bunnell Inc.
Salt Lake City, Utah
INTRODUCTION
High frequency ventilators (HFVs) were designed to eliminate many of the problems that conventional ventilators
HIGH FREQUENCY VENTILATION |
497 |
create as they try to mimic normal breathing. When we breathe normally, we draw gas into the lungs by creating a negative pressure with our diaphragm. Iron lungs were created to replicate that activity, and they worked very well for thousands of polio patients. However, when patients are sealed off in airtight iron lungs numerous practical problems unrelated to breathing arise.
Positive pressure ventilators made assisting ventilation much easier. Attaching the ventilator to the patient’s lungs via an endotracheal (ET) tube greatly simplified patient care. But, lungs, especially premature lungs, are not designed to tolerate much positive pressure.
The lungs of prematurely borne infants have yet to be fully formed, and they lack the surfactant that enables alveoli to expand with very little pressure gradient. Hence, a considerable pressure gradient must be applied to ventilate them. Applying that pressure from the outside in, as conventional ventilators (CVs) have been doing since the early 1970s, causes problems. Tiny infant’s airways get distended, alveoli are ruptured, and inflammatory sensors are triggered. Even if an infant receives artificial surfactant to lessen the need for assisted ventilation, and they grow new lung fast enough to survive, they may well develop chronic lung disease at a time when most of us were just taking our first breaths. Most premature infants outgrow their chronic lung disease, but many struggle mightily with every virus they encounter in their first few years of life, and they have an increased incidence of neurodevelopmental problems, such as cerebral palsy. Some infants lose those struggles and die of pneumonia.
Acute respiratory distress syndrome (ARDS) is the primary problem for mechanical ventilation of adults. This disease affects 50 people per 100,000 with a mortality of 30–50%, and there have been few improvements in this mortality rate over the past several decades.
High frequency ventilators were developed in response to problems associated with CVs, but HFVs do not try to replicate normal breathing. They assist ventilation using much smaller tidal volumes delivered at rates 10 times higher than normal. Animal and clinical studies indicate that smaller tidal volumes cause less lung injury (1,2).
Swedish anesthesiologists in the 1970s turned up the rate of their anesthesia ventilators to enable them to use smaller breaths to assist patients during neurosurgery (3). Their regular ventilators caused pulsations in blood pressure, causing brain movement every time the ventilator pushed in a breath, which was an obvious problem during microsurgery.
Auto accident victims whose heads went through car windshields also pose problems during surgery when access to the lungs has to pass right through the area of the face and neck where major reconstruction is required. So, another anesthesiologist, Dr. Miroslav Klain, began sticking needles into patients’ necks to gain access to their tracheas, and he made it work by delivering very tiny breaths at very rapid rates (4).
The HFVs have shown great promise in supporting premature infants where fragile, underdeveloped and surfactant deficient lungs need to be gently ventilated until growth and maturation allow the newborn to catch up both anatomically and physiologically. In newborn infants,

498 HIGH FREQUENCY VENTILATION
where various modes of positive pressure ventilation have produced lung injury, HFVs have been widely accepted. Early studies using HFV to treat adults with severe ARDS have also shown promise in lessening lung injury and improving survival (5,6).
This article will review our current understanding of how HFVs work, the types of HFV equipment that are available for treating infants and adults, the results of several key animal and clinical studies that indicate how to optimize applications of HFVs, and what controversies remain to be resolved before HFVs can be considered as a primary mode of ventilation.
THEORETICAL BASIS FOR HFV: HOW HFVs WORK
High frequency ventilators are different from all other types of mechanical ventilators. They do not mimic normal breathing; rather, they facilitate gas exchange in a manner similar to panting in animals.
There are two elements to explaining how HFVs work with the higher than normal frequencies and smaller than normal tidal volumes. We begin with the assumption that ventilation or CO2 elimination is proportional to minute volume or frequency times tidal volume, as
|
|
ð1Þ |
VCO2 / Vmin ¼ f VT |
||
|
|
|
where VCO2 ¼ rate of carbon dioxide elimination; Vmin ¼ minute volume; f ¼ ventilator frequency; and VT ¼ tidal volume.
If the practical limits of the high frequency end of this relationship are considered, there must be a lower limit on tidal volume size that will effectively provide alveolar ventilation. That lower limit is related to the effective or physiologic dead space of the lungs by the following equation:
|
ð2Þ |
VA ¼ f ðVT VDÞ |
where VA ¼ alveolar ventilation, and VD ¼ effective or physiologic dead space.
Thus, as tidal volume approaches the size of the effective dead space of the lungs, ventilation of the alveoli becomes nil.
Physiologic Dead Space and the Lower Limit of Tidal Volume
Anatomic dead space of mammalian lungs is generally considered to be 2 mL kg 1 body weight (7). When one breathes normally, effective or physiologic dead space must be at least as large as anatomic dead space, because one pushes the dead space gas back into the alveoli ahead of the fresh gas during inhalation. What happens in the panting animal is another matter, as Henderson and associates described in 1915 (8).
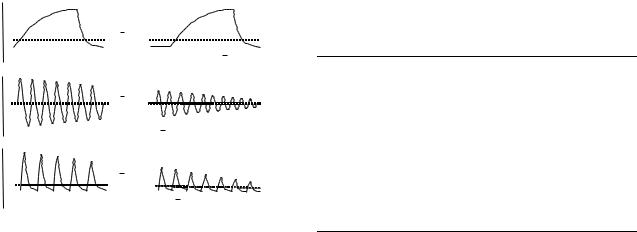
Henderson et al. (8) demonstrated that panting animals breathe very shallowly as well as rapidly. They hypothesized that physiologic dead space changes at rapid respiratory rates in mammals, and they measured these effects on themselves. They also performed a series of experiments using smoke to demonstrate how inhaled gas penetrates through the anatomic dead space with rapid inhalations in a manner that makes physiologic dead space become less than anatomic dead space (Fig. 1).
Figure 1. Henderson’s smoke experiment. (a) A long thin spike or jet stream of smoke shoots downstream when suddenly blown into a glass tube. (b) The jet stream disappears when flow stops and diffusion takes place. (c) This effect can be duplicated in the opposite direction if fresh gas is drawn back into the smoke filled tube with a sudden inhalation. (d) Imperfections in the tube walls (such as a bulb) have little effect on the shape of the jet stream. (Adapted with permission from Ref. 8, p. 8. # 1915, American Physiology Society.)
Regardless of how much physiologic dead space can be reduced in the panting animal, there is still the matter of providing adequate alveolar ventilation as defined by Eq. 2. Thus, the extent to which smaller tidal volumes can be used to ventilate the alveoli has to be balanced by an increase in breathing frequency, as defined by Eq. 1. Panting animals breathe very rapidly, of course, but humans do not pant as a rule. So, how can the benefits of increasing ventilator frequency for humans be explained?
The Natural Frequency of the Lungs
There is a mechanical advantage to ventilating lungs at frequencies higher than normal breathing frequency. This phenomenon was revealed by a diagnostic technique for measuring airway resistance called forced oscillations (9).
Applying forced oscillations to measure airway resistance requires a person to hold a large bore tube in their mouth and allow small volumes of gas to be oscillated in and out of their lungs by a large loudspeaker. The frequency of oscillations produced by the speaker is varied through a spectrum from low ( 1 Hz) to high ( 60 Hz), and the pressure amplitudes of the oscillations are measured along with the flow rate of the gas that is passing in and out of the lungs (Fig. 2 depicts the test set up). Although the volume of gas moving in and out of the lungs is a constant, pressure amplitude varies with frequency and is minimized at the resonant or natural frequency of the lungs.
The concept that the lungs have a natural frequency is explained by consideration of lung mechanics. There are three elements to lung impedance (those things that impede the flow of gas in and out of the lungs): airway resistance, lung compliance, and inertance. We normally are not concerned about inertance, since it is concerned with the energy involved in moving the mass in the system,

P |
. |
Constant volume generator |
|
(loudspeaker) |
|||
|
|||
|
V |
|
.
Figure 2. Measuring airway resistance at the resonant or natural frequency of the lungs using forced oscillations. (Used with permission. # 2003, Bunnell Inc.)
most of which is gas, and gas does not have much mass. Therefore,itdoesnottakemuchenergytoovercomeinertance when one breathes: unless one is breathing very rapidly.
In the forced oscillations determination of airway resistance, the point of minimum pressure amplitude marks the frequency at which the energy necessary to overcome the elasticity of the lungs is supplied by the energy temporarily stored in the inertial elements of the system (i.e., the gas rushing in). (We normally measure lung elasticity inversely as lung compliance.) As the lungs recoil at the end of the gas-in phase, the elasticity of the lungs imparts its energy to turn the gas around and send it back out to the loudspeaker.
When the natural frequency or resonance is reached, the speaker and lungs exchange the gas being forced in and out of the lungs with ease. The lungs and the speaker accept the gas and recoil at just the right times to keep the gas oscillating back and forth with minimal energy required to keep the gas moving. At this point, the only element impeding gas flow is frictional airway resistance, which works against the gas coming in and going out. Its value can be calculated by dividing pressure amplitude by the gas flow rate when pressure amplitude is minimized.
The smaller the lungs are, the higher the natural frequency. The natural frequency of adult lungs is 4 Hz, while that of premature infant lungs is closer to 40 Hz.
Putting Two and Two Together: How Can We HFV?
Combining the two concepts that describe the relationships of gas velocity, physiologic dead space, breathing frequency, and lung mechanics led us to HFV. We reported that one can then achieve adequate minute ventilation and compensate for very small tidal volumes in paralyzed animals by increasing ventilatory frequency to several hundred breaths per minute in 1978 (10). Pushing small volumes of gas into the lungs at high velocities reduced effective dead space volume and pushed the lower limit of effective tidal volume below anatomic dead space volume
HIGH FREQUENCY VENTILATION |
499 |
( 2 mL kg 1). Increasing frequency to near resonant frequency also allowed us to minimize airway pressure.
As HFVs were developed and clinical use in newborn intensive care units (NICUs) became widespread in the 1980 and 1990s, numerous theories and experiments refined our concepts of how it all works. A number of prominent physiologists and bioengineers tackled the analysis and interpretation of gas exchange within the lungs during HFV while clinicians were seeking to identify appropriate applications of the new technique and all its intricacies. A few notable contributions will be discussed here.
Fredberg (11) and Slutsky et al. (12) analyzed mechanisms affecting gas transport during high frequency oscillation, expanding traditional concepts of convection and diffusion to include their combined effects, and termed the collection: augmented transport. Their analyses and those of Venegas et al. (13) and Permutt et al. (14) revealed that our traditional appreciation of the relationship between minute volume and CO2 elimination must be modified during HFV to reflect the increased contribution of tidal volume, as
VCO2 / f a VTb |
ð3Þ |
where the exponent b is greater than the exponent a. For practical purposes, most people now accept this relationship as
|
2 |
ð4Þ |
VCO2 / f VT |
||
Slutsky also explored the limitations of HFV by measuring the effect of bronchial constriction on gas exchange. When the peripheral airways of dogs were constricted by administration of histamine, HFOV was no longer as effective at higher frequencies. (This issue is discussed later when the effectiveness of various types of HFVs for different pathophysiologies are explored.)
Venegas and Fredberg explored the importance of frequency during HFV in their classic paper of 1994, subtitled: ‘‘Why does high frequency ventilation work?’’ (15). They found that the resonant frequency of the smallest prematurely born infant with RDS (respiratory distress syndrome) is approximately 40 Hz. At that frequency, the minimum pressure amplitude is required to ventilate the lungs. However, the shape of theoretical curves of pressure amplitude measured at the carina versus frequency for infants with various lung conditions is most interesting as illustrated in Fig. 3, which was constructed using their concepts.
Figure 3 illustrates four essential considerations concerning the application of HFV for newborn infants.
1. Decreasing lung compliance moves the optimal frequency for HFV to the right (i.e., toward higher frequencies).
2. Increasing airway resistance moves the optimal frequency for HFV to the left (i.e., toward lower frequencies); and
3. There is diminishing value in applying HFV for infants at frequencies above ~ 10 Hz as far as airway pressure is concerned.
4. Choosing to operate at the ‘‘corner frequency’’ is an appropriate choice for HFV since there is little benefit

500 HIGH FREQUENCY VENTILATION
40 |
|
|
|
30 |
|
Preemie with |
|
fc |
RDS and PIE |
|
|
|
fopt |
|
|
|
|
|
|
20 |
fc |
Preemie with RDS |
|
PIP |
|
|
|
(cm H2O) |
|
|
fopt |
10 |
Preemie with |
|
|
|
|
||
|
|
|
|
|
normal lungs |
|
|
|
10 |
20 |
30 |
Frequency (Hz)
Figure 3. Theoretical peak carinal pressures for infants with normal lungs and lungs with poor compliance (RDS), poor airway resistance (asthma), and both conditions (RDS þ PIE). Note how infants with RDS are well served using the corner frequency (fc) of10 Hz (600 breaths per minute, bpm). Larger patients will exhibit curves with nearly identical shapes, but they will all be shifted to the left. (Adapted with permission from Ref. 15.)
above that frequency and more chance for gas trapping. Venegas and Fredberg define corner frequency as that frequency above which airway pressure required to provide adequate ventilation no longer rapidly decreases.
In other words, ventilating premature babies at 10 breaths s 1 is practically as efficient as ventilating them at their theoretical resonant frequency of 40 ‘‘breaths’’ s 1, where the danger of gas trapping is greatly increased. Patients with increased airway resistance require more careful consideration of the decreased benefits of exceeding the corner frequency and are more safely ventilated at lower frequencies.
One can calculate the resonant and corner frequencies if values of lung compliance (CL), airway resistance (Raw), and inertance (I) are known, but that is rarely the case with patients in intensive care. Venegas and Fredberg provided the following formulas:
¼ |
ð |
q |
|
f0 |
|
¯ |
ð5Þ |
1= 2p CIÞ |
|||
where f0 ¼ resonant frequency, and |
|
||
fc ¼ 1=ð2pCRÞ |
ð6Þ |
||
where fc ¼ corner frequency. (Plug in typical values for lung compliance, inertance, and airway resistance of a pre-
mature |
infant, |
0.5 mL cm 1 H2O, |
0.025 cm H2O L s 2, |
||||
and 50 |
cm H |
1 |
1 |
|
1 |
and |
|
1 |
2O L |
s |
|
, respectively, and f0 ¼ 45 s |
|||
fc ¼ 6.4 s |
.) |
|
|
|
|
|
|
Finally, Venegas and Fredberg illustrated the value of using appropriate levels of positive end-expiratory pressure (PEEP). The PEEPs of 5–10 cm H2O dramatically decrease the pressure amplitude necessary to ventilate premature infants at all frequencies when lung compliance is normal, and at all frequencies above 6 Hz when lung compliance is reduced.
High-frequency |
PEEP / HFV valve |
||||||||||
Constant valve |
|
|
|
|
|
|
|
PEEP or |
|||
pressure |
|
|
|
|
|
|
|
|
|
|
synchronized |
|
|
|
|
|
|
|
|
|
|
||
or flow |
|
|
|
|
|
|
|
|
closure |
||
|
|
|
|
|
|
|
|
||||
|
|
|
|
Exhaust |
|||||||
|
Patient |
||||||||||
|
|
|
|
|
|
|
|
|
|||
Figure 4. Basic design of HFPPVs. (Used with permission.
# 2003, Bunnell Inc.)
HFV EQUIPMENT
Design Classifications
Figures 4–7 illustrate four different ways HFVs have been created. Figure 4 illustrates high frequency positivepressure ventilation (HFPPV), which is basically a CV that operates at HFV rates. Early devices worked in this manner, but they seldom worked at the very high frequencies used with infants.
Figure 5 illustrates high frequency flow interruption (HFFI), where positive pressure oscillations are created by releasing gas under pressure into the breathing circuit via an HFV valve mechanism. The valve may be a solenoid valve or valves, a spinning ball with a hole in it, and so on. Early HFVs used long, small diameter exhaust tubes to increase impedance to gas flow oscillating at HFV frequencies in the expiratory limb so that the HFV oscillations would preferentially flow in and out of the patient.
High frequency oscillatory ventilators (HFOVs) work in a similar manner to HFFIs, as shown in Fig. 6, except that the pressure oscillations in the patient’s breathing circuit are caused by an oscillating piston or diaphragm. Again, the impedance of the expiratory limb of the circuit tubing must be higher than the impedance of the patient and his ET tube when the gas flowing through the circuit is oscillating at HFV frequencies. The major difference between HFOV and HFFI is that pressure in the ventilator circuit during HFOV oscillates below atmospheric pressure in an effort to actively assist the patient’s expiration. (This topic is discussed further below when gas trapping is addressed.)
Finally, Fig. 7 illustrates high frequency jet ventilation (HFJV), where inspiratory gas is injected into the patient’s ET tube via a jet nozzle. Jet nozzles have been fashioned out of needles or built into special ET tubes or ET tube adapters as discussed below.
Each HFV approach introduces fresh gas into the patient’s airway at about 10 times the patient’s normal breathing frequency. The last three designs incorporate a separate constant flow of gas that passes by the patient’s
|
|
High-frequency |
|
|
|
|
|
|
|
|
||||
Constant |
flow-interrupting valve |
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||
pressure |
|
|
|
|
|
|
IMV / PEEP valve |
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
PEEP or IMV |
Constant flow |
|
|
|
|
|
|
|
|
|
|
|
|
pressure |
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
Exhaust |
|||||||||
|
|
|
|
|
|
|
||||||||
|
|
|
|
Patient |
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|||
Figure 5. Basic design of HFFIs. (Used with permission. # 2003, Bunnell Inc.)

Oscillating piston or diaphragm
Paw valve

 Paw Pressure
Paw Pressure
Constant flow
Exhaust
Patient
Figure 6. Basic design of HFOVs. (Used with permission. # 2003, Bunnell Inc.)
ET tube and out a large orifice valve to control baseline PEEP and mean airway pressure in the circuit. The patient may also breathe spontaneously from this gas stream, which may be provided by a built-in mechanism or by a separate conventional ventilator. Conventional IMV (intermittent mandatory ventilation) may be combined with HFV in this way. Additional hybrid devices that are more difficult to characterize have also been created, but the currently most common used HFVs are HFOVs, HFJVs, and conventional ventilators with built-in HFOV modules.
In the early 1980s, the FDA (U.S. Food and Drug Administration) decided that 150 breaths min 1 would be the lower limit of what they would define as an HFV, and they placed rigorous Class III restrictions on any ventilator that operates above that frequency. As a result, there have been only six HFVs approved for use in the United States, three for infants and children, two for adults, and one HFV that was granted Class II approval (i.e., not needing proof of safety and efficacy since it was substantially equivalent to devices marketed before 1976, the date U.S. law was amended to require proof of safety and efficacy before new products can be marketed). At least four other HFVs are available outside the United States.
Of the FDA approved devices, two HFVs have been withdrawn from the market by the major corporation that acquired the smaller companies that developed them. Therefore, we are left with one HFJV, one HFOV for infants and children, one HFOV for children and adults, and a Class II HFV hybrid device designed for patients of all sizes. These four devices will be discussed in more detail below.
HFJV: High Frequency Jet Ventilators
The HFJVs inject inspired gas into the endotracheal tube via a jet nozzle. The Bunnell LifePulse High Frequency
High-frequency |
|
|
|
|
|
|
|
|
|
|
||||
Flow-interrupting valve |
|
|
|
|
|
|
|
|
|
|
||||
Constant |
Jet |
|
|
|
|
|
|
|
|
|
||||
pressure |
IMV / PEEP valve |
|||||||||||||
|
|
|
|
|
nozzle |
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
PEEP or IMV |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
pressure |
Constant flow |
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|||||
Exhaust
Patient
Figure 7. Basic design of HFJVs. (Used with permission. # 2003, Bunnell Inc.)
HIGH FREQUENCY VENTILATION |
501 |
Figure 8. Bunnell life pulse HFV. (Used with permission.
# 2003, Bunnell Inc.)
Ventilator is the only HFJV currently available for intensive care in the United States (Fig. 8). It was designed for infants and children up to 10 years of age and operates at rates between 240 and 660 bpm. It is also used in tandem with a conventional ventilator, which provides for the patient’s spontaneous breathing, delivery of occasional sigh breaths, and PEEP.
The LifePulse is a microprocessor controlled, pressure limited, time cycled ventilator that delivers heated and humidified breaths to the ET tube via a LifePort adapter (Fig. 9). A small Patient Box placed close to the patient’s head contains an inhalation valve and pressure transducer for monitoring airway pressures in conjunction with the LifePort adapter. Peak inspiratory pressure (PIP) is feedback controlled by regulating the driving pressure (servo pressure) behind the jet nozzle. More detailed information on the device can be found on the manufacturer’s website: www.bunl.com.
The theory of operation behind the LifePulse is that pulses of high velocity fresh gas stream down the center of the airways, penetrating through the dead-space gas, while exhaled gas almost simultaneously moves outward in the annular space along the airway walls. This countercurrent action facilitates mucociliary clearance while it minimizes effective dead space volume.
The pressure amplitude (DP) of LifePulse HFJV breaths is determined by the difference between PIP and the CVcontrolled PEEP. Its value is displayed continuously on the LifePulse front panel along with mean airway pressure, PIP, PEEP, and Servo Pressure.
Servo Pressure on the LifePulse is a direct reflection of the gas flow needed to reach the set PIP, so it varies with the patient’s changing lung mechanics. Alarm limits are automatically set around the Servo Pressure to alert the operator to significant changes in the patient’s condition as well as the tubing connecting the patient to the HFJV.

502 HIGH FREQUENCY VENTILATION
Figure 9. LifePort ET tube adapter for HFJV. (Used with permission. # 2003, Bunnell Inc.)
A low limit alarm would infer that the patient’s lung compliance or airway resistance has worsened, and the LifePulse is using less gas (i.e., smaller tidal volumes) to reach the set PIP in that circumstance. A high limit alarm would infer that the patient’s condition has improved, larger tidal volumes are being delivered, and the operator should consider weaning PIP in order to avoid hyperventilation.
Alarm limits are also automatically set around monitored mean airway pressure.
HFOV: High Frequency Oscillatory Ventilators
The SensorMedics 3100A HFOV for infants and children and its sister model, the 3100B for adults, are the only pure HFOVs currently available in the United States. Sinusoidal oscillatory ventilation is produced by an electromagnetically driven floating piston with adjustable frequency and amplitude. Inspiratory gas is supplied as bias flow, which escapes from the very large diameter (1.5 in. ID, 38 mm) patient breathing circuit via a traditional dome valve that
controls mean airway pressure. All pressures are monitored at the connection to the ET tube. The SensorMedics 3100A HFOV is illustrated in Fig. 10 and more information is available at www.sensormedics.com.
The 3100 HFOVs operate on the principle that high frequency oscillations that are in tune with the natural frequency of the patients lungs will preferentially move in and out of the lungs, as opposed to the exhaust system of the patient circuit. Haselton and Scherer illustrated a new gas transport principle that applies to HFOV (16). Differences in the velocity profiles of inspiration and expiration during HFOV created by the branching architecture of the lungs enables inspiratory gas to advance down the center of the airways while exhaled gas moves up along the airway walls as the piston of the HFOV pulls the gas back. The net effect of many oscillations is similar to, but less pronounced than, the flow characteristics of HFJV flow in the airways. Fresh gas tends to flow down the center of the airways while exhaled gas recedes back along the airway walls.

Figure 10. SensorMedics 3100A high frequency oscillatory ventilator. (Used with permission. # 2003, SensorMedics Inc.)
The 3100 HFOVs have six control settings:
1.Frequency, which is adjusted to suit patient size and lung time constants.
2–4. Bias gas flow rate, Mean Pressure Adjust and Mean Pressure Limit, which together set mean airway pressure.
5.Power, which sets DP.
6.% Inspiratory Time, which sets I:E (inspiratory to expiratory time ratio; typically set at 33%).
Mean airway pressure is the primary determinant of oxygenation, and DP is the primary determinant of tidal volume and ventilation (CO2 removal). However, all controls are open-loop: increasing frequency decreases tidal volume and visa versa, and changing bias gas flow rate or power may change mean airway pressure. Mean airway pressure is monitored in the HFOV circuit, so changes there are apparent, but changes in tidal volume due to setting changes or changes in a patient’s lung mechanics are not apparent. Given the squared relationship between tidal volume and CO2 removal noted above, changes in frequency move PaCO2 in the opposite direction of what one would anticipate with conventional ventilation. (Increasing frequency increases PaCO2; decreasing frequency decreases PaCO2.) Thus, continuous or frequent monitoring of arterial PaCO2 is recommended during HFOV, as it is with all HFVs and conventional ventilation of premature infants due to the potential for cerebral injury associated with hyperventilation (more on that topic later).
HFFIs and Other Hybrids
Conventional infant ventilators with built-in HFV modules, such as the Dra¨ger Babylog 8000 plus (Dra¨ger Medical AG & Co. KGaA) and the popular Infant Star Ventilator, which will no longer be supported by its manufacturer after May 2006, have been widely used in the
HIGH FREQUENCY VENTILATION |
503 |
United States, Canada, Europe, and Japan. In general, these hybrid HFVs are not as powerful as the stand-alone HFVs, so their use is limited to smaller premature infants (<2 kg). The VDR Servolator Percussionator (Percussionaire Corporation, Sand Point ID), however, was designed to ventilate adults as well as premature infants. (Detailed information on these devices can be viewed on the manufacturers’ websites: www.draeger-medical.com and www. percussionaire.com.) The mechanical performances of these devices vary widely, as do their complexities of operation and versatilities as infant ventilators. (See the section Equipment Limitations.)
Design Philosophy for Clinical Applications
The philosophy for controlling arterial blood gases with HFVs is similar to that used with pressure-limited conventional ventilation, especially when HFVs are used to treat homogeneous lung disorders such as RDS (respiratory distress syndrome) in prematurely born infants. The alveoli of these surfactant-deficient lungs must be opened with some type of recruitment maneuver and kept open with appropriate mean airway pressure (Paw) or PEEP in order for the lungs to make oxygen available to the blood stream. Ventilation is accomplished at a frequency proportionate to the patient’s size and lung mechanics using a peak airway pressure or pressure amplitude above PEEP that creates a tidal volume that produces an appropriate arterial PCO2. Pulse oximeters, which report the oxygen percent saturation of arterial blood, are great indirect indicators of when lungs have opened up, because oxygenation is highly dependent on the number of alveoli that are open and participating in gas exchange with the blood stream. Chest wall motion is a good indirect indicator of ventilation since it reflects the amount of gas that is passing in and out of the lungs.
The usual approach to initiation of HFV is to choose a frequency that is appropriate for the size of the patient and his lung mechanics, starting with 10 Hz or 600 bpm for the smallest premature infant with RDS and working downward as the size of the patient increases and lung mechanics improve. A good rule of thumb is to choose a frequency 10 times greater that the patient’s normal breathing frequency, which would put HFV for adults at rates <200 bpm. Higher rates may be used with HFOV since exhalation is nonpassive, but gas trapping can still result unless mean airway pressure is kept high enough to keep the airways open during the active exhalation phase. Operating an HFOV with a 33% inspiratory time (I:E ¼ 1:2) lessens negative pressure during exhalation compared to longer I-times (e.g., I:E ¼ 1:1) thereby decreasing the potential for causing airway collapse.
With HFJV, the shortest possible inspiratory time ( 0.020 s) usually works best; it maximizes inspiratory velocity, which helps reduce effective dead space, and minimizes I:E, which allows more time for exhalation to avoid gas trapping. These characteristics also minimize mean airway pressure, which is very useful when treating airleaks and for ventilation during and after cardiac surgery. The high velocity inspirations also enable ventilation of patients with upper airway leaks and tracheal tears.
504 HIGH FREQUENCY VENTILATION
Treatment of obstructive lung disorders absolutely requires longer exhalation times, so HFV must be used at lower frequencies on these patients. HFJV I:E varies from 1:3.5 to 1:12 as frequency is reduced from 660 to 240 bpm when inspiratory time is held constant at its shortest value.
The HFV is not intended and may in fact be contraindicated for patients with asthma, unless helium–oxygen mixtures become part of the mix (17).
Once a frequency and duty cycle (% I-time or I:E) is chosen, airway pressure settings (PIP, PEEP, or DP) are set to provide HFV tidal volumes that noticeably move the chest. If chest wall movement is not apparent, ventilation is probably not adequate. Use of transcutaneous CO2 monitoring is of great benefit here.
Finally, mean airway pressure (Paw) or PEEP must be optimized. Too little Paw or PEEP will lead to atelectasis and hypoxemia, and too much Paw or PEEP will interfere with cardiac output. One of the true benefits of HFV, however, is that higher Paw and PEEP can be used without increasing the risk of iatrogenic lung injury. (The small HFV tidal volumes do not create the same potential for creating alveolar ‘‘stretch’’ injury as larger CV tidal volumes do.) Pulse oximeters can be great indirect indicators of appropriate lung volume, but one must be vigilant in detecting signs of decreased cardiac output.
Conventional ventilation is sometimes required or available for tandem use with certain HFVs. The CV breaths are most useful with nonhomogeneous lung disorders and to facilitate alveolar recruitment with atelectatic lungs. The usual strategy is to reduce CV support when starting HFV (assuming the patient is on CV prior to HFV) to 5–10 bpm while optimal Paw and PEEP is being sought, and then reduce CV support further.
Now some of the performance differences in HFV equipment and how those differences may affect successful HFV implementation will be examined.
HFV Equipment Limitations
There have been few head-to-head comparisons of HFV equipment. The most recent comparison were by Hatcher et al. and Pillow et al. where they compared several neonatal HFOVs and found wide variations in performance, complexity, and versatility (18,19). Pillow et al. concluded that the clinical effects of manipulating ventilator settings may differ with each HFOV device. In particular, the pressure amplitude required to deliver a particular tidal volume varies with device, and the effect of altering frequency may result in very different effects on tidal volume
and PaCO2.
The first rigorous analysis of HFVs was undertaken by Fredberg et al. in preparation for the HiFi Study (20). They bench tested eight HFVs in an effort to provide the clinicians who were to participate in the study comparative data that they could use to select an HFV for use in their study. (They selected the Hummingbird HFOV, manufactured by MERA of Japan.) Despite the wide diversity of ventilator designs tested, certain common features emerged. In almost all devices, delivered tidal volume was sensitive to endotracheal tube size and airway resis-
tance and invariant with respiratory system compliance. These results supported the theoretical basis for why high frequency ventilation may be a better treatment for RDS compared to pressure-limited CV (conventional ventilation), because low lung compliance is its paramount pathophysiologic feature.
These HFV bench tests also found that tidal volume decreased with increasing frequency with all HFOVs where I:E (inspiratory to expiratory time ratio) was held constant and was invariant with HFJV and HFFI devices where I-time was held constant. Peak inspiratory flow rates for a given tidal volume and frequency were significantly higher with the HFJV and HFFI as well. Proximal airway pressure was also a poor indicator of distal pressure with all devices.
Two other studies compared HFJV to HFOV. Boros and associates compared the pressure waveforms measured at the distal tip of the endotracheal tube of the Bunnell LifePulse HFJV and the Gould 4800 HFOV (precursor to the SensorMedics 3100A HFOV) in normal, paralyzed, and anesthetized cats (21). They found that the HFOV required higher PIP, DP, and Paw to get the same PaCO2, PaO2, and pH compared to HFJV. Likewise, PaCO2 was higher and pH and PaO2 were lower with HFOV when the same airway pressures were used. However, different frequencies were used with the two ventilators; 400 bpm with HFJV and 900 bpm (15 Hz) with HFOV.
Zobel and associates also found that HFJV was effective at lower airway pressure compared to HFOV (22). They used a piglet model of acute cardiac failure and respiratory failure and also measured airway pressure at the distal tip of the endotracheal tube. The HFJV used was an Acutronic AMS-10001 (Acutronic Medical Systems AG, Switzerland) operating at 150 bpm with an I:E of 1:2. The HFOV was a SensorMedics 3100A operating at 10 Hz and 1:2.
Why do HFOVs (presumably) operate at higher Paw compared to HFJV? The answer to this question may be related to gas trapping, HFV rates, and what happens during exhalation. In both of the animal studies just discussed, HFJV rate was considerably lower than HFOV rate. Exhalation is passive during HFJV, so lower rates must be employed to allow sufficient exhalation time to avoid gas trapping. The HFOVs suck the gas back out of the lungs during the expiratory phase, and the physiologic consequence can be, not surprisingly, airway collapse. However, the Paw employed during HFOV determines the importance of this effect.
Bryan and Slutsky set the tone for the future of HFVs when they noted that this mode of ventilation is ideally designed for treatment of patients with poor lung compliance (23). The higher Paw required to match the pathophysiology of such patients also serves to splint the airways open during HFOV so that the choking effect of active expiration is mitigated.
In conclusion, both modes of HFV can cause gas trapping; they just do it by different mechanisms. The HFOV can choke off airways when Paw is insufficient to mitigate the effect of active expiration, and HFJV will trap gas when expiratory time is insufficient to allow complete exhalation of inspired tidal volume. One cannot lower Paw during HFOV beyond the point where choking is made evident by
a rise in a patient’s PCO2. With HFJV, one should not increase frequency beyond the point where PEEP monitored in the endotracheal tube, as it is with the Bunnell LifePulse, begins to rise inadvertently. If the automatically set upper alarm limit on mean airway pressure with the LifePulse is activated, there is a good chance that this rise in Paw is due to inadvertent PEEP. The remedy for that circumstance is to decrease HFJV frequency, which lengthens exhalation time and allows the PEEP to fall back to set level.
Airway Pressure Monitoring During HFV
While airway pressures were monitored at the distal tip of the ET tube in the animal studies noted above, monitoring at this location is seldom done currently, because the Hi-Lo ET tubes (formerly manufactured by Mallinckrodt, Inc.) are no longer available. Thus, airway pressure monitoring is done either at the standard ET tube adapter connection during HFOV or at the distal tip of the special LifePort adapter during HFJV. In either case, the pressure waveform measured deep in the lungs at the alveolar level is greatly damped (Fig. 11). Gerstmann et al. reported that measurement of pressure amplitude in the alveoli of rabbits during HFOV at 15 Hz was only 10% of that measured proximal to the ET tube (24).
Meaningful monitoring of airway pressure during HFOV is limited to mean airway pressure, and that is only representative of mean alveolar pressure in the absence of gas trapping, as noted above. Relative values of pressure amplitude at the proximal end of the ET tube are indicative of tidal volume size, and they are typically expressed as such by the various HFOVs.
Peak inspiratory pressure (PIP) and PEEP as well as Paw are measured during HFJV at the distal tip of the LifePort ET adapter. The PEEP is representative of alveolar PEEP at this location in the absence, again, of gas trapping. However, the PIP at this location is a gross overestimate of peak pressure in the alveoli. Mean airway pressure may slightly overestimate mean alveolar pressure as shown by the study of Perez-Fontan et al. (25).
|
Trachea & Proximal Airways |
Distal Airways & Alveoli |
|
|
20 |
|
|
P |
15 |
CMV |
|
10 |
|
||
Paw |
|
||
|
5 |
|
|
|
|
|
|
|
0 |
|
Amplitude fixed; Paw fixed. |
|
20 |
HFOV |
|
|
15 |
|
|
P |
Paw |
|
|
10 |
|
||
|
|
|
|
|
5 |
|
|
|
0 |
Amplitude attenuates; Paw fixed when I:E = 1:1. |
|
|
20 |
|
|
P |
15 |
HFJV |
|
10 |
Paw |
|
|
|
5 |
|
|
|
0 |
Amplitude attenuates; PEEP fixed; Paw slightly declines. |
|
Figure 11. HFV Airway Pressure Waveform Dampening. (Used with permission. # 2003, Bunnell Inc.)
HIGH FREQUENCY VENTILATION |
505 |
HFV APPLICATIONS IN NEONATES AND CLINICAL OUTCOMES
Homogeneous Atelectatic Lung Disease (e.g., RDS) and Prevention of Lung Injury
Ever since the completion of the first multicenter, randomized, controlled HFV trial was published in 1989, reporting no benefit for premature infants with RDS and an increased risk of severe cerebral injury (26), the choice of HFV to prevent lung injury in preterm infants has been hotly debated. Some recent trails have demonstrated that if HFVs are implemented within hours of a premature infant’s birth with the proper strategy, results are positive. Other recent studies have not been positive.
The HiFi Trial, as the first multicenter, randomized, controlled trial was labeled, was criticized for the general lack of clinical experience of the investigators and failure to adhere to the most appropriate strategy for recruiting and maintaining appropriate lung volume (15). Later multicenter, randomized controlled trials conducted in the 1990s using both HFJV and HFOV demonstrated significant reductions in chronic lung disease (CLD) measured at 36 weeks postconceptional age (PCA) in this patient population with practically no difference in adverse effects (27,28). (There was a slightly higher incidence of PIE in the experimental group of the HFOV study.) The demographics and results of these two trials are illustrated in Tables 1 and 2.
The results of the HFJV study were criticized for a lack of well-defined ventilator protocols for the conventionally ventilated control group, whereas protocols for both the HFOV and SIMV control groups in the HFOV study, conducted several years later, were well conceived and monitored during the study. Therefore, it is interesting to note that the major outcome measures of CLD at 36 weeks PCA in the control groups of the two studies were almost identical.
Other HFV studies revealed an increase in severe cerebral injury that appears to be related to hyperventilation and hypocarbia during HFV (29–32). Other criticisms of recent trials with negative or equivocal results include the same strategy issues plus choice of HFV devices, limited time on HFV before weaning back to CV, and so on (33).
Because of these mixed results, HFVs have yet to be generally accepted for early treatment of premature infants with RDS and prevention of lung injury.
Table 1. Demographics of Two Multicenter, Randomized Controlled Trials with HFOV and HFJV
|
|
HFJV |
|
|
|
HFOV |
|
Design/Demographics |
Studya |
|
Study10 |
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
Treatment Groups |
|
HFJV |
CV |
HFOV |
SIMV |
||
|
|
|
|
|
|
|
|
Number of Patients |
|
65 |
65 |
|
244 |
254 |
|
|
|
||||||
Mean Birth Weight, kg |
1.02 |
1.02 |
0.86 |
0.85 |
|||
Mean Gestational Age |
27.3 |
27.4 |
26.0 |
26.1 |
|||
Age at Randomization, h |
8.1 |
8.3 |
|
2.7 |
2.7 |
||
1 min/5 min Apgar Scores |
3.5/7 |
4/7 |
|
5/7 |
5/7 |
||
FIO2 at Entry |
0.62 |
0.69 |
0.57 |
0.60 |
|||
Mean Airway Pressure |
10 |
10 |
|
8.2 |
8.3 |
||
at Entry |
|
|
|
|
|
|
|
aSee Ref. 9.

506 HIGH FREQUENCY VENTILATION
Table 2. Significant Respiratory and Clinical Outcomes of HFOV and HFJV Early Application Trials on Premature Infants with RDS
Significant Respiratory and |
HFOV Study |
HFJV Study |
|
|
|||
|
|
|
|
|
|
|
|
HFOV |
SIMV |
HFJV |
CV |
|
|
||
Clinical Outcomes |
|
|
|||||
|
|
|
|
|
|
|
|
Alive w/o CLD at 36 weeks PCA |
56% |
47% |
|
68% |
48% |
|
|
|
p ¼ 0.046 |
p ¼ 0.037 |
|
|
|||
Age at extubation, days |
13 |
21 |
|
- |
|
|
|
|
p < 0.001 |
|
|
|
|
||
Crossovers or Exitsa |
25/244 (10%) |
49/254 (19%) |
|
3/65 (5%) |
21/65 (32%) |
||
|
p ¼ 0.07 |
|
p < 0.01 |
||||
Success after Crossover |
|
|
|
|
14/21 (67%) |
0/3 (0%) |
|
|
|
|
|
|
p ¼ 0.06 |
||
Supplemental O2 |
27% |
31% |
|
|
5.5% |
23% |
|
|
p ¼ 0.37 |
|
p ¼ 0.019 |
||||
PIE |
20% |
13% |
|
|
|
|
|
|
p ¼ 0.05 |
|
|
|
|
||
Pulmonary Hemorrhage |
2% |
7% |
|
|
6.3% |
10% |
|
|
p ¼ 0.02 |
|
p > 0.05 |
||||
aSimilar failure criteria were prospectively defined in both studies. Those who met the criteria in the HFJV study were crossed over to the other mode, while those who met the criteria in the HFOV study exited the study and were treated with whatever mode of ventilation the investigators deemed appropriate, including HFJV (personal communication, David Durand, MD). Data on all patients were retained in their originally assigned group in both studies.
Homogeneous Restrictive Lung Disease (e.g., Congenital
Diaphragmatic Hernia)
While theories support use of HFV in cases where the lungs are uniformly restricted by acute intra-abdominal disease or postsurgically in infants with congenital diaphragmatic hernia, omphalocele, or gastroschisis, there are no randomized controlled trials due to the rarity of these disorders. Despite this lack of controlled trials, HFV has been widely accepted as an appropriate treatment for this category of lung disease due to the futility of CV treatment in severe cases.
Keszler et al. demonstrated improved gas exchange and better hemodynamics with HFJV in an animal model of chest wall restriction (34) and later reported improved ventilation and hemodynamics in a series of 20 patients with decreased chest wall compliance (35). Fok et al. reported improved gas exchange with HFOV in eight similar patients who were failing CV (36).
Nonhomogeneous Atelectatic and Restrictive Lung Disease (e.g., RDS with Tension PIE)
Pulmonary interstitial emphysema (PIE) in the premature infant creates a non-homogeneous lung disease: parts of the lungs are collapsed as a result of surfactant deficiency while other parts become overexpanded with gas trapped in interstitial areas. Air leaks like PIE originate most commonly in premature infants near the terminal bronchial (37). As gas dissects into interstitial spaces, it invades and dissects airway and vascular walls moving towards the larger airways and vessels and the pleural space where pneumothoraces are formed (38). While positive-pressure CV may successfully penetrate such restricted airways, the consequence may well be accumulation of trapped gas in the alveoli and subsequent alveolar disruption, which produces the classical picture of PIE on X ray.
The HFJV quickly gained a reputation for superior treatment of PIE in the early days of its clinical application. A multicenter randomized trial of HFJV compared to rapid rate (60–100 bpm), short I-time (0.20–0.35 s) CV for the treatment of PIE confirmed anecdotal findings of faster and more frequent resolution of PIE on HFJV. Survival in the stratified group of 1000–1500 g birth weight infants was most evident (79% with HFJV vs. 44% with CV; p < 0.05). There was no difference in the incidence of adverse side effects.
There is, as yet, no comparable randomized trial of HFOV treatment for PIE. While anecdotal success has been reported, attempts to show an advantage with HFOV in a randomized controlled trial have so far been unsuccessful. It may be that the physical characteristics of the two types of HFVs coupled with the pathophysiologic characteristics of PIE are the reasons for this lack of success. Recall that one difference between HFV devices reported in the pre-HiFi bench studies by Fredberg et al. was that HFJVs squirt gas into the lungs at much higher flow rates compared to HFOV. That fact may make HFJV more sensitive to airway patency compared to HFOV.
Since CV breath distribution may be more affected by lung compliance while HFV breaths may be more affected by airway resistance, especially HFJV breaths with their high velocity inspirations, the distribution of ventilation in the nonhomogeneous PIE lung may be markedly affected by mode of ventilation. While the path of least resistance for CV breaths may lead to more compliant, injured areas of the lungs, HFJV breaths may automatically avoid injured areas where airway and vascular resistances are increased. Therefore, HFJV breath distribution may favor relatively normal airways in the uninjured parts of the lungs where ventilation/perfusion matching is more favorable.
The CV tidal volumes delivered with higher PEEP and Paw may dilate airways enough to help gas get into
restricted areas in babies with PIE, but those larger tidal volumes take longer to get back out. Much smaller HFV tidal volumes are more easily expired, especially those that were unable to penetrate the restricted airways where the lungs are injured.
Upper Airway Fistulas and Pneumothoraces
Theoretically, the small tidal volumes, high inspiratory velocities, and short inspiratory times of HFJV are ideally suited for treating pneumothoraces and broncho-pleural and tracheal-esophageal fistulae. Gonzalez et al. found that gas flow in chest tubes, inserted in a series of infants with pneumothoraces, dropped an average of 54% when six infants were switched from CV to HFJV (39). Their mean PaCO2 dropped from 43 to 34 Torr at the same time that their peak and mean airway pressures measured at the distal tip of the ET tube dropped from means of 41–28 and 15 to 9.7 cm H2O, respectively.
Goldberg et al. (40) and Donn et al. (41) similarly reported improved gas exchange and reduced flow through tracheal–esophageal fistulas.
Homogeneous Obstructive Lung Disease (e.g., Reactive
Airway Disease, Asthma)
The HFV should theoretically not be of much benefit in treating lung disorders such as asthma wherein airway resistance is uniformly increased. Low rates and long expiration times should be more effective. However, recent work with HFJV and helium-oxygen mixtures (heliox) demonstrated interesting potential for treating such disorders in patients requiring no more than 80% oxygen.
Tobias and Grueber improved ventilation in a one-year old infant with respiratory syncytial virus and progressive respiratory failure related to bronchospasm with HFJV by substituting a mixture of 80% helium/20% oxygen for compressed air at the air/oxygen blender (42). They hypothesized that the reduced density of helium compared to nitrogen enhanced distal gas exchange. Gupta and associates describe another case where HFJV and heliox rescued a 5 month old infant with acute respiratory failure associated with gas trapping, hypercarbia, respiratory acidosis, and air leak (43). The combination of HFJV with heliox led to rapid improvements in gas exchange, respiratory stabilization, and the ability to wean the patient from mechanical ventilation.
Nonhomogeneous Obstructive Lung Disease (e.g., MAS) and
ECMO Candidates
Clinical studies of infants with meconium aspiration syndrome (MAS) provide support for the use of HFV with this type of lung disease. These patients are potential candidates for extracorporeal membrane oxygenation (ECMO), so ability to avoid ECMO is a typical outcome variable in such studies.
Clark et al. randomized 94 full-term infant ECMO candidates to HFOV or CV in a multicenter study (44). Prospectively defined failure criteria were met by 60% of those infants randomized to CV while only 44% of those randomized to HFOV failed. Cross-overs to the alternate mode by those who failed were allowed, and 63% of those
HIGH FREQUENCY VENTILATION |
507 |
who failed CV were rescued by HFOV, while only 23% of those who failed HFOV were rescued by CV. (The latter comparison was statistically significant.) Overall, 46% of the infants who met ECMO criteria required ECMO.
A similar single-center study of HFJV versus CV involved 24 ECMO candidates with respiratory failure and persistent pulmonary hypertension of the newborn (PPHN) (45). Most of the infants in the HFJV-treated group (8 of 11) and 5 of 13 of the conventionally treated infants had either MAS or sepsis pneumonia. Treatment failure within 12 h of study entry occurred in only two of the HFJV-treated infants versus seven of the conventionally treated infants. The ECMO was used to treat 4 of 11 HFJV infants versus 10 of 13 control infants. Zero of nine surviving HFJV-treated infants developed chronic lung disease compared to four of 10 surviving controls ( p ¼ 0.08). Survival without ECMO in the HFJV group was 5 of 11 (45%) versus 3 of 13 (23%) in the control group. There was no statistical significance in any of these comparisons due to the small number of patients.
The degree to which pathophysiology predicts positive outcomes with respect to the ability of HFVs to rescue infants that become ECMO candidates has been explored in two additional clinical studies. Baumgart et al. evaluated their success with HFJV prior to instituting an ECMO program in 73 infants with intractable respiratory failure who by age and weight criteria may have been ECMO candidates (46). They found survival after HFJV treatment to be much higher in infants with RDS and pneumonia (32/38, 84%) compared to MAS/PPHN (10/26, 38%) or congenital diaphragmatic hernia (3/9, 33%). All patients initially responded rapidly to HFJV as measured by oxygen index (O.I., calculated as mean airway pressure in cm H2O multiplied by fraction of inhaled O2 divided by PaO2 in Torr). However, that improvement in survivors was realized and sustained during the first 6 h of HFJV treatment.
Paranka et al. studied 190 potential ECMO candidates treated with HFOV during 1985–1992 (47). All patients were born at 35 weeks gestational age or more and developed severe respiratory failure, as defined by an arterial to alveolar oxygen ratio ðPðA aÞO2 Þ < 0:2 or the need for a peak pressure of >35 cm H2O on CV. Fifty-eight percent (111 patients) responded to HFOV and 42% (79 patients) were placed on ECMO. Gas exchange improved in 88% of the infants with hyaline membrane disease (RDS), 79% of those with pneumonia, 51% with meconium aspiration, and 22% of those with congenital diaphragmatic hernia. They also found failure to demonstrate an improvement in PðA aÞO2 after six hours on HFOV to be predictive of failure.
During and After Cardiac Surgery
The ability of HFJV to hyperventilate while using lower mean airway pressure is a great asset when treating patients with cardiac problems. During surgery, the small tidal volumes and low mean airway pressure allow the surgeon to move the lungs out of the way, in order to visualize and work on the heart. After surgery, HFJV can gently hyperventilate the patient to encourage increased pulmonary blood flow while mean airway pressure is kept down (48–51).
508 HIGH FREQUENCY VENTILATION
PPHN and Nitric Oxide Therapy
Kinsella et al. demonstrated the potential of HFV to enhance delivery of nitric oxide (NO) for the treatment of PPHN in a large, multicenter, randomized controlled trial (52). Nitric oxide delivered with HFOV to infants with significant parenchymal lung disease was more effective than NO delivered by CV. NO has also been delivered successfully with HFJV (53). However, NO must be administered via the HFJV circuit in order for the patient to realize any beneficial effect from the gas (54). Inhaled NO does not work with HFJV when administered exclusively through the conventional ventilator circuit (55).
HFV APPLICATIONS IN CHILDREN AND ADULTS
While the bulk of the research and application of HFV has been aimed at the benefit of infants to date, the sheer number of potential applications for children and adults is far greater. Unfortunately, the number of HFVs available to treat adults is severely limited. There is only one instrument currently available in the United States specifically designed for ARDS in children and adults, the SensorMedics 3100B. (The Percussionaire VDR4-F00008 ventilator also provides HFV for adults. It was approved as a Class II device by the FDA.)
Acute respiratory distress syndrome is the obvious target for HFV treatment in adult intensive care. This syndrome affects 50 per 100,000 population with a mortality of 30–50%. It is a clinical syndrome of noncardiogenic pulmonary edema associated with pulmonary infiltrates, stiff lungs, and severe hypoxemia (56). Although the pathology of ARDS involves a number of features similar to RDS in infants, such as hyaline membranes, endothelial and epithelial injury, loss of epithelial integrity, and increased alveolar-capillary permeability, it may have a much greater inflammatory component.
The only treatment shown to positively impact mortality over the past several decades came from the ARDSnet Trial where CVs were used with a low tidal volume ventilatory strategy designed to reduce iatrogenic lung injury (57). Comparative treatments in this multicenter study of 861 patients included an experimental group where mean tidal volumes for the first 3 days of their treatments were 6.2 mL kg 1 body weight and a control group where tidal volumes were 11.8 mL kg 1. The experimental group had lower mortality and fewer days on mechanical ventilators.
With ARDSnet trial pointing in the general direction of smaller tidal volumes, it is not surprising that recent HFV trials appear very promising, especially since HFV investigators focused on NICU patients and worked their way up the learning curve. The most important lesson learned, and one that took many years to learn in the treatment of infants, was the importance of recruiting and maintaining adequate lung volume during HFV. Adult trials of HFV for ARDS now begin with a Paw 5 cm H2O greater than that currently being used with CV. Just as was learned with infants, it is safe to use higher PEEPs and mean airway pressures with HFVs smaller tidal volumes.
HFV Clinical Trails with Children and Adults
The importance of starting early with HFV on adults and children with ARDS was highlighted in several anecdotal and pilot trials. Smith et al. treated 29 children with severe ARDS complicated by pulmonary barotrauma with HFJV (58). Twenty (69%) survived, and the only statistically significant difference between survivors and nonsurvivors was the mean time on CV before initiating HFJV (3.7 days in survivors vs. 9.6 days in nonsurvivors). Fort et al. similarly found that survivors in a pilot study of HFOV for adults with ARDS were on CV 2.5 days before initiation of HFOV, while nonsurvivors were on CV for 7.2 days (59). Expected survival in the pilot study was <20%, actual survival was 47%.
Arnold et al. compared HFOV to CV in children with respiratory failure (60). Optimizing lung volume was emphasized in both the experimental and control groups. The strategy for optimizing lung volume in the CV group was to lengthen inspiratory times and increase PEEP in order to decrease required PIPs. They found significant improvement in oxygenation in the HFOV group as well as a lower need for supplement oxygen at 30 days postenrollment.
A recent prospective trial of HFOV for ARDS had similar results. Mehta et al. treated a series of 24 adults with severe ARDS with HFOV (61). Five of the patients were burn victims. Within 8 h of HFOV initiation, FIO2 and PaCO2 were lower and PaO2/FIO2 was higher than baseline values during CV throughout the duration of the trial. An obvious focus was placed on recruiting and maintaining adequate lung volume while on HFOV, since Paw was also significantly higher than that applied during CV throughout the HFOV trial. Unfortunately, this increase in Paw was associated with significant changes in hemodynamic variables including an increase in pulmonary artery occlusion pressure (at 8 and 40 h) and central venous pressure (at 16 and 40 h), and a reduction in cardiac output throughout the study. Thus, Paw may not have been optimized. However, 10 patients were successfully weaned from HFOV and 7 survived. Again, there was a statistically significant difference in the time spent on CV prior to initiation of HFV: 1.6 days for survivors versus 5.8 days for the nonsurvivors.
Noting the importance of early intervention, Derdak et al. designed a multicenter, randomized, controlled trial comparing the safety and effectiveness of HFOV versus CV in adults with less severe ARDS (62). (The authors nicknamed their trial: the MOAT Study.) Inclusion criteria included PaO2/FIO2 200 mmHg (26.66 kPa) on 10 cm H2O PEEP, and 148 adults were evenly randomized. Applied Paw was significantly higher in the HFOV group compared with the CV group throughout the first 72 h. The HFOV group showed improvement in PaO2/FIO2 at <16 h, but this difference did not persist beyond 24 h. Thirty day mortality was 37% in the HFOV group and 52% in the CV group ( p ¼ 0.102). At 6 months, mortality was 47% in the HFOV group and 59% in the CV group ( p ¼ 0.143). There were no significant differences in hemodynamic variables, oxygenation failure, ventilation failure, barotraumas, or mucus plugging between treatment groups.
The MOAT Study indicates that HFOV is safe and effective for ARDS, and the FDA approved the SensorMedics 3100B for ARDS. Outcome data from this study are comparable to those of the ARDSnet Trial. The control group in the MOAT study was not ventilated with tidal volumes as small as those used in the experimental group of the ARDSnet trial (6–10 vs. 6.2 mL kg 1), but they were generally smaller than the ARDSnet control group (11.8 mL kg 1). Mortality at 30 days in the MOAT Study was not quite as good as that in the ARDSnet Trial (37 vs. 31%, respectively), but sepsis was much more prevalent in the MOAT Study compared to the ARDSnet Trial (47 vs. 27%, respectively).
STATUS OF HFV, RISKS, AND OUTLOOK FOR THE FUTURE
Are HFVs Safe and Effective?
Use of HFVs for newborn infants and adults began in the early 1980s. Fifteen randomized controlled trials with infants and about one-half that many randomized studies with children and adults were conducted over the next 20þ years. Over 1000 articles about HFV have been published. Yet, there are still questions about HFV safety and efficacy.
There are certainly adequate data to suggest that HFVs are effective in lessening chronic lung injury. The fact that not all studies have been successful in this regard is a reflection of differences in infant populations, ventilator strategies, and devices used. There is little argument that use of antenatal steroids, exogenous surfactant, and ventilator strategies using smaller tidal volumes have greatly improved mortality and morbidity of premature infants.
Not surprisingly, as clinicians have become more successful with HFV and other small tidal volume strategies, the age of viability of premature infants has gone down. Thus, the challenge of preventing chronic lung disease in NICU patients never gets easier, because the patients keep getting more premature.
What Are the Risks Associated with HFV in the NICU?
The greatest controversy in consideration of HFVs as a primary mode of ventilation of premature infants is safety, particularly whether HFV use increases the risk of cerebral injury. Clark et al. evaluated the probability of risk of premature infants suffering from intraventricular hemorrhage (IVH) or periventricular leukomalacia (PVL) by conducting a meta-analysis of all prospective randomized controlled trials of HFV published by 1996
HIGH FREQUENCY VENTILATION |
509 |
(63). The meta-analysis showed that use of HFV was associated with an increased risk of PVL (odds ratio ¼ 1.7 1.7 with a confidence interval of 1.06–2.74), but not IVH or severe ( grade 3) IVH. In addition, since the largest study in the group by far was the HiFi Trial (14), where implementation strategy was reputed to be less than optimal, they repeated the analysis without that study. When the results of the HIFI study were excluded, there were no differences between HFV and conventional ventilation in the occurrence of IVH or PVL.
Since 1996, seven additional randomized controlled trials of early use of HFV have been conducted on 1726 patients. Only one of the newer studies demonstrated a possible increased risk of cerebral injury (64), and that study included 273 patients or 16% of the total in these 7 studies. Thus, a more current meta-analysis would be even more convincingly positive today, and one could even say that there is little evidence of increased risk of cerebral injury during HFV. Why then, is this matter still controversial?
The risk of causing cerebral injury in premature infants is associated with hyperventilation and hypocarbia as noted earlier. There will never be a randomized controlled trial to prove cause and effect here, for obvious reasons. Therefore, all we can do is try to avoid hyperventilation and hypocarbia and see if outcomes get better over time.
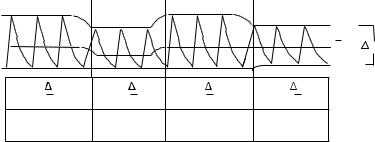
Avoiding hyperventilation and hypoxemia first requires proper monitoring. Pulse oximetry, transcutaneous CO2 monitoring, and continuous or frequent arterial blood gas monitoring are essential during HFV. Control of PaCO2 during HFV often requires optimization of PEEP, Paw, and pressure amplitude (DP) as shown in Fig. 12. The HFVs are noted for their ease of blowing off CO2 at lower airway pressures compared to CV, so PEEP and Paw must often be increased above those used during CV, if hypoxemia is to be avoided.
With HFOV, one often adjusts mean airway pressure without knowing the resulting baseline pressure or PEEP, whereas with HFJV, PEEP is adjusted to get an appropriate Paw. Therefore, one must not be fearful of higher PEEP when higher mean airway pressure is required. PEEP as high as 10 cm H2O is not unusual when HFJV is being used to treat premature infants.
One must also recognize that raising PEEP will reduce DP when PIP is held constant, as shown in Fig. 12, which causes both PaO2 and PaCO2 to rise.
Other safety concerns with early use of HFVs for preventing lung injury are interference with cardiac output by using too much PEEP or Paw. Since interference with
|
|
|
|
PIP |
|
|
|
|
|
|
Paw P |
|
|
|
|
|
|
PEEP |
|
|
Higher P |
Lower P |
Higher P |
Lower P |
|
|
|
Higher Paw |
Lower Paw |
Higher Paw |
Higher Paw |
Figure 12. Adjusting pressure waveforms to correct |
||
Hypocapnia |
Good PaCO2 |
Hypocapnia |
Good PaCO2 |
|||
arterial blood gases. (Used with permission. # |
2003, |
|||||
Good PaO2 |
Hypoxemia |
Good PaO2 |
Good PaO2 |
|||
Bunnell Inc.) |
|
|||||
|
|
|
|
|
||

510 HIGH FREQUENCY VENTILATION
venous return by elevated intrathoracic pressure raises intracranial pressure, there is associated fear of causing IVH by this mechanism as well.
A related issue, for those HFVs with that capability, is using too many CV breaths or using overly large CV tidal volumes during HFV. The latter use increases the risk of causing lung injury when HFV is implemented with higher PEEP.
Optimizing PEEP and minimizing the risk of using too many CV breaths during HFV can be achieved at the same time. The following flowchart for finding optimal PEEP during HFJV illustrates this point (Fig. 13).
The flowchart in Fig. 13 is based on the concept that CV breaths will be most effective in opening up collapsed alveoli, while PEEP or baseline pressure will prevent alveoli from collapsing during exhalation. The longer I times and tidal volumes of CV breaths provide a greater opportunity to reach the critical opening pressure of collapsed alveoli, and if PEEP is set above the critical closing pressure of those alveoli, they will remain open throughout the ventilatory cycle. Once PEEP is optimized, there is less value in using CV in tandem with HFV.
Although Fig. 13 was designed for use during HFJV, its principles are equally applicable to HFOV when CV may not be available. In this case, mean airway pressure is raised until an improvement in oxygenation makes it apparent that alveolar recruitment has occurred. At that point, it should be possible to decrease mean airway pressure somewhat without compromising oxygenation. However, the appropriate strategy here would be to set a goal for lowering the fraction of inhaled oxygen (FIO2) before
attempting to lower Paw. In this way, one should avoid inadvertently weaning Paw too fast and risking catastrophic collapse of alveoli. An appropriate FIO2 goal in this circumstance might be 0.3–0.4 depending on the vulnerability of the patient to high airway pressures and the magnitude of the mean airway pressure present at the time.
The final risk to be mentioned here will be the greatest risk associated with HFV: inadequate humidification and airway damage. In unsuccessful applications of HFV in infants in the early 1980s, necrotizing tracheal bronchitis (NTB) was frequently noted at autopsy (65). First discovered during HFJV, it was subsequently discovered during HFOV as well (66). Fortunately, the development of better humidification systems coupled with earlier implementation of HFV seems to have eradicated this problem as an extraordinary adverse side effect. None of the 14 randomized controlled trials has found an increase in NTB associated with HFV treatment.
Humidification during HFV is challenging, especially for HFOVs that use high gas flow rates and HFJVs. Gas humidified under pressure will not hold as much water as unpressurized gas, so HFJVs must humidify their inspiratory gas at higher than normal temperatures in order to reach anything near 100% relative humidity at body temperature.
Bunnell Incorporated’s HFJV addressed this inherent problem by minimizing the driving pressure behind their jet nozzle using an inspiratory pinch valve in a little box placed near the patient’s head. The pinch valve reduces the pressure drop through that part of the system because of
Starting Assumptions:
1.Patient is on HFJV + CV with 5 – 10 IMV bpm, PEEP < 8 cm H2O.
2.Patient is being monitored with a pulse oximetry.
Figure 13. Optimal PEEP flowchart. (Used with permission. # 2005, Bunnell Inc.) (Warnings: Lowering PEEP may improve SaO2 in some cases. Optimal PEEP may be lower in patients with active air leaks or hemodynamic problems. Do not be shocked if optimal PEEP ¼ 8 12 cm H2O. Using IMV PIP with high PEEP is hazardous. Do not assume high PEEP causes overexpansion.)
 Switch CV to CPAP mode.
Switch CV to CPAP mode.
Does SaO2 drop? |
NO |
(Wait 1- 5 min.) |
|
YES
PEEP is too low.
Switch back to IMV.
Increase PEEP by 1 – 2.
Wait for SaO2 to return to acceptable value.
PEEP is high enough, for the moment.
Hours later…
Does FIO2 |
NO |
need to be increased? |
|
YES
Increase PEEP by 1 – 2. Add IMV at 3-5 bpm as necessary until FIO2 can be reduced, then go back to CPAP mode.
Keep PEEP at this level
until FiO2 < 0.30
the relatively large internal diameter (ID) of the tubing in the valve (0.13 in., 3.2 mm). Placing the pinch valve within 35 cm of the patient where inspired gas is delivered via a jet nozzle embedded in a special ET tube adapter also enables the LifePulse Ventilator to deliver its tiny tidal volumes without much driving pressure. (A typical driving pressure needed for HFJV with the LifePulse on a premature infant is between 1.5 and 3.5 psi.) The HFVs that work at higher pressures and gas flow rates sometimes provide humidity with liquid water. (See Acutronic jet ventilation systems on their website: www.acutronicmedical.ch.)
Working Within the Limitations of HFVs in the NICU
The HFVs have been widely accepted for treating newborn infants with lung injury. Whether HFV will ever be widely accepted as a primary mode of ventilation is another matter. There have been many advances in conventional ventilator therapy over the past several years and, to a certain extent, techniques used to optimize HFVs have been applied to CVs (67).
Like most therapies, the skill with which HFV is implemented is probably the most critical determinant of clinical success. Starting HFV on patients sooner rather than waiting for worsening lung disease to become nearly hopeless is also extraordinarily important. Having written protocols defining the patient population and how HFV is optimized for that patient population can also be very helpful.
Early-to-moderately early stage treatment of homogeneously noncompliant lungs is the most obvious choice as an appropriate indication for HFV. If hyperventilation and gas trapping are avoided and appropriate resting lung volume is achieved, better outcomes should result. The ability of all HFVs to ventilate lungs using less pressure, independent of lung compliance, is nearly universal as long as the lungs are not too large for the ventilator’s output. Many of the HFV modes built into CVs are not powerful enough to ventilate even a term infant, so operators need to know the relative output capacities of their HFVs.
Using HFVs as rescue ventilators usually means that the underlying lung disorder has become nonhomogeneous or even obstructive. Ironically, most clinicians will only use HFVs as rescue ventilators even though these disorders are much harder to treat with HFV. Gas trapping is a much greater risk with heterogeneous obstructive disorders, and it may be avoided via use of small HFV tidal volumes. However, even HFV tidal volumes can be trapped if expiratory times are not several times greater than inspiratory times. The HFJVs with their very short I:E ratios and very high velocity inspiratory flows have been demonstrated to be more effective with these types of lung disease. Combined CV and HFJV have also been shown to work even better than pure HFJV in severe nonhomogeneous lung disorders (68).
What Are the Risks and Limitations Associated with HFV in Treating Children and Adults?
The only risks unique to HFV for children and adults are those associated with the necessity for delivering larger
HIGH FREQUENCY VENTILATION |
511 |
tidal volumes at higher flow rates compared to HFV for infants. Humidification of HFV gases is crucial as was learned with the early trials in infants, and gas is more difficult to humidify under pressure, as discussed above. The most challenging mode of HFV in this respect is HFJV, since it takes elevated pressure to push gas through a jet nozzle. Perhaps this is one reason for the lack of success of the only HFJV for adults approved by the FDA for use with adults (the APT 1010 Ultrahigh Frequency Ventilator, developed by the Advanced Pulmonary Technologies, Inc., Glastonbury, CT). Humidification with this device was only provided via supersaturated entrained gas. However, the same corporation that pulled this product off the market is also planning to discontinue manufacturing the Infant Star HFV for infants, and that ventilator had no such humidification issues. Thus, it appears likely that these products were discontinued for other (i.e., business) reasons.
There is also danger of operator error when any machine is used by untrained or unskilled operators. Given the tendency for some hospitals to only use HFVs as last resort rescue ventilators, one must consider those types of risks. Since HFOV is most successful in homogeneous lung disorders, it only makes sense for it to be used relatively early in the course of ARDS before significant lung injury results from CV treatment. Once the patient’s condition deteriorates into ARDS complicated by airleaks, chances for HFOV success may be significantly decreased.
Treating children and adults with HFVs also has to take optimal frequency into account. The primary determinant of optimal frequency is lung compliance, which is primarily determined by the patient’s size. An adult’s lung is larger than that of a child, which is larger than that of a term newborn, which is larger than that of a preemie. Thus, HFV frequency should be reduced as patient size increases. Optimal HFV for adults may occur at 150 bpm, whereas operation at that frequency with infants would not even be considered HFV.
Given the evidence that HFJVs have been more successful with nonhomogeneous lung disorders in infants, as described above, it would seem likely that HFJVs would find a role for treating adult patients with ARDS as well. Unfortunately, the application of HFJV for adults has been tarnished by a lack of success in very early studies.
Carlon et al. conducted a randomized controlled trial of HFJV versus volume-cycled CV on adults with acute respiratory failure (69). While they reported patients failing on CV improved more rapidly and in greater number when switched to HFJV compared to those who were failing on HFJV and crossed to CV, there were no advantages with respect to survival and total duration of stay in the ICU. Thus, they concluded that HFJV offered no obvious benefits over CV. The study was published in 1983, long before there was much appreciation of the need to optimize PEEP and maintain adequate lung volume during HFV. One wonders what results would come from a similar trial 20 years later, but such a trial will probably never happen now. The cost, time, and effort of seeking FDA approval for any Class III device is so high now, that HFJV may never find its way back into an adult ICU in the United States.
512 HIGH FREQUENCY VENTILATION
What Is the Outlook for HFV in the Future?
The HFVs evolved as people discovered problems with trying to replicate breathing in compromised patients. Unlike conventional ventilation, HFV is designed to facilitate gas exchange rather than mimic how people breathe normally. The differences between HFV and CV have led to creative solutions for many of the problems that investigators set out to solve, but they have also created their own problems.
It was discovered how HFVs can ventilate much more effectively than CV, and in the process, it was discovered how hypocapnia can lead to severe cerebral injury in premature infants.
The HFV still uses positive pressure where we create negative pressure to draw gas into the lungs. So, the problems related to use of ET tubes and its bypassing the normal humidification system of the body (i.e., the nose) are still there.
The HFV uses much smaller tidal volumes than CV, so the damage we have come to call volutrauma has lessened. In the process, it was discovered that more mean airway pressure or PEEP is required to keep sick lungs open while they are being ventilated. Then it was discovered that the new problems were associated with too much pressure in the thorax interfering with cardiac output.
So, HFVs do not solve all the problems, and they require increased vigilance to avoid creating new problems. However, the basic differences between HFV and CV provide a very reliable alternative to CV in circumstances where those differences are critical to survival.
The HFV tidal volumes are minimally affected by lung compliance and maximally affected by airway resistance when they are delivered via a jet nozzle. Therefore, in lung disorders where these conditions dictate treatment success or failure, wise users of HFVs have been very successful. When HFV users are not adequately trained or aware of the differences in HFV gas distribution caused by lung pathophysiology, success can be elusive.
Premature infants with RDS represent a large population with the potential of leading long and rich lives if they survive their first months of life with undamaged or even minimally damaged lungs and brains. Many randomized controlled trials have demonstrated the potential of HFVs to help these infants realize that potential.
The tens of thousands of adults who succumb to ARDS every year also have the potential of increased survival with less morbidity thanks to HFVs. These patients, when successfully treated with an HFV, will be considered rescued from a well-recognized disorder with a chronically high mortality rate.
Given the skill and training needed to master HFVs, their use may be considered risky indefinitely. As HFVs and associated monitoring equipment become better designed to help their users optimize assisted ventilation, HFV use should increase and evolve into earlier, more prophylactic applications to prevent lung injury. The HFVs are inherently a kinder, gentler form of mechanical ventilation. Hopefully, their true potential will someday be realized.
BIBLIOGRAPHY
Cited References
1.Lee PC, Helsmoortel CM, Cohn SM, Fink MP. Are low tidal volumes safe? Chest 1990;97:430–434.
2.Kacmarek RM, Chiche J.D. Lung protective ventilatory strategies for ARDS—the data are convincing! Resp Care 1998; 43:724–727.
3.Sjo¨strand U. Review of the physiological rationale for and development of high-frequency positive-pressure ventila- tion—HFPPV. Acta Anaesthesiol Scand (Suppl) 1977;67: 7– 27.
4.Klain M. Clinical applications of high-frequency jet ventilation, Part A: clinical use in the operating room. In: Carlon GC, Howland WS, editors. High-frequency ventilation in intensive care and during surgery. New York: Marcel Dekker; 1985. p 137–149.
5.Singh JM, Stewart TE. High-frequency mechanical ventilation principles and practices in the era of lung-protective ventilation strategies. Respir Care Clin N Am 2002;8:247–60.
6.MacIntyre NR. Setting the frequency-tidal volume pattern. Respir Care 2002;47:266–274.
7.Radford E. Ventilation standards for use in artificial respiration. J Appl Physiol 1955;7:451–460.
8.Henderson Y, Chillingworth FP, Whitney JL. The respiratory dead space. Am J Physiol 1915;38:1–19.
9.Dubois AB, Brody AW, Lewis DH, Burgess BF. Oscillation mechanics of lungs and chest in man. J Appl Physiol 1956;8: 587–594.
10.Bunnell JB, Karlson KH, Shannon DC. High-frequency positive pressure ventilation in dogs and rabbits. Am Rev Respir Dis 1978;117:289.
11.Fredberg JJ. Augmented diffusion in the airways can support pulmonary gas exchange. J Appl Physiol 1980;49:232–238.
12.Slutsky AS. Mechanisms affecting gas transport during highfrequency oscillation. Crit Care Med 1984;12:713–717.
13.Venegas JG, Hales CA, Strieder DJ. A general dimensionless equation of gas transport by high-frequency ventilation. J Appl Physiol 1986;60:1025–1030.
14.Permutt S, Mitzner W, Weinmann G. Model of gas transport during high-frequency ventilation. J Appl Physiol 1985;58: 1956–1970.
15.Venegas JG, Fredberg JJ. Understanding the pressure cost of high frequency ventilation: why does high-frequency ventilation work? Crit Care Med 1994;22:S49–S57.
16.Haselton FR, Scherer PW. Bronchial bifurcations and respiratory mass transport. Science 1980;208:69–71.
17.Tobias JD, Grueber RE. High-frequency jet ventilation using a helium-oxygen mixture. Paediatr Anaesth 1999;9:451–455.
18.Hatcher D, et al. Mechanical performance of clinically available, neonatal, high-frequency, oscillatory-type ventilators. Crit Care Med 1998;26:1081–1088.
19.Pillow JJ, Wilkinson MH, Neil HL, Ramsden CA. In vitro performance characteristics of high-frequency oscillatory ventilators. Resp Crit Care Med 2001;164:1019–1024.
20.Fredberg JJ, Glass GM, Boynton BR, Frantz 3rd ID. Factors influencing mechanical performance of neonatal highfrequency ventilators. J Appl Physiol 1987;62:2485–2490.
21.Boros SJ, et al. Comparison of high-frequency oscillatory ventilation and high-frequency jet ventilation in cats with normal lungs. Ped Pulmonol 1989;7:35–41.
22.Zobel G, Dacar D, Rodl S. Proximal and tracheal airway pressures during different modes of mechanical ventilation: An animal model study. Ped Pulmonol 1994;18:239–243.
23.Bryan AC, Slutsky AS. Lung volume during high frequency oscillation. Am Rev Resp Dis 1986;133:928–930.
24.Gerstmann DR, et al. Proximal, tracheal, and alveolar pressures during high-frequency oscillatory ventilation in a normal rabbit model. Pediatr Res 1990;28:367–373.
25.Perez Fontan JJ, Heldt GP, Gregory GA. Mean airway pressure and mean alveolar pressure during high-frequency jet ventilation in rabbits. J Appl Physiol 1986;61:456–463.
26.HIFI Study Group, HFOV compared with conventional mechanical ventilation in the treatment of respiratory failure in preterm infants. N Engl J Med 1989;320:88–93.
27.Keszler M, et al. Multicenter controlled clinical trial of highfrequency jet ventilation in preterm infants with uncomplicated respiratory distress syndrome. Pediatrics 1997;100:593–599.
28.Courtney SE, et al. Early high-frequency oscillatory ventilation versus conventional ventilation in very-low-birth- weight-infants. N Engl J Med 2002;347:643–653.
29.Johnson AH, et al. High-frequency oscillatory ventilation for the prevention of chronic lung disease of prematurity. N Engl J Med 2002;347:633–642.
30.Wiswell TE, et al. HFJV in the early management of RDS is associated with a greater risk for adverse outcomes. Pediatrics 1996;98:1035–1043.
31.Wiswell TE, et al. Effects of hypocarbia on the development of cystic periventricular leukomalacia in premature infants treated with HFJV. Pediatrics 1996;98:918–924.
32.Bryan AC, Froese AB. Reflections on the HiFi trial. Pediatrics 1991;87:565–567.
33.Stark AR. High-frequency oscillatory ventilation to prevent bronchopulmonary dysplasia—are we there yet? N Engl J Med 2002;347:682–683.
34.Keszler M, Goldberg LA, Wallace A. High frequency jet ventilation in subjects with low chest wall compliance. Pediatr Res 1993;33:331.
35.Keszler M, Jennings LL. High frequency jet ventilation in infants with decreased chest wall compliance. Pediatr Res 1997;41:257.
36.Fok TF, et al. High frequency oscillatory ventilation in infants with increased intra-abdominal pressure. Arch Dis Child 1997;76:F123–125.
37.Thibeault DW. Pulmonary barotrauma: Interstitial emphysema, pneumomediastinum, and pneumothorax. In: Thibeault DW, Gregory GA, editors. Neonatal Pulmonary Care. 2nd ed. New York : Appleton-Century-Crofts; 1986. p 499–517.
38.Macklin MT, Macklin CC. Malignant interstitial emphysema of the lung and mediastinum as an important occult complication in many respiratory diseases and other conditions: An interpretation of clinical literature in light of laboratory experiment. Medicine 1944;23:281.
39.Gonzalez F, Harris T, Richardson P. Decreased gas flow through pneumothoraces in neonates receiving highfrequency jet versus conventional ventilation. J Pediatr 1987;110:464–466.
40.Goldberg L, Marmon L, Keszler M. High-frequency jet ventilation decreases flow through tracheo-esophageal fistula. Crit Care Med 1992;20:547–550.
41.Donn SM, et al. Use of high-frequency jet ventilation in the management of congenital tracheoesophageal fistula associated with respiratory distress syndrome. J Pediatr Surg 1990;12:1219–1221.
42.Tobias JD, Grueber RE. High-frequency jet ventilation using a helium-oxygen mixture. Paediatr Anaesth 1999;9:451–455.
43.Gupta VK, Grayck EN, Cheifetz IM. Heliox administration during high-frequency jet ventilation augments carbon dioxide clearance. Respir Care 2004;49:1038–1044.
44.Clark RH, Yoder BA, Sell MS. Prospective, randomized comparison of high-frequency oscillation and conventional ventilation in candidates for extracorporeal membrane oxygenation. J Pediatr 1994;124:447–454.
HIGH FREQUENCY VENTILATION |
513 |
45.Engle WA, et al. Controlled prospective randomized comparison of HFJV and CV in neonates with respiratory failure and persistent pulmonary hypertension. J Perinat 1997;17: 3–9.
46.Baumgart S, et al. Diagnosis-related criteria in the consideration of ECMO in neonates previously treated with HFJV. Pediatrics 1992;89:491–494.
47.Paranka MS, Clark RH, Yoder BA, Null DM. Predictors of failure of high-frequency oscillatory ventilation in term infants with severe respiratory failure. Pediatrics 1995;95: 400–404.
48.Meliones JN, et al. High-frequency jet ventilation improves cardiac function after the Fontan procedure. Circulation 1991;84(Suppl III):III-364–III-368.
49.Kocis KC, et al. High-frequency jet ventilation for respiratory failure after congenital heart surgery. Circulation 1992; 86(Suppl II):II-127–II-132.
50.Greenspan JS, et al. HFJV: Intraoperative application in infants. Ped Pulmonol 1994;17:155–160.
51.Davis DA, et al. High-frequency jet versus conventional ventilation in infants undergoing Blalock-Taussig shunts. Ann Thorac Surg 1994;57:846–849.
52.Kinsella JP, et al. Randomized, multicenter trial of inhaled nitric oxide and high-frequency oscillatory ventilation in severe, persistent pulmonary hypertension of the newborn. J Pediatr 1997;131:55–62.
53.Day RW, Lynch JM, White KS, Ward RM. Acute response to inhaled nitric oxide in newborns with respiratory failure and pulmonary hypertension. Pediatrics 1996;98:698–705.
54.Platt D, Swanton D, Blackney D. Inhaled nitric oxide delivery with high-frequency jet ventilation. J Perinatol: (in press) 2003.
55.Mortimer TW, Math MCM, Rajardo CA. Inhaled nitric oxide delivery with high-frequency jet ventilation: A bench study. Respir Care 1996;41:895–902.
56.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000;342:1334–1349.
57.The Acute Respiratory Distress Syndrome Network, Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 2000;342:1301–1308.
58.Smith DW, et al. High-frequency jet ventilation in children with the adult respiratory distress syndrome complicated by pulmonary barotrauma. Ped Pulmonol 1993;15:279–286.
59.Fort P, et al. High-frequency oscillatory ventilation for adult respiratory distress syndrome—a pilot study. Crit Care Med 1997;25:937–47.
60.Arnold JH, et al. Prospective, randomized comparison of high-frequency oscillatory ventilation and conventional mechanical ventilation in pediatric respiratory failure. Crit Care Med 1994;22:1530–1539.
61.Mehta S, et al. Prospective trial of high-frequency oscillation in adults with acute respiratory distress syndrome. Crit Care Med 2001;29:1360–1369.
62.Derdak S, et al. High-frequency oscillatory ventilation for acute respiratory distress syndrome in adults: A randomized, controlled trial. Am J Respir Crit Care Med 2002;166:801– 808.
63.Clark RH, Dykes FD, Bachman TG, Ashurst JT. Intraventricular hemorrhage and high-frequency ventilation: A metaanalysis of prospective clinical trials. Pediatrics 1996;98: 1058–1061.
64.Moriette G, et al. Prospective randomized multicenter comparison of high-frequency oscillatory ventilation and conventional ventilation in preterm infants of less than 30 weeks with respiratory distress syndrome. Pediatrics 2001;107: 363–72.
