
- •Contents
- •Preface
- •How to use this book
- •Chapter 1 Units, constants, and conversions
- •1.1 Introduction
- •1.2 SI units
- •1.3 Physical constants
- •1.4 Converting between units
- •1.5 Dimensions
- •1.6 Miscellaneous
- •Chapter 2 Mathematics
- •2.1 Notation
- •2.2 Vectors and matrices
- •2.3 Series, summations, and progressions
- •2.5 Trigonometric and hyperbolic formulas
- •2.6 Mensuration
- •2.8 Integration
- •2.9 Special functions and polynomials
- •2.12 Laplace transforms
- •2.13 Probability and statistics
- •2.14 Numerical methods
- •Chapter 3 Dynamics and mechanics
- •3.1 Introduction
- •3.3 Gravitation
- •3.5 Rigid body dynamics
- •3.7 Generalised dynamics
- •3.8 Elasticity
- •Chapter 4 Quantum physics
- •4.1 Introduction
- •4.3 Wave mechanics
- •4.4 Hydrogenic atoms
- •4.5 Angular momentum
- •4.6 Perturbation theory
- •4.7 High energy and nuclear physics
- •Chapter 5 Thermodynamics
- •5.1 Introduction
- •5.2 Classical thermodynamics
- •5.3 Gas laws
- •5.5 Statistical thermodynamics
- •5.7 Radiation processes
- •Chapter 6 Solid state physics
- •6.1 Introduction
- •6.2 Periodic table
- •6.4 Lattice dynamics
- •6.5 Electrons in solids
- •Chapter 7 Electromagnetism
- •7.1 Introduction
- •7.4 Fields associated with media
- •7.5 Force, torque, and energy
- •7.6 LCR circuits
- •7.7 Transmission lines and waveguides
- •7.8 Waves in and out of media
- •7.9 Plasma physics
- •Chapter 8 Optics
- •8.1 Introduction
- •8.5 Geometrical optics
- •8.6 Polarisation
- •8.7 Coherence (scalar theory)
- •8.8 Line radiation
- •Chapter 9 Astrophysics
- •9.1 Introduction
- •9.3 Coordinate transformations (astronomical)
- •9.4 Observational astrophysics
- •9.5 Stellar evolution
- •9.6 Cosmology
- •Index

110 |
Thermodynamics |
|
|
5.3 Gas laws
Ideal gas
Joule’s law |
U = U(T ) |
(5.55) |
|||||
|
|
|
|
|
|
|
|
Boyle’s law |
pV |T = constant |
(5.56) |
|||||
|
|
|
|
|
|
|
|
Equation of state |
pV = nRT |
(5.57) |
|||||
(Ideal gas law) |
|||||||
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
pV γ = constant |
(5.58) |
|||||
Adiabatic |
T V (γ−1) = constant |
(5.59) |
|||||
T γp(1−γ) = constant |
|
||||||
equations |
(5.60) |
||||||
|
1 |
(p2V2 − p1V1) |
|
||||
|
∆W = |
|
|
(5.61) |
|||
|
γ − 1 |
||||||
Internal energy |
nRT |
(5.62) |
|||||
U = |
|
|
|||||
γ − 1 |
|||||||
Reversible |
|
|
|
|
|
|
|
isothermal |
∆Q = nRT ln(V2/V1) |
(5.63) |
|||||
expansion |
|
|
|
|
|
|
|
|
|
|
|||||
Joule expansiona |
∆S = nR ln(V2/V1) |
(5.64) |
|||||
|
|
|
|
|
|
|
|
Uinternal energy
Ttemperature
ppressure
Vvolume
nnumber of moles
Rmolar gas constant
γratio of heat capacities (Cp/CV )
∆W work done on system
∆Q heat supplied to system
1,2 initial and final states
∆S change in entropy of the system
aSince ∆Q = 0 for a Joule expansion, ∆S is due entirely to irreversibility. Because entropy is a function of state it has the same value as for the reversible isothermal expansion, where ∆S = ∆Q/T .
Virial expansion
|
|
|
|
|
B2(T ) |
|
|
p |
pressure |
|
|
pV = RT |
1 + |
|
|
V |
volume |
||||
|
|
|
|
|||||||
Virial expansion |
|
|
|
|
V |
(5.65) |
R |
molar gas constant |
||
|
|
|
|
B3(T ) |
|
T |
temperature |
|||
|
|
|
+ |
|
|
+ · · · |
|
|||
|
|
|
V 2 |
|
Bi |
virial coe cients |
||||
Boyle |
B2 |
(TB) = 0 |
|
|
|
|
|
(5.66) |
TB |
Boyle temperature |
temperature |
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|

5.3 Gas laws |
111 |
|
|
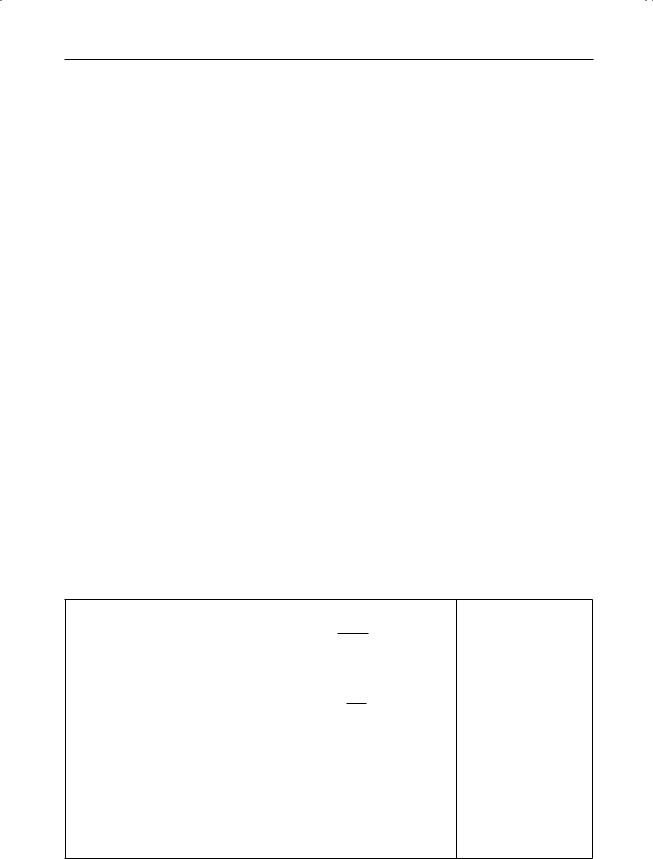
Van der Waals gas
|
|
|
|
|
|
|
p |
pressure |
|
|
|
|
a |
|
Vm |
molar volume |
|||
Equation of state |
|
|
|
R |
molar gas constant |
||||
|
|
|
|
|
|
||||
p+ Vm2 (Vm − b) = RT |
(5.67) |
||||||||
|
T |
temperature |
|||||||
|
|
|
|
|
|
|
a,b |
van der Waals’ constants |
|
|
|
|
|
|
|
|
|||
|
Tc = 8a/(27Rb) |
(5.68) |
Tc |
critical temperature |
|||||
|
pc = a/(27b2) |
|
|||||||
Critical point |
(5.69) |
pc |
critical pressure |
||||||
|
Vmc = 3b |
(5.70) |
Vmc |
critical molar volume |
|||||
|
|
|
|
|
|
|
pr |
= p/pc |
|
Reduced equation |
3 |
|
|
|
|||||
|
|
(5.71) |
Tr |
= T /Tc |
|||||
of state |
pr + Vr2 (3Vr − 1) = 8Tr |
||||||||
|
|
|
|
|
|
|
Vr |
= Vm/Vmc |
|
Dieterici gas
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5 |
|
|
|
|
|
|
|
|
|
|
|
|
p |
pressure |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
RT |
|
|
|
−a |
|
|
|
Vm |
molar volume |
|
|
Equation of state |
p = |
|
exp |
|
|
|
(5.72) |
R |
molar gas constant |
|
||||
Vm − b |
|
|
|
|||||||||||
|
|
|
|
RT Vm |
|
|
T |
temperature |
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
a ,b Dieterici’s constants |
|
|
|
Tc = a /(4Rb ) |
|
|
|
|
|
(5.73) |
Tc |
critical temperature |
|
||||
Critical point |
pc = a /(4b 2e2) |
|
|
|
|
|
(5.74) |
pc |
critical pressure |
|
||||
|
Vmc = 2b |
|
|
|
|
|
|
|
(5.75) |
Vmc |
critical molar volume |
|
||
|
|
|
|
|
|
|
|
e |
= 2.71828... |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Reduced equation |
|
|
Tr |
|
|
|
2 |
|
|
pr |
= p/pc |
|
||
pr = |
|
|
|
(5.76) |
Tr |
= T /Tc |
|
|||||||
of state |
2Vr − 1 exp 2 |
− VrTr |
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
Vr |
= Vm/Vmc |
|
|
|
Van der Waals gas |
|
|
2 |
||
|
1.4 |
|
|
|
|
|
|
|
1.1 |
Tr = 1.2 |
|
|
|
1.8 |
|
|
|
|
|
|
|||
|
1.2 |
1.0 |
|
|
|
|
1.6 |
|
1 |
|
|
|
|
1.4 |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
1.2 |
|
|
0.8 |
|
|
|
|
|
|
r |
|
|
|
|
r |
1 |
|
p |
|
0.9 |
|
|
|
p |
|
|
0.6 |
|
|
|
|
0.8 |
|
|
|
|
|
|
|
||
|
0.4 |
|
|
|
|
|
0.6 |
|
|
|
|
|
|
0.4 |
|
|
0.2 |
0.8 |
|
|
|
|
|
|
|
|
|
|
0.2 |
||
|
|
|
|
|
|
|
|
|
0 |
1 |
2 |
|
4 |
5 |
0 |
|
0 |
3 |
|
||||
|
|
|
Vr |
|
|
|
|
Dieterici gas
|
|
Tr = 1.2 |
|
|
|
|
1.1 |
|
|
|
|
|
1.0 |
|
|
|
|
|
0.9 |
|
|
|
|
|
0.8 |
|
|
|
|
0 |
1 |
2 |
3 |
4 |
5 |
|
|
Vr |
|
|
|
112 Thermodynamics
5.4 Kinetic theory Monatomic gas
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
p |
|
pressure |
|
|
|
|
1 |
|
|
|
|
|
2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
n |
|
number density = N/V |
||||
|
Pressure |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
p = 3 nm c |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.77) |
m |
2 |
particle mass |
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
c |
|
mean squared particle |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
velocity |
|
|
|
Equation of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
V |
|
volume |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
k |
|
Boltzmann constant |
|||
|
state of an ideal |
pV = NkT |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.78) |
|
||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
|
number of particles |
|||||||||||||
|
gas |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
T |
|
temperature |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
Internal energy |
3 |
|
|
|
|
|
|
N |
|
|
|
|
|
2 |
|
|
|
|
|
U |
|
internal energy |
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
U = 2 NkT |
= 2 m c |
|
|
|
|
|
(5.79) |
|
|||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
|
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.80) |
|
|
|
|
|
||||||
|
|
CV = |
|
|
Nk |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CV |
heat capacity, constant V |
||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5 |
|
|
|
|
|
|
|
|
|
||||||
|
Heat capacities |
Cp = CV + Nk = |
Nk |
|
|
|
|
(5.81) |
Cp |
heat capacity, constant p |
||||||||||||||||||||
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
|
2 |
|
|
|
|
||||||||||||||||||||||||
|
|
|
Cp |
|
|
5 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
γ |
|
ratio of heat capacities |
||||||
|
|
γ = |
= |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.82) |
|
|
|
|
|
|||||||
|
|
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
CV |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
Entropy |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
S |
|
entropy |
|
|
|
(Sackur– |
|
|
|
|
|
|
|
|
|
mkT |
|
|
3/2 |
|
V |
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
5/2 |
|
¯h |
|
= (Planck constant)/(2π) |
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
Tetrode a |
S = Nkln 2π¯h2 |
|
|
|
|
e |
N |
(5.83) |
|
||||||||||||||||||||
|
|
|
|
e |
|
= 2.71828... |
|
|
||||||||||||||||||||||
|
equation) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
a |
For the uncondensed gas. The factor |
|
mkT |
! |
3/2 |
is the quantum concentration of the particles, n |
|
. Their thermal de |
||||||||||||||||||||||
|
|
|
|
Q |
||||||||||||||||||||||||||
|
|
2π¯h2 |
|
|
|
|||||||||||||||||||||||||
|
|
|
|
|
1/3 |
|
|
|
|
|
|
|
|
|
||||||||||||||||
Broglie wavelength, λT , approximately equals nQ− |
|
|
|
. |
|
|
|
|
|
|
|
|
|
|||||||||||||||||
Maxwell–Boltzmann distributiona
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
pr |
probability density |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
3/2 |
|
|
|
|
|
|
|
2 |
|
m |
particle mass |
|||||
Particle speed |
pr(c) dc = |
|
|
m |
|
|
exp |
|
|
−mc |
|
4πc2 dc |
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
distribution |
|
|
|
|
|
|
2πkT |
! |
|
|
|
|
|
2kT |
|
|
k |
Boltzmann constant |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.84) |
T |
temperature |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
c |
|
particle speed |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Particle energy |
pr(E) dE = |
|
|
2E1/2 |
|
exp |
|
|
− |
E |
|
dE (5.85) |
E |
particle |
kinetic |
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
||||||||||||
distribution |
π1/2(kT )3/2 |
|
|
|
|
|
|
|
|||||||||||||||||||||
|
|
|
|
|
|
|
|
|
kT |
|
|
|
energy (= mc /2) |
||||||||||||||||
|
c = |
|
kT |
|
1/2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Mean speed |
8 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.86) |
c |
mean speed |
|
|||||||
πm |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
|
crms = |
kT |
|
1/2 |
= |
|
π |
|
1/2 |
|
|
|
|
c |
rms |
root mean squared |
|||||||||||||
rms speed |
3 |
|
|
|
|
3 |
|
|
|
c |
|
(5.87) |
|
speed |
|
|
|||||||||||||
m |
|
|
8 |
|
|
|
|
|
|
|
|||||||||||||||||||
Most probable |
cˆ = |
kT |
1/2 |
|
|
π |
1/2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
speed |
2 |
|
|
|
|
= |
|
|
! |
|
|
c |
|
|
|
|
|
(5.88) |
cˆ |
|
most probable speed |
||||||||
m |
|
|
|
4 |
|
|
|
|
|
|
|
|
|||||||||||||||||
aProbability density functions normalised so that %0∞ pr(x) dx = 1.

5.4 Kinetic theory |
113 |
|
|
Transport properties
Mean free patha |
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
l = |
√ |
|
πd2n |
|
|
|
|
|
|
|
|
|
|
|
|
(5.89) |
||||||||||||
2 |
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
Survival |
pr(x) = exp(−x/l) |
|
|
|
|
|
|
|
(5.90) |
|||||||||||||||||||
equationb |
|
|
|
|
|
|
|
|||||||||||||||||||||
Flux through a |
J = |
1 |
n c |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.91) |
||||||||||
planec |
4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Self-di usion |
J = −D n |
|
|
|
|
|
|
|
|
|
|
|
|
(5.92) |
||||||||||||||
(Fick’s law of |
|
|
2 |
|
|
|
|
|
|
|
|
|||||||||||||||||
where D |
l c |
|
|
|
|
|
|
|
|
|||||||||||||||||||
di usion)d |
|
|
|
|
|
|
|
(5.93) |
||||||||||||||||||||
|
|
|
|
|
|
|
|
|
||||||||||||||||||||
3 |
|
|
|
|
|
|
|
|||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
H = −λ T |
|
|
|
|
|
|
|
|
|
|
|
|
(5.94) |
||||||||||||||
Thermal |
2 |
|
|
|
|
|
|
|
1 ∂T |
|
|
|
|
|
|
|
|
|
|
|
||||||||
conductivityd |
|
|
T = |
D |
|
∂t |
|
|
|
|
|
|
5 |
|
|
|
(5.95) |
|||||||||||
|
for monatomic gas |
λ |
ρl c cV |
(5.96) |
||||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||
|
4 |
|||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Viscosityd |
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
η 2 ρl c |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.97) |
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
Brownian |
|
|
|
|
|
|
|
|
|
|
kT t |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
motion (of a |
x |
2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5.98) |
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
= 3πηa |
|
|
|
|
|
|
|
|
|
|
||||||||||||||
sphere) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free molecular |
|
|
|
|
|
|
|
|
|
|
|
|
|
3 |
|
|
|
|
|
|
|
1/2 |
|
|
|
|
|
|
|
M |
|
|
|
|
4R |
|
|
|
|
πm |
|
|
p1 |
|
p2 |
|
|||||||||||
flow (Knudsen |
|
d |
|
= |
p |
|
2 |
− |
||||||||||||||||||||
|
dt |
|
3L |
k |
T 1/2 |
T 1/2 |
||||||||||||||||||||||
flow)e |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
2 |
(5.99) |
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
lmean free path
dmolecular diameter
nparticle number density
pr probability
xlinear distance
Jmolecular flux
c mean molecular speed
Ddi usion coe cient
Hheat flux per unit area
λthermal conductivity
Ttemperature
ρdensity
cV |
specific heat capacity, V |
5 |
|
|
constant |
||
η |
dynamic viscosity |
||
|
xdisplacement of sphere in x direction after time t
kBoltzmann constant
ttime interval
asphere radius
dM mass flow rate dt
Rp pipe radius
Lpipe length
mparticle mass
ppressure
aFor a perfect gas of hard, spherical particles with a Maxwell–Boltzmann speed distribution. bProbability of travelling distance x without a collision.
cFrom the side where the number density is n, assuming an isotropic velocity distribution. Also known as “collision number.”
dSimplistic kinetic theory yields numerical coe cients of 1/3 for D, λ and η.
eThrough a pipe from end 1 to end 2, assuming Rp l (i.e., at very low pressure).
Gas equipartition
Classical |
a |
Eq = 1 kT |
|
|
|
|
|
|
(5.100) |
Eq |
energy per quadratic degree of |
||||||||
|
|
|
|
|
|
|
freedom |
||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
equipartition |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
k |
Boltzmann constant |
|
|
|
2 |
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
T |
temperature |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CV |
heat capacity, V constant |
|
|
|
|
|
|
|
1 |
|
|
|
1 |
|
|
||||||
|
|
CV = |
fNk = |
|
fnR |
(5.101) |
Cp |
heat capacity, p constant |
|||||||||||
|
|
|
|
2 |
|||||||||||||||
|
|
|
|
2 |
|
|
|
|
|
|
|||||||||
Ideal gas heat |
|
|
|
|
|
|
|
|
|
|
f |
|
|
N |
number of molecules |
||||
C |
|
= Nk 1 + |
|
(5.102) |
f |
number of degrees of freedom |
|||||||||||||
|
|
|
|
|
|
|
|||||||||||||
capacities |
|
|
p |
|
|
|
|
|
|
|
|
2 |
|
n |
number of moles |
||||
|
|
|
|
|
Cp |
|
2 |
|
|
|
(5.103) |
R |
molar gas constant |
||||||
|
|
γ = |
|
|
|
|
= 1 + |
|
|
|
|
|
|
||||||
|
|
CV |
f |
|
|
|
γ |
ratio of heat capacities |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
aSystem in thermal equilibrium at temperature T .
