
- •Англійська мова для професійного спілкування
- •Передмова
- •Brief contents
- •Unit 1 structure and bonding
- •1. You are going to read three texts which are all connected with chemistry. Read the texts and be able to make intelligent guesses about:
- •2. Decide what books the texts come from. What helped you to make up your mind? Choose from the following:
- •3. Which sentence could be the opening sentence of the text?
- •4. Think about the first sentences above and decide which you think are likely to introduce a paragraph with:
- •6. Give the definitions of the following terms:
- •2. Look at Appendix 3 and Render the following text.
- •3. Read the following text. Discuss the point with your colleagues. What do you know about the methods of scientific investigation? The Scientific Method
- •The Scientific Method
- •1. Culture clips: London life
- •2.What museums are there in your city/town? Have you ever visited any?
- •3.Have you ever visited science museum of the “kpi”? Are there any in your university? Imagine that you are a guide at such museum, tell about the most interesting museum piece.
- •2. What was said in the text about:
- •3. Render the following text.
- •1. Imagine that you are starting a presentation. What phrases might you use?
- •2. Listen totwowaysofopeningpresentationsandseeifyoucanhearsomeofthephrasesabove.
- •3. Read some advices on delivering effective presentations in the Appendix 7 and write your own opening for the topic “Stereochemistry”.
- •Imagine that you are a major distributor of the following product. Look at Business English section and write a letter asking more information about the product presented below.
- •Unit 3 molecular symetry
- •2. Find five things in the texts to finish the sentence: “It reminds me of…”
- •2. Read the flowcharts given in the figure 1 and 2.
- •3. Read some information about creation of the flow charts in the Appendix 4-6 and create your own describing any experiment you made in the laboratory.
- •4. Create a list of rules related to the theme of the text given in the exercise 1. Share and compare the rules with your partners and think how they might be improved, choose the best ones.
- •5. Render the text given in the exercise 1.
- •2. Listen to two ways of giving presentations and see if you can hear some of the phrases above.
- •3. Read some advices on delivering effective presentations in the Appendix 7 and write your own presentation for the topic “Molecular symmetry”.
- •You ordered: Beckman du64 uv/VisSpectrophotometer
- •Unit 4 stereochemistry of reactions
- •Chiral Drug
- •1.Presentation: questions.
- •Unit 5 resolution of enantiomers
- •Resolution of enantiomers
- •1. Method of resolution is the title of the text in this section. What is the likely content of the article? Predict the methods which might be described.
- •3. Mark and talk about five things from the text you are glad to find out about. Talk in pairs about these things and why you chose them.
- •5.Render the text.
- •4. Think of three reasons you liked the text and three reasons you didn’t like it. Share and compare your reasons with other students. Find out how many other students share your opinion.
- •1.Presentation: useful tips.
- •3.Complete the sentence with the correct phrase.
- •Principles of Stereochemistry
- •Enantiomeric Relationships
- •Diastereomeric Relationships
- •Methods of determining configuration
- •The Cause of Optical Activity
- •Molecules With More Than One Chiral (Stereogenic) Center
- •Asymmetric Synthesis
- •Business english
- •Formal letter
- •1.Titles and addresses
- •2Covering the issues
- •3 Beginning your letter
- •4 Ordering ideas
- •5 Range
- •6 Ending the letter
- •Sample formal letter
- •Informal letter or email
- •1 Titles and addresses
- •2 Openings
- •3 Covering all the issues
- •4 Using informal language
- •5 Range
- •6 Connectors
- •7 Closing statements
- •Writing a tactful advice letter
- •How to write a request letter
- •Complaint letter
- •If necessary, add any further information:
- •Writing claim letter
- •Inquiry letter
- •Establish Your Objective
- •Determine Your Scope
- •Organize Your Letter
- •Draft Your Letter
- •Close Your Letter
- •Review and Revise Your Inquiry Letter
- •Sample Inquiry Letter __________Better Widget Makers, Inc.__________
- •5555 Widget Avenue
- •Appendices appendix 1 exclamations
- •Appendix 2 general conversation gambits
- •Appendix 3 the scheme of rendering the text
- •Appendix 4 flow charts
- •Appendix 5 graph
- •Appendix 6 reading and interpreting graphs
- •Types of Graphs
- •Appendix 7 presentations
- •Typescripts
- •Bbc Learning English. Talking Business
- •(Bbclearningenglish. Com)
- •Bibliography 1
- •Bibliography 2
1. Culture clips: London life
Welcome to London Life. This time, we find out about London's science museum. It is one of London's most popular attractions. During the programme, we look at some useful science vocabulary. Write down at least 5 questions that
you think or hope will be answered by the programme.As you listen, try to answer the following questions:
1: What kinds of things are children most interested in?
2: What is William's favorite thing at the museum?
2.What museums are there in your city/town? Have you ever visited any?
3.Have you ever visited science museum of the “kpi”? Are there any in your university? Imagine that you are a guide at such museum, tell about the most interesting museum piece.
Writing
Imagine that you are Margarita. This is a part of the letter that your friend has sent you. Read it, then look at Business English section and write him/her a letter giving your advice. Use the following useful expressions and plan.
Dear Margarita,
I’ve just found out that I’ve failed to pass organic chemistry. I haven’t told my parents yet because I’m too scared. The most difficult for me is the topic “Bonding and structure”. I’ll have to retake the exams, but I know I’ll fail again! What can I do? Please help me!
Useful expressions:
Start with: I just got your letter and I think I can help you. / I was sorry to hear about your problem.
Giving advice: If I were you…, You should…, Why don’t you…, It would be a good idea to…, The best thing you can do is…, I strongly advise you to…
Finish with: I hope this helps you./ Let me know what happens./ Hope this advice is of some help to you./ Things will get better soon.
Plan
Introduction
Dear (the persons first name)
Paragraph 1: express sympathy
Main body
Paragraph 2: give your advice (tell parents…)
Paragraph 3: give your advice how to master the topic
Conclusion
Paragraph 4: end the letter offering some encouragement
Good luck
Margarita
UNIT 2
PRINCIPLES, NOMENCLATURE & SYMBOLS
Reading
Section A
1. Scan the text, find and read the definitions of the following terms:
Stereochemistry, stereoisomer, enantiomer , chiral, achiral, stereogenic center.
***
The Greek word stereos means “solid,” and stereochemistry refers to chemistry in three dimensions. The foundations of organic stereochemistry were laid by Jacobusvan’t Hoff and Joseph Achille Le Bel in 1874. Independently of each other, van’t Hoff and Le Bel proposed that the four bonds to carbon were directed toward the corners of a tetrahedron. One consequence of a tetrahedral arrangement of bonds to carbon is that two compounds may be different because the arrangement of their atoms in space is different. Isomers that have the same constitution but differ in the spatial arrangement of their atoms are called stereoisomers. We have already had considerable experience with certain types of stereoisomers—those involving cis and trans substitution patterns in alkenes and in cycloalkanes.
****
Everything has a mirror image, but not all things are superposable on their mirror images. Mirror-image superposability characterizes many objects we use every day. Cups and saucers, forks and spoons, chairs and beds are all identical with their mirror images. Many other objects though—and this is the more interesting case—are not. Your left hand and your right hand, for example, are mirror images of each other but can’t be made to coincide point for point, palm to palm, knuckle to knuckle, in three dimensions. In 1894, William van’t Hoff was the recipient of the first Nobel Prize in chemistry in 1901 for his work in chemical dynamics and osmotic pressure—two topics far removed from stereochemistry.
Our major objectives in this chapter are to develop a feeling for molecules as three-dimensional objects and to become familiar with stereochemical principles, terms, and notation. A full understanding of organic and biological chemistry requires an awareness of the spatial requirements for interactions between molecules; this chapter provides the basis for that understanding.
Thomson (Lord Kelvin) coined a word for this property. He defined an object as chiral if it is not superposable on its mirror image. Applying Thomson’s term to chemistry, we say that a molecule is chiral if its two mirror-image forms are not superposable in three dimensions. The work “chiral” is derived from the Greek word “cheir”, meaning “hand”, and it essentially appropriate to speak of the “handedness” of molecules. The opposite of chiral isachiral. Amolecule that is superposable on its mirror image is achiral.
In organic chemistry, chirality most often occurs in molecules that contain a carbon that is attached to four different groups. An example is bromochlorofluoromethane (BrClFCH).
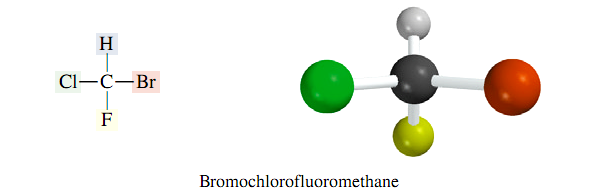
The two mirror images of bromochlorofluoromethane cannot be superposed on each other. Since the two mirror images of bromochlorofluoromethane are not superposable, BrClFCH is chiral. The two mirror images of bromochlorofluoromethane have the same constitution. That is, the atoms are connected in the same order. But they differ in the arrangement of their atoms in space; they are stereoisomers. Stereoisomers that are related as an object and its nonsuperposable mirror image are classified as enantiomers. The word “enantiomer” describes a particular relationship between two objects. One cannot look at a single molecule in isolation and ask if it is an enantiomer any more than one can look at an individual human being and ask, “Is that person a cousin?” Furthermore, just as an object has one, and only one, mirror image, a chiral molecule can have one, and only one, enantiomer.
The two enantiomers of bromochlorofluoromethane are similarly oriented, that the difference between them corresponds to an interchange of the positions of bromine and chlorine. It will generally be true for species of the type C(w, x, y, z), where w, x, y, and z are different atoms or groups, that an exchange of two of them converts a structure to its enantiomer, but an exchange of three returns the original structure, albeit in a different orientation.
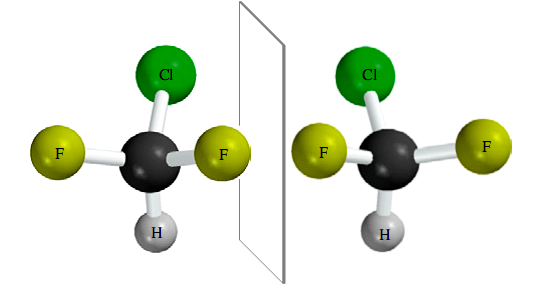
Consider next a molecule such as chlorodifluoromethane (ClF2CH), in which two of the atoms attached to carbon are the same. Since mirror-image representations of chlorodifluoromethane are superposable on each other, ClF2CH is achiral.
****
Molecules of the general type
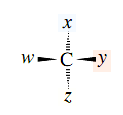
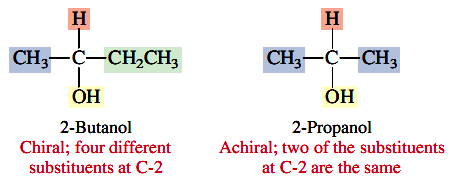
are chiral when w, x, y, and z are different substituents. A tetrahedral carbon atom that bears four different substituents is variously referred to as a chiral center, a chiral carbon atom, an asymmetric center, or an asymmetric carbon atom. A more modern term is stereogenic center, and that is the term that we’ll use. (Stereocenter is synonymouswith stereogenic center.) Noting the presence of one (but not more than one) stereogenic center in a molecule is a simple, rapid way to determine that it is chiral. For example, C-2 is a stereogenic center in 2-butanol; it bears a hydrogen atom and methyl, ethyl, and hydroxyl groups as its four different substituents. By way of contrast, none of the carbon atoms bear four different groups in the achiral alcohol 2-propanol.
Molecules with stereogenic centers are very common, both as naturally occurring substances and as the products of chemical synthesis. (Carbons that are part of a double bond or a triple bond can’t be stereogenic centers.)
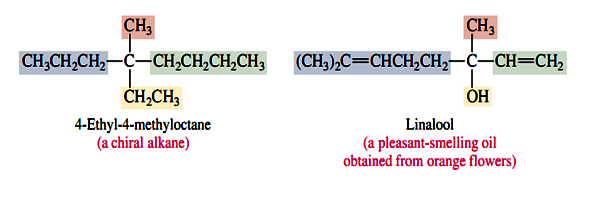
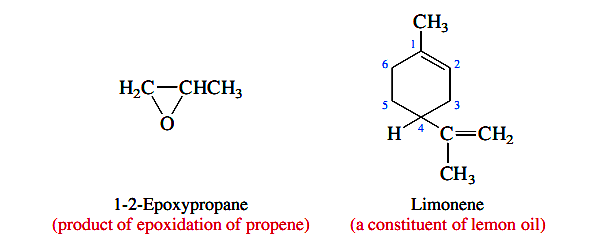
A carbon atom in a ring can be a stereogenic center if it bears two different substituents and the path traced around the ring from that carbon in one direction is different from that traced in the other. The carbon atom that bears the methyl group in 1,2-epoxypropane, for example, is a stereogenic center. The sequence of groups is O±CH2 as one proceeds clockwise around the ring from that atom, but is CH2±O in the anti-clockwise direction. Similarly, C-4 is a stereogenic center in limonene.
Even isotopes qualify as different substituents at a stereogenic center. The stereochemistry of biological oxidation of a derivative of ethane that is chiral because of deuterium (D = 2H) and tritium (T= 3H) atoms at carbon, has been studied and shown to proceed as follows:
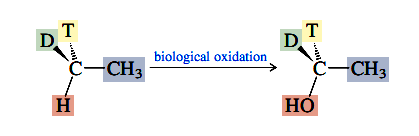
The stereochemical relationship between the reactant and the product, revealed by the isotopic labeling, shows that oxygen becomes bonded to carbon on the same side from which H is lost.
One final, very important point is about stereogenic centers. Everything we have said in this section concerns molecules that have one and only one stereogenic center; molecules with more than one stereogenic center may or may not be chiral.
