
- •I. Lead-in
- •II. Vocabulary
- •III. Reading
- •IV. Speaking
- •V. Reading
- •VI. Listening
- •VIII. Writing
- •VII. Extra activities
- •IX. Grammar: Present Tenses
- •Lead-in
- •II. Reading
- •III. Speaking
- •IV. Reading
- •V. Listening
- •VI. Extra activities
- •VII. Write 10 equations and ask your partner to read them.
- •VIII. Project work
- •IX. Grammar: Past Tenses
- •I. Lead –in
- •II. Reading
- •III.Speaking
- •IV. Listening
- •V. Extra activities
- •VI. Speaking
- •VII. Grammar: will/going to
- •II. Reading
- •Read the text and check the pronunciation of the new words and elements in the dictionary.
- •Vocabulary
- •IV. Reading
- •Scan the text, find all the elements mentioned there. Then skim the text and say in what context they are used.
- •V. Writing
- •VI. Listening
- •Now listen to a “Periodic Table” song and check if you were right.
- •Iron is the 26th then cobalt, nickel ………you get
- •VII. Extra activities
- •Check the pronunciation of the elements in a dictionary or in appendix and read them aloud:
- •VIII. Grammar. Passive Voice
- •The Law of Conservation of Mass
- •I. Lead-in
- •II. Reading
- •The Law of Conservation of Mass
- •III. Speaking
- •IV. Reading
- •Read the text and check the new words in the dictionary.
- •Put the events into chronological order according to the text.
- •Find the synonyms to the words in bold from the text.
- •V. Speaking. Discuss with your partner which you think are the most important scientific discoveries of the past. Talk about:
- •VI. Video
- •Before watching the video match the words with their definitions.
- •Watch the video and complete the sentences using the words from ex. VI a.
- •VII. Translate into English.
- •VIII. Grammar: Participles
- •IX. Write a short essay to answer the question: “What are the main differences between the phlogiston theory and the Law of Conservation of mass”? Use these notes to write four paragraphs.
- •II. Reading
- •Read the text and choose the correct answer/answers and find the proof in the text.
- •VI. Reading
- •VII. Speaking /Project work
- •VIII. Video
- •Match the following words and word combinations with their Russian equivalents
- •IX. Writing
- •X. Grammar. Gerund
- •I. Lead- in
- •Reading
- •States of matter
- •IV. Translate into English.
- •V. Speaking
- •VI. Reading
- •Measuring matter: mass, weight, and volume
- •Volume V
- •Match the words and their definitions.
- •Measuring matter crossword
- •Fill in the table.
- •VIII. Listening
- •XI. Writing
- •X. Grammar. Infinitive
- •II. Reading
- •IV. Insert the missing prepositions into the blanks.
- •V. Speaking
- •VI. Reading
- •Answer the following questions.
- •VII. Fill in the blanks with the words and expressions listed:
- •Give the English equivalents for the following.
- •IX. Listening
- •X. Writing
- •XI. Grammar: Modal Verbs
- •Reading
- •Read the text and check if your definition is correct.
- •Speaking
- •Before you read
- •Reading
- •Comprehension
- •Read the text again and decide if the following statements are true or false.
- •Find synonyms for the following words in the text.
- •Speaking
- •Discuss these questions with your partner.
- •Do you agree with the following quotes? Why? Why not? Try to explain what Albert Einstein meant by saying them.
- •Writing
- •X. Extra activities
- •XI. Grammar: Reported speech.
- •Lead-in
- •Reading
- •Comprehension
- •Reading
- •Speaking
- •IX. Writing
- •Grammar: Conditionals
- •Lead-in
- •II. Reading
- •Read the text and check if your answers were correct.
- •Read the text thoroughly with a dictionary and answer the following questions.
- •Find in the text English equivalents to the given Russian words, word combinations and chemical terms.
- •III. Reading
- •Read the text and divide it into logical parts and entitle them.
- •Think of synonyms for the words in bold.
- •IV. Video
- •Look through the words before watching the video:
- •Now watch the video “Organic molecules” and find more information about carbon. Combine the information from the text and the video and tell the class about carbon.
- •Speaking
- •Translate the following sentences into English.
- •Match the names of organic chemistry reactions with their descriptions.
- •VIII. Grammar: Questions
- •IX. Choose one of the topics below and write a report (150-200 words): Properties of Carbon, Carbon bonds, Hydrocarbons and their isomers, Derivatives of hydrocarbons, Types of polymers.
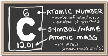
- •I. Elements (symbol, atomic number, English pronunciation).
- •II. How to read chemical formulas in inorganic chemistry.
- •III. How to read chemical formulas in organic chemistry.
- •IV. Notes on reading chemical formulas.
- •V. Some abbreviations in common use in chemistry.
-
Find in the text English equivalents to the given Russian words, word combinations and chemical terms.
nouns: исключение, образование, правило, причина, сырье
verbs: означать, рассматривать(считать), узнать, преобразовывать, соединять(ся), разделить, производить
adjectives and prepositions: внутри, некоторым образом, просто, непосредственно, слишком много
word combinations: нельзя не удивиться, можно сказать, в настоящее время
chemical terms: сахар, этиловый спирт, метан, мочевина, углерод, цепочка атомов, углеводород
-
Fill in the gaps in the sentences with the following words: organic, living, branch, cleavage, carbon, organism, properties, chain.
-
Organic chemistry is the………….of chemistry dealing with compounds containing carbon-carbon bonds.
-
Every living…………., irrespective of plants and animals are composed of organic compounds.
-
…………..chemistry reactions are different from inorganic chemistry reactions.
-
…………….can form a long chain due to its catenation ability.
-
The root word of organic chemistry, 'Organic' means that the compounds were synthesized from………… organisms in the past.
-
All the organic compounds with same functional group have same chemical……… which make the study of organic chemistry much easier.
-
The organic chemistry reactions involve the………. of the covalent bonds and forming of new bonds.
-
The physical properties may differ with the number and nature of carbon…........
III. Reading
Text B
-
Read the text and divide it into logical parts and entitle them.
Carbon
Carbon is to be ranked along with hydrogen and oxygen as one of the most important of all the elements to man. Carbon occurs in nature as a free element and in many compounds. It constitutes only about 0.03 percent of the Earth’s crust, but this relatively small amount of the 300,000 or more compounds of the element which exists naturally or which have been prepared. It is proved that this number is approximately ten times the number of compounds of all the elements put together. For a long time it was believed that these compounds might have never been produced except with the aid of organic life, in other words, by living plants and animals. For this reason they were called organic compounds. It is known that carbon occurs in two crystalline forms which differ strikingly by their properties. Graphite is black, soft, a good conductor of electricity. Diamond, on the contrary is colourless and transparent, the hardest of known substances, a non-conductor of electricity. It is the crystal structure, as determined by X-rays, which gives an explanation of this contrast of properties. The four valence electrons of each carbon atom enable it, by sharing electrons with four of its neighbours, to be linked with them in a covalent union. It may be shown by X-rays examination that in the diamond the four nearest neighbours of each carbon atom are symmetrically arranged about it in space. All atoms in a diamond are thus firmly linked together; hence the whole crystal acts as a giant molecule. Thus we account for the extreme hardness of the diamond, its high melting point, and its failure to dissolve in any solvent. On the other hand, it is found that graphite possesses parallel planes of atoms, and each is at a considerable distance from its neighbours. Each carbon atom in graphite has three nearest neighbours and they all are present in its own plane. Only three of the four valence electrons of each atom are needed for furnishing bonds with these nearest neighbours and the fourth is available for producing a bond with a neighbouring plane. A certain portion of the electrons in graphite are relatively free to move as it is true of metals. Hence, graphite is a conductor of electricity.
