Industrial downstream processing
might need to recover the soluble fraction as well as the insoluble fraction. In some, but not all instances fast processing and addition of components preventing precipitation or crystallisation can solve these problems. Substrate or substrate analogues are examples of such components.
The continuing developments within production organisms and fermentation technology [12] with higher product yield and cleaner background will in the years to come accelerate these process problems. Where in resent years the ability for an enzyme to crystallise was seen with great pleasure, in the coming years spontaneous enzyme precipitation and crystallisation will bring unpleasant challenges to the field.
5. Summary.
Pharmaceutical and bulk enzyme productions differ significantly in relation to process requirements and solutions.
Where pharmaceutical downstream processing is centred on chromatographic purification, bulk enzyme production is based on the techniques known from the chemical industries with flocculation, membrane concentration, adsorption, precipitation and crystallisation as the dominant technologies.
Pharmaceutical production benefits from the continuing research in refining chromatography based systems. Many well designed columns, matrixes and procedures therefore exist, making removal of host impurities a not too difficult matter. However, as sensitivity of impurity detection increases and focus increases for producing safe pharmaceuticals with little/controlled micro heterogeneity research and development within the high-resolution area will be of significant value. High-resolution chromatographic systems therefore still deserve focused research and development, as do new high-resolution technologies. Also focus on analytical tools, protein chemistry and on protein interactions with surfaces and soluble components will be of clear value.
For bulk enzymes low production cost, high capacity and environmental friendliness are very important parameters. As price competition within the field can be fierce, much refinement is going on to ensure efficient low cost production. However, a hard challenge enzyme producers is going to handle in the near future - besides general cost and waste reductions, is efficient processing of enzymes partially precipitated or crystallised in the fermentation broth. Research within enzyme solubility and crystallisation will therefore be of value for the enzyme field. Research and development within enclosed, sanitary and environmentally friendly technologies do also have significant value for the enzyme producer.
References.
1.Wenisch, E., Schneider P., Hansen S. A., Rezzonico R. and Righetti P. G. (1993) Isoelectric focusing in a multicompartment electrolyser with zwitterionic membranes, exemplified by purification of glucoamylase, Journal of Biochemical and Biophysical Methods 27, 199-213
2.Horvath Z. S., Corthals G. L., Wrigley C. W. and Margolis J (1994) Multifunctional apparatus for electrokinetic processing of proteins, Electrophoresis 15, 968-971
Mads Laustsen
3. Laustsen M. (1993) Methods and systems tor high molecular weight electrodialysis, U. S. Patent
Application 08/176,037
4 Nielsen N. (1993) Method for purification of an aqueous enzyme solution, WO 94/01537
5.Laustsen M. and Nielsson S. (1997) Selective Inactivation of Enzyme Activities, PCT/DK96/00489
6.Becker, Nathaniel, T.; Braunstein, Edit, L.; Gros, Ernst, H.; Fewkes, Robert; Heng, Meng, H. (1996) Crystalline cellulase and method for producing same, WO 97/15660
7.Gros, Ernst, H. and Cunefare, Lerry, L. (1997) Crystalline protease and method for producing same, WO
97/33983
8. Nilsson B. M., Laustsen M. and Pahle C. (1998) Separation of proteins EP 0691 982
9.Nilsson B. M., Laustsen M. and Rancke-Madsen A. (1998) Separation of proteins US 5,728,559
10.Lovrien R. and Matolis D. (1996) Hard and soft sulphate and sulfonate anions in protein precipitationbiorecognition 211t h ACS National Meeting New Orleans, LA
11. Becker N. T. and Anderson K. A. (1998) Recovery of proteins by precipitation using lignosulfonates,
WO 98/30580
12De Laat, Wilhelmus, Theodorus, Antonius, Maria; Preusting, Johannes, Cornelis, Gerardus; Koekman, Bertus, Pieter. (1998) Fermentative production of valuable compounds on an industrial scale using chemically defined media, WO 98/37179
SEPARATION OF 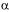 -LACTALBUMIN AND
-LACTALBUMIN AND 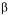 -LACTOGLOBULIN BY
-LACTOGLOBULIN BY
PREPARATIVE CHROMATOGRAPHY USING SIMULATED MOVING BEDS
S.L. LUCENA, P.T.V. ROSA, L.T. FURLAN AND C.C. SANTANA
DPB/FEQ/UNICAMP, C.P. 6066, CEP 13083-970 Campinas, SP, Brazil
Abstract
Simulated moving bed (SMB) is an important separation process that uses a series of columns of adsorption connected being formed a circuit that is divided in, for example, four zones defined by two entrances (feeding and desorbant) and two exits (extract and rafinate). Those entrances and exits are periodically moved in the direction of the liquid flow simulating a countercurrent movement with the phase solid adsorbent. In this work, experimental data for equilibrium isotherms in an ion-exchange resin were coupled to mathematical formulation leading to a computational routine developed for the estimation of the concentration profiles in the extract and in the rafinate of constituted by the proteins 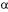 -lactalbumin and
-lactalbumin and  -lactoglobulin in a binary mixture. Those proteins are present in cheese whey with concentrations of the order of 1,5 g/L to 3,0 g/L, respectively, and, in high levels of concentrations, exhibit application in the veterinary medicine and as supplement for of culture of cells media, being generally residues of milk processing industries. Chromatographic profiles obtained for a SMB with twelve columns indicates conditions that meets the complete separation of the two components from the mixture.
-lactoglobulin in a binary mixture. Those proteins are present in cheese whey with concentrations of the order of 1,5 g/L to 3,0 g/L, respectively, and, in high levels of concentrations, exhibit application in the veterinary medicine and as supplement for of culture of cells media, being generally residues of milk processing industries. Chromatographic profiles obtained for a SMB with twelve columns indicates conditions that meets the complete separation of the two components from the mixture.
1. Introduction
The products of biotechnological origin possess great diversity and are generally present in fermentation broths and of cultures of cells in low concentrations. Diluted systems coupled to considerable amounts of chemical species that interfere in the recovery processes, concentration and final purification turn those difficult and onerous tasks, answering in most of the processes for the largest part of the costs of biomolecules production. In the special case of the proteins the percentages of the purification costs reaches values of the order of 60% in relation to the total costs of the production process, could located in the range of up to 80 to 90% for original fermentation products of recombinant DNA (Blanch and Clark, 1997). The
325
M. Hofman and P. Thonart (eds.), Engineering and Manufacturing for Biotechnology, 325–337. © 2001 Kluwer Academic Publishers. Printed in the Netherlands.
S.L. Lucena, P.T.V. Rosa, L.T. Furlan and C.C. Santana
conventional liquid chromatographic (Ganetsos and Barker, 1993). The largest disadvantages of the usual chromatographic separations lasts in the discontinuity of the process and in the dilution of the product. It is a well-known fact in the operations of adsorption that continuous systems in which the phase solid is contacted in the direction opposed the one of the flowing phase that the profile of mass transfer stays stationary and the adsorbent is used in a more efficient way.
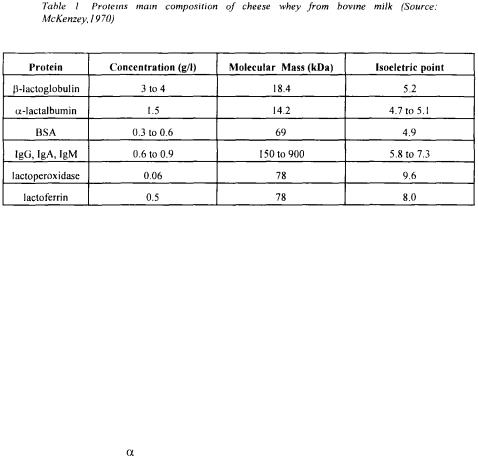
Purified proteins derived from milk products like whey are acquiring important applications in medicine, veterinary, as food functional products and for cells media culture. In many countries, whey from milk products like cheese are discarded in mainstreams, causing pollution problems. As restrictions on the discharge of pollutants increase, new strategies for waste treatment must be found. Future initiatives to improve recovery of by-products will require additional research to investigate possible uses for valuable waste stream components and to develop cost-effective techniques for their recovery (Beszedits,1982). A typical composition of a cheese whey is shown in Table 1.
Increasingly opportunities exist for the commercial extraction of these bioproducts even in diluted solutions. The utilisation of chromatographic processes based on ionexchange resins coupled to chemical engineering principles is one example of process that can be applied to the design , scale-up and optimisation of large scale systems
(Carrére, 1993). A fundamental understanding of adsorption processes as well as of
novel equipment configurations enables foresee the fractionation of individual proteins |
in economic basis. The search of preparative methods |
to |
separate and purify fragile |
products like proteins is an important issue connected |
to |
the increasing demand and |
higher throughput of proteins in the biotechnological industry. The utilisation of large |
scale expanded bed columns and the search of continuous methods of separation are |
examples of trying to meet |
some of |
the preparative techniques requirements. |
Continuous processes using |
Simulated |
Moving Beds (SMB) as a chromatographic |
procedure was utilised by Huang et al. (1986) who demonstrated the feasibility of SMB as a chromatographic procedure to purify enzymes. Gottschlich et al. (1997) applied an
SMB system to purify -chymotrypsin on immobilised soybean trypsin inhibitor, based
Separation of  -lactalbumin and
-lactalbumin and  -lactoglobulin by preparative chromatography
-lactoglobulin by preparative chromatography
therefore on biospecific affinity phenomena. This paper addresses the utilisation of adsorption kinetics and equilibrium data in ion-exchange adsorbents connected to a linear model for SMB performance A model binary system produced artificially by using 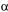 ,-lactalbumin and
,-lactalbumin and 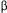 -lactoglobulin was chosen to demonstrate the feasibility of separation of these major bovine milk serum components.
-lactoglobulin was chosen to demonstrate the feasibility of separation of these major bovine milk serum components.
2. Basic concepts of processes of separation with simulated moving beds
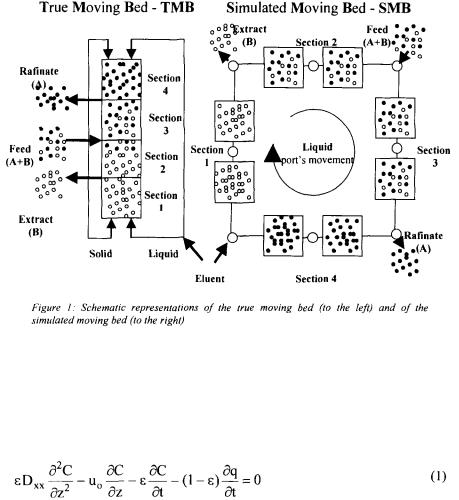
In Figure 1 two different processes of adsorption are presented in a countercurrent mode of operation. The well-known process called true moving bed (TMB) allows the obtaining of a continuous operation, distinctly of the classic chromatographic elution process. On the other hand, due to the difficulties of implementing the circulation of the solids, efforts have been driven to develop processes that maintain the advantages of the countercurrent operation but that avoid the circulation of the solids. In most of the innovations in that sense the movement of the solids is obtained by periodic changes in the feeding and discharge in a system of multiple columns resulting the outline of well- known process as simulated moving bed (SMB).
Since 1964, continuous chromatographic systems have been used mainly in industrial scale in the petrochemical industries (Processes SORBEX and PAREX, developed by United Oil Products), and of processing of sugar (Barker and Abusabah,
1985). The technology of SMB has also been studied and applied the products of fine chemistry (Ganetsos and Barker, 1993) and of biotechnological origin (Yamamoto et al., 1992), especially in enantiomers separation. SMB presents economic advantages over other chromatographic systems for several reasons: it is a continuous process and it allows to separate starting from a similar composed mixture, allowing high productions and low solvent consumption. In general in that system type the volume of requested adsorbent is approximately 25% of the requested in batch chromatography
(Gottschlich et al., 1996).
As depicted in the Figure 1, SMB uses a series of columns of adsorption (eight columns or twelve columns, for example) with an appropriate adsorbent. The columns are connected to recipients that contain the feeding and the eluent and that receive the currents of exit of the product through lines controlled by a group of valves of multiple positions. That group of controlling valves allows that they are alternate, in regular intervals of time, the points of entrance of the feeding, of the eluent and of the exit currents. The system changes therefore the positions between the entrance points and exit, simulating the countercurrent flow.
From the point of view of the operational variables, the project of SMB is relatively complex because it involves at least ten specific parameters to know: diameter of the columns, four lengths of separation zones, four flowing currents and a average velocity associated to the control of the opening of the valves of multiple positions. LMS is usually used for a mixture that contains two similar products, of the which it is attempted the separation. The use of SMB in the separation of multicomponent mixtures is not still very well known (Ganetsos and Barker, 1993). The main claim of this
S.L. Lucena, P.T.V. Rosa, L.T. Furlan and C.C. Santana
separation method consists |
in its ability to separate mixtures of difficult resolution and |
for products of high added |
value . |
3. Mathematical formulation
The equation of the mass balance for the solute in the flowing phase is given by the equation of the rate as deduced by Blanch and Clark (1997):
where C is the concentration of the component i in the liquid phase, q is the concentration of the component i in the surface of the particles, z is the axial position, and 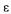 it is the bed porosity, uo it is the superficial speed and
it is the bed porosity, uo it is the superficial speed and 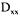 is the coefficient of axial dispersion.
is the coefficient of axial dispersion.
The terms of the equation (1) represents the axial dispersion, the convection in the bed, the accumulation in the liquid and accumulation in the particles, respectively. If it be considered that there are no resistances for the mass transfer of the adsorbed component in the film of the particle and in the pores of the resins, the concentrations
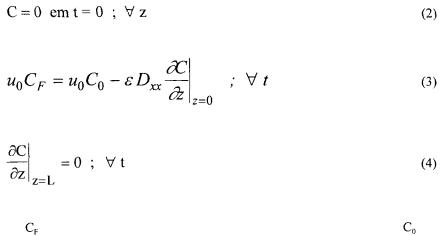
Separation of 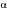 -lactalbumin and
-lactalbumin and  -lactoglobulin by preparative chromatography
-lactoglobulin by preparative chromatography
of the material to be adsorbed in the solid |
and in the liquid will be directly related for |
the isotherm of adsorption of the material. |
|
The boundary conditions for the system are given for:
where |
is the concentration of the component i in the feeding and |
is the |
concentration of the component i in the beginning of the bed and  is the superficial liquid velocity. Boundary condition described by equation (3) is known as a Dankwerts type condition (Dankwerts, 1953) and takes in account the effect of axial dispersion at the column entrance.
is the superficial liquid velocity. Boundary condition described by equation (3) is known as a Dankwerts type condition (Dankwerts, 1953) and takes in account the effect of axial dispersion at the column entrance.
3.1. APPLICATION FOR A COLUMN OF ADSORPTION
As pointed out above, the value of the concentration of the solute in the solid (q) it is related with the concentration of the solute in the liquid through the isotherms of adsorption. Dimensionless forms previous equations with the application of classical forms of isotherms for the concentration q of the component i in the surface of the particles are presented in Appendix. Equations (5) and (6) presents the equations in the dimensionless form obtained starting from the equation (1) for isotherms of adsorption of the linear type and Langmuir type, while the equations (7) and (8) represents the equations for the isotherm of adsorption of competitive Langmuir for two components. The set of equations were solved for the study of adsorption in one column using the method of the orthogonal collocation method for the axial position and the Runge-Kutta adaptive method with control step (Press et al., 1992) for the time.
3.2. APPLICATION TO THE SIMULATED MOVING BED
The simulated moving bed was considered as a group of interlinked columns of adsorption as shown in Figure 1. It was considered that the moving bed is formed by 3
columns in each section, resulting in a total of |
12 columns for the system. The operation |
is started with the whole system |
filled |
with the solvent and, therefore, the |
concentrations of the components in all the columns were considered as being the same
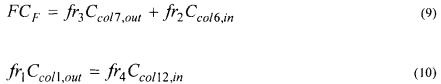
S.L. Lucena, P.T.V. Rosa, L.T. Furlan and C.C. Santana
and equal to zero in this period. As the columns are connected in series, the liquid flowrate is constant in each section of the bed. The twelve columns were jointly simulated and the feed concentration of the columns are different from the feeding of the system. The eluent was considered to be pure, without proteins. In the feeding columns and at the point of the solvent introduction the concentrations were obtained as described in equations (5) and (6):
where frl, fr2, fr3 and fr4 are the flow rates in the sections 1,2,3 and 4, and Ccol7,out and Ccoll,out are the concentrations in the exit of the columns 7 and 1.
Equations (1) through (4) are applied for each column and integrated along the time.
When switching time is reached, the positions of the currents of the system were rotated, passing for the columns to the left. The concentration profiles in each column was maintained constant during the change of the currents. This procedure was used up to reach the required final number of rotations.
4. Adsorption isotherms
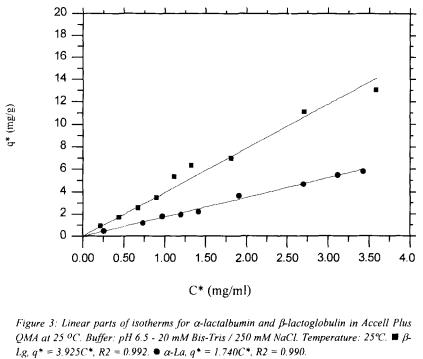
The isotherms were obtained to 25 °C after determination of the kinetics of adsorption so that the time to reach the equilibrium was known. About 4 ml of the solutions of proteins with different concentrations were incubated in a rotative agitator with a certain amount of the resin (about 20 mg) in syringes of 5 ml. After 45 minutes (time of equilibrium) the concentrations in the supernatant were measured in an spectrophotometer to 280 nm being taken as reference a calibration curve. Using a mass balance, the concentrations of proteins adsorbed in the resin could be ascertained. The concentrations of proteins in the supernatant and in the resin were then adjusted being used the model of Langmuir, as shown in Figure 2 , while Figure 3 depicts the linear part of the experimentally obtained isotherms.
5. Results and discussion
5.1. INDIVIDUAL COLUMN OF ADSORPTION
The simulated moving bed consists basically of a group of interlinked individual columns of adsorption in a recurrent way, and the material that leaves a column it is fed in an adjacent column. The understanding of the behaviour of an adsorption column is of vital importance for the understanding of the operation of the simulated moving bed.
Several factors can alter the form of the profile of concentration of a certain solute in
S.L. Lucena, P.T.V. Rosa, L.T. Furlan and C.C. Santana
Symbols shown in the figure are results of the simulation process. The competition of the two solutes in adsorption of the resins results in a smaller liquid rate of adsorption, resulting in smaller breakthrough times. In this value of feeding concentration the term of KC cannot be disregarded in the Langmuir equation, because there is a great variation among the profiles obtained by this model of adsorption and for the linear model. The choice among the model of Langmuir and of competitive Langmuir depends on the system under study. In the case of affinity resins that presents non-specific adsorption, the model of Langmuir can be used . Considering the case of resins that don't have great specificity for a certain solute, the isotherm of competitive Langmuir should be used since it is possible to determine the adsorption characteristics of the competitive species.
5.2. SIMULATED MOVING BED
In a SMB, the compound that interacts more intensely with the adsorbent will move in the solid phase direction and will be recovered in the extract stream. Conversely, the material that exhibits lower tendency to accumulate on the solid particles will be carried on by the liquid flow and will be recovered in the raffinate port. The 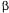 -lactoglobulin
-lactoglobulin

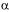 -LACTALBUMIN AND
-LACTALBUMIN AND 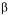 -LACTOGLOBULIN BY
-LACTOGLOBULIN BY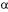 -lactalbumin and
-lactalbumin and  -lactoglobulin in a binary mixture. Those proteins are present in cheese whey with concentrations of the order of 1,5 g/L to 3,0 g/L, respectively, and, in high levels of concentrations, exhibit application in the veterinary medicine and as supplement for of culture of cells media, being generally residues of milk processing industries. Chromatographic profiles obtained for a SMB with twelve columns indicates conditions that meets the complete separation of the two components from the mixture.
-lactoglobulin in a binary mixture. Those proteins are present in cheese whey with concentrations of the order of 1,5 g/L to 3,0 g/L, respectively, and, in high levels of concentrations, exhibit application in the veterinary medicine and as supplement for of culture of cells media, being generally residues of milk processing industries. Chromatographic profiles obtained for a SMB with twelve columns indicates conditions that meets the complete separation of the two components from the mixture.
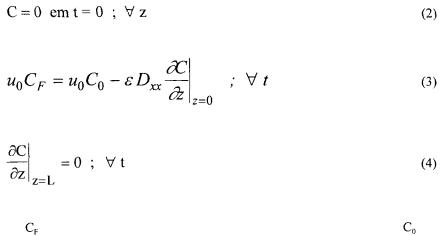
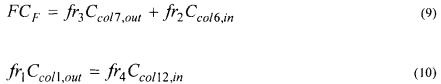



 -lactalbumin and
-lactalbumin and  -lactoglobulin by preparative chromatography
-lactoglobulin by preparative chromatography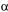
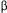
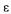 it is the bed porosity, uo it is the superficial speed and
it is the bed porosity, uo it is the superficial speed and 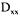 is the coefficient of axial dispersion.
is the coefficient of axial dispersion.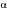 -lactalbumin and
-lactalbumin and  -lactoglobulin by preparative chromatography
-lactoglobulin by preparative chromatography
 -lactalbumin and
-lactalbumin and 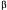 -lactoglobulin by preparative chromatography
-lactoglobulin by preparative chromatography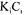 if compared with 1), practically the phenomenon of competition of adsorption is not observed and the linear isotherm represents well the process of adsorption. With the increase of the solute concentration, the linear isotherm doesn't represent more the process of adsorption and the models of Langmuir and of competitive Langmuir describes better the equilibrium. Figure 4 presents the breakthrough curve obtained through the simulation of the equations (5) the (8) together with the initial condition (2) and with the boundary conditions (3) and (4) for the adsorption of two solutes that are fed in the column of adsorption with a such concentration that the value of
if compared with 1), practically the phenomenon of competition of adsorption is not observed and the linear isotherm represents well the process of adsorption. With the increase of the solute concentration, the linear isotherm doesn't represent more the process of adsorption and the models of Langmuir and of competitive Langmuir describes better the equilibrium. Figure 4 presents the breakthrough curve obtained through the simulation of the equations (5) the (8) together with the initial condition (2) and with the boundary conditions (3) and (4) for the adsorption of two solutes that are fed in the column of adsorption with a such concentration that the value of 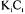 is not small if compared with the unit.
is not small if compared with the unit.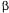 -lactoglobulin
-lactoglobulin