
- •CONTENTS
- •Preface
- •Contributors
- •1 Introduction to Toxicology
- •1.1 Definition and Scope, Relationship to Other Sciences, and History
- •1.1.2 Relationship to Other Sciences
- •1.1.3 A Brief History of Toxicology
- •1.3 Sources of Toxic Compounds
- •1.3.1 Exposure Classes
- •1.3.2 Use Classes
- •1.4 Movement of Toxicants in the Environment
- •Suggested Reading
- •2.1 Introduction
- •2.2 Cell Culture Techniques
- •2.2.1 Suspension Cell Culture
- •2.2.2 Monolayer Cell Culture
- •2.2.3 Indicators of Toxicity in Cultured Cells
- •2.3 Molecular Techniques
- •2.3.1 Molecular Cloning
- •2.3.2 cDNA and Genomic Libraries
- •2.3.3 Northern and Southern Blot Analyses
- •2.3.4 Polymerase Chain Reaction (PCR)
- •2.3.5 Evaluation of Gene Expression, Regulation, and Function
- •2.4 Immunochemical Techniques
- •Suggested Reading
- •3.1 Introduction
- •3.2 General Policies Related to Analytical Laboratories
- •3.2.1 Standard Operating Procedures (SOPs)
- •3.2.2 QA/QC Manuals
- •3.2.3 Procedural Manuals
- •3.2.4 Analytical Methods Files
- •3.2.5 Laboratory Information Management System (LIMS)
- •3.3 Analytical Measurement System
- •3.3.1 Analytical Instrument Calibration
- •3.3.2 Quantitation Approaches and Techniques
- •3.4 Quality Assurance (QA) Procedures
- •3.5 Quality Control (QC) Procedures
- •3.6 Summary
- •Suggested Reading
- •4 Exposure Classes, Toxicants in Air, Water, Soil, Domestic and Occupational Settings
- •4.1 Air Pollutants
- •4.1.1 History
- •4.1.2 Types of Air Pollutants
- •4.1.3 Sources of Air Pollutants
- •4.1.4 Examples of Air Pollutants
- •4.1.5 Environmental Effects
- •4.2 Water and Soil Pollutants
- •4.2.1 Sources of Water and Soil Pollutants
- •4.2.2 Examples of Pollutants
- •4.3 Occupational Toxicants
- •4.3.1 Regulation of Exposure Levels
- •4.3.2 Routes of Exposure
- •4.3.3 Examples of Industrial Toxicants
- •Suggested Reading
- •5 Classes of Toxicants: Use Classes
- •5.1 Introduction
- •5.2 Metals
- •5.2.1 History
- •5.2.2 Common Toxic Mechanisms and Sites of Action
- •5.2.3 Lead
- •5.2.4 Mercury
- •5.2.5 Cadmium
- •5.2.6 Chromium
- •5.2.7 Arsenic
- •5.2.8 Treatment of Metal Poisoning
- •5.3 Agricultural Chemicals (Pesticides)
- •5.3.1 Introduction
- •5.3.3 Organochlorine Insecticides
- •5.3.4 Organophosphorus Insecticides
- •5.3.5 Carbamate Insecticides
- •5.3.6 Botanical Insecticides
- •5.3.7 Pyrethroid Insecticides
- •5.3.8 New Insecticide Classes
- •5.3.9 Herbicides
- •5.3.10 Fungicides
- •5.3.11 Rodenticides
- •5.3.12 Fumigants
- •5.3.13 Conclusions
- •5.4 Food Additives and Contaminants
- •5.5 Toxins
- •5.5.1 History
- •5.5.2 Microbial Toxins
- •5.5.3 Mycotoxins
- •5.5.4 Algal Toxins
- •5.5.5 Plant Toxins
- •5.5.6 Animal Toxins
- •5.6 Solvents
- •5.7 Therapeutic Drugs
- •5.8 Drugs of Abuse
- •5.9 Combustion Products
- •5.10 Cosmetics
- •Suggested Reading
- •6 Absorption and Distribution of Toxicants
- •6.1 Introduction
- •6.2 Cell Membranes
- •6.3 Mechanisms of Transport
- •6.3.1 Passive Diffusion
- •6.4 Physicochemical Properties Relevant to Diffusion
- •6.4.1 Ionization
- •6.5 Routes of Absorption
- •6.5.1 Extent of Absorption
- •6.5.2 Gastrointestinal Absorption
- •6.5.3 Dermal Absorption
- •6.5.4 Respiratory Penetration
- •6.6 Toxicant Distribution
- •6.6.1 Physicochemical Properties and Protein Binding
- •6.7 Toxicokinetics
- •Suggested Reading
- •7 Metabolism of Toxicants
- •7.1 Introduction
- •7.2 Phase I Reactions
- •7.2.4 Nonmicrosomal Oxidations
- •7.2.5 Cooxidation by Cyclooxygenases
- •7.2.6 Reduction Reactions
- •7.2.7 Hydrolysis
- •7.2.8 Epoxide Hydration
- •7.2.9 DDT Dehydrochlorinase
- •7.3 Phase II Reactions
- •7.3.1 Glucuronide Conjugation
- •7.3.2 Glucoside Conjugation
- •7.3.3 Sulfate Conjugation
- •7.3.4 Methyltransferases
- •7.3.7 Acylation
- •7.3.8 Phosphate Conjugation
- •Suggested Reading
- •8 Reactive Metabolites
- •8.1 Introduction
- •8.2 Activation Enzymes
- •8.3 Nature and Stability of Reactive Metabolites
- •8.4 Fate of Reactive Metabolites
- •8.4.1 Binding to Cellular Macromolecules
- •8.4.2 Lipid Peroxidation
- •8.4.3 Trapping and Removal: Role of Glutathione
- •8.5 Factors Affecting Toxicity of Reactive Metabolites
- •8.5.1 Levels of Activating Enzymes
- •8.5.2 Levels of Conjugating Enzymes
- •8.5.3 Levels of Cofactors or Conjugating Chemicals
- •8.6 Examples of Activating Reactions
- •8.6.1 Parathion
- •8.6.2 Vinyl Chloride
- •8.6.3 Methanol
- •8.6.5 Carbon Tetrachloride
- •8.6.8 Acetaminophen
- •8.6.9 Cycasin
- •8.7 Future Developments
- •Suggested Reading
- •9.1 Introduction
- •9.2 Nutritional Effects
- •9.2.1 Protein
- •9.2.2 Carbohydrates
- •9.2.3 Lipids
- •9.2.4 Micronutrients
- •9.2.5 Starvation and Dehydration
- •9.2.6 Nutritional Requirements in Xenobiotic Metabolism
- •9.3 Physiological Effects
- •9.3.1 Development
- •9.3.2 Gender Differences
- •9.3.3 Hormones
- •9.3.4 Pregnancy
- •9.3.5 Disease
- •9.3.6 Diurnal Rhythms
- •9.4 Comparative and Genetic Effects
- •9.4.1 Variations Among Taxonomic Groups
- •9.4.2 Selectivity
- •9.4.3 Genetic Differences
- •9.5 Chemical Effects
- •9.5.1 Inhibition
- •9.5.2 Induction
- •9.5.3 Biphasic Effects: Inhibition and Induction
- •9.6 Environmental Effects
- •9.7 General Summary and Conclusions
- •Suggested Reading
- •10 Elimination of Toxicants
- •10.1 Introduction
- •10.2 Transport
- •10.3 Renal Elimination
- •10.4 Hepatic Elimination
- •10.4.2 Active Transporters of the Bile Canaliculus
- •10.5 Respiratory Elimination
- •10.6 Conclusion
- •Suggested Reading
- •11 Acute Toxicity
- •11.1 Introduction
- •11.2 Acute Exposure and Effect
- •11.3 Dose-response Relationships
- •11.4 Nonconventional Dose-response Relationships
- •11.5 Mechanisms of Acute Toxicity
- •11.5.1 Narcosis
- •11.5.2 Acetylcholinesterase Inhibition
- •11.5.3 Ion Channel Modulators
- •11.5.4 Inhibitors of Cellular Respiration
- •Suggested Reading
- •12 Chemical Carcinogenesis
- •12.1 General Aspects of Cancer
- •12.2 Human Cancer
- •12.2.1 Causes, Incidence, and Mortality Rates of Human Cancer
- •12.2.2 Known Human Carcinogens
- •12.3 Classes of Agents Associated with Carcinogenesis
- •12.3.2 Epigenetic Agents
- •12.4 General Aspects of Chemical Carcinogenesis
- •12.5 Initiation-Promotion Model for Chemical Carcinogenesis
- •12.6 Metabolic Activation of Chemical Carcinogens and DNA Adduct Formation
- •12.7 Oncogenes
- •12.8 Tumor Suppressor Genes
- •12.8.1 Inactivation of Tumor Suppressor Genes
- •12.8.2 p53 Tumor Suppressor Gene
- •12.9 General Aspects of Mutagenicity
- •12.10 Usefulness and Limitations of Mutagenicity Assays for the Identification of Carcinogens
- •Suggested Reading
- •13 Teratogenesis
- •13.1 Introduction
- •13.2 Principles of Teratology
- •13.3 Mammalian Embryology Overview
- •13.4 Critical Periods
- •13.5 Historical Teratogens
- •13.5.1 Thalidomide
- •13.5.2 Accutane (Isotetrinoin)
- •13.5.3 Diethylstilbestrol (DES)
- •13.5.4 Alcohol
- •13.6 Testing Protocols
- •13.6.1 FDA Guidelines for Reproduction Studies for Safety Evaluation of Drugs for Human Use
- •13.6.3 Alternative Test Methods
- •13.7 Conclusions
- •Suggested Reading
- •14 Hepatotoxicity
- •14.1 Introduction
- •14.1.1 Liver Structure
- •14.1.2 Liver Function
- •14.2 Susceptibility of the Liver
- •14.3 Types of Liver Injury
- •14.3.1 Fatty Liver
- •14.3.2 Necrosis
- •14.3.3 Apoptosis
- •14.3.4 Cholestasis
- •14.3.5 Cirrhosis
- •14.3.6 Hepatitis
- •14.3.7 Oxidative Stress
- •14.3.8 Carcinogenesis
- •14.4 Mechanisms of Hepatotoxicity
- •14.5 Examples of Hepatotoxicants
- •14.5.1 Carbon Tetrachloride
- •14.5.2 Ethanol
- •14.5.3 Bromobenzene
- •14.5.4 Acetaminophen
- •14.6 Metabolic Activation of Hepatotoxicants
- •Suggested Reading
- •15 Nephrotoxicity
- •15.1 Introduction
- •15.1.1 Structure of the Renal System
- •15.1.2 Function of the Renal System
- •15.2 Susceptibility of the Renal System
- •15.3 Examples of Nephrotoxicants
- •15.3.1 Metals
- •15.3.2 Aminoglycosides
- •15.3.3 Amphotericin B
- •15.3.4 Chloroform
- •15.3.5 Hexachlorobutadiene
- •Suggested Reading
- •16 Toxicology of the Nervous System
- •16.1 Introduction
- •16.2 The Nervous system
- •16.2.1 The Neuron
- •16.2.2 Neurotransmitters and their Receptors
- •16.2.3 Glial Cells
- •16.3 Toxicant Effects on the Nervous System
- •16.3.1 Structural Effects of Toxicants on Neurons
- •16.3.2 Effects of Toxicants on Other Cells
- •16.4 Neurotoxicity Testing
- •16.4.1 In vivo Tests of Human Exposure
- •16.4.2 In vivo Tests of Animal Exposure
- •16.4.3 In vitro Neurochemical and Histopathological End Points
- •16.5 Summary
- •Suggested Reading
- •17 Endocrine System
- •17.1 Introduction
- •17.2 Endocrine System
- •17.2.1 Nuclear Receptors
- •17.3 Endocrine Disruption
- •17.3.1 Hormone Receptor Agonists
- •17.3.2 Hormone Receptor Antagonists
- •17.3.3 Organizational versus Activational Effects of Endocrine Toxicants
- •17.3.4 Inhibitors of Hormone Synthesis
- •17.3.5 Inducers of Hormone Clearance
- •17.3.6 Hormone Displacement from Binding Proteins
- •17.4 Incidents of Endocrine Toxicity
- •17.4.1 Organizational Toxicity
- •17.4.2 Activational Toxicity
- •17.4.3 Hypothyroidism
- •17.5 Conclusion
- •Suggested Reading
- •18 Respiratory Toxicity
- •18.1 Introduction
- •18.1.1 Anatomy
- •18.1.2 Cell Types
- •18.1.3 Function
- •18.2 Susceptibility of the Respiratory System
- •18.2.1 Nasal
- •18.2.2 Lung
- •18.3 Types of Toxic Response
- •18.3.1 Irritation
- •18.3.2 Cell Necrosis
- •18.3.3 Fibrosis
- •18.3.4 Emphysema
- •18.3.5 Allergic Responses
- •18.3.6 Cancer
- •18.3.7 Mediators of Toxic Responses
- •18.4 Examples of Lung Toxicants Requiring Activation
- •18.4.1 Introduction
- •18.4.2 Monocrotaline
- •18.4.3 Ipomeanol
- •18.4.4 Paraquat
- •18.5 Defense Mechanisms
- •Suggested Reading
- •19 Immunotoxicity
- •19.1 Introduction
- •19.2 The Immune System
- •19.3 Immune Suppression
- •19.4 Classification of Immune-Mediated Injury (Hypersensitivity)
- •19.5 Effects of Chemicals on Allergic Disease
- •19.5.1 Allergic Contact Dermatitis
- •19.5.2 Respiratory Allergens
- •19.5.3 Adjuvants
- •19.6 Emerging Issues: Food Allergies, Autoimmunity, and the Developing Immune System
- •Suggested Reading
- •20 Reproductive System
- •20.1 Introduction
- •20.2 Male Reproductive Physiology
- •20.3 Mechanisms and Targets of Male Reproductive Toxicants
- •20.3.1 General Mechanisms
- •20.3.2 Effects on Germ Cells
- •20.3.3 Effects on Spermatogenesis and Sperm Quality
- •20.3.4 Effects on Sexual Behavior
- •20.3.5 Effects on Endocrine Function
- •20.4 Female Reproductive Physiology
- •20.5 Mechanisms and Targets of Female Reproductive Toxicants
- •20.5.1 Tranquilizers, Narcotics, and Social Drugs
- •20.5.2 Endocrine Disruptors (EDs)
- •20.5.3 Effects on Germ Cells
- •20.5.4 Effects on the Ovaries and Uterus
- •20.5.5 Effects on Sexual Behavior
- •Suggested Reading
- •21 Toxicity Testing
- •21.1 Introduction
- •21.2 Experimental Administration of Toxicants
- •21.2.1 Introduction
- •21.2.2 Routes of Administration
- •21.3 Chemical and Physical Properties
- •21.4 Exposure and Environmental Fate
- •21.5 In vivo Tests
- •21.5.1 Acute and Subchronic Toxicity Tests
- •21.5.2 Chronic Tests
- •21.5.3 Reproductive Toxicity and Teratogenicity
- •21.5.4 Special Tests
- •21.6 In vitro and Other Short-Term Tests
- •21.6.1 Introduction
- •21.6.2 Prokaryote Mutagenicity
- •21.6.3 Eukaryote Mutagenicity
- •21.6.4 DNA Damage and Repair
- •21.6.5 Chromosome Aberrations
- •21.6.6 Mammalian Cell Transformation
- •21.6.7 General Considerations and Testing Sequences
- •21.7 Ecological Effects
- •21.7.1 Laboratory Tests
- •21.7.2 Simulated Field Tests
- •21.7.3 Field Tests
- •21.8 Risk Analysis
- •21.9 The Future of Toxicity Testing
- •Suggested Reading
- •22 Forensic and Clinical Toxicology
- •22.1 Introduction
- •22.2 Foundations of Forensic Toxicology
- •22.3 Courtroom Testimony
- •22.4.1 Documentation Practices
- •22.4.2 Considerations for Forensic Toxicological Analysis
- •22.4.3 Drug Concentrations and Distribution
- •22.5 Laboratory Analyses
- •22.5.1 Colorimetric Screening Tests
- •22.5.2 Thermal Desorption
- •22.5.6 Enzymatic Immunoassay
- •22.6 Analytical Schemes for Toxicant Detection
- •22.7 Clinical Toxicology
- •22.7.1 History Taking
- •22.7.2 Basic Operating Rules in the Treatment of Toxicosis
- •22.7.3 Approaches to Selected Toxicoses
- •Suggested Reading
- •23 Prevention of Toxicity
- •23.1 Introduction
- •23.2 Legislation and Regulation
- •23.2.1 Federal Government
- •23.2.2 State Governments
- •23.2.3 Legislation and Regulation in Other Countries
- •23.3 Prevention in Different Environments
- •23.3.1 Home
- •23.3.2 Workplace
- •23.3.3 Pollution of Air, Water, and Land
- •23.4 Education
- •Suggested Reading
- •24 Human Health Risk Assessment
- •24.1 Introduction
- •24.2 Risk Assessment Methods
- •24.2.2 Exposure Assessment
- •24.2.3 Dose Response and Risk Characterization
- •24.3 Noncancer Risk Assessment
- •24.3.1 Default Uncertainty and Modifying Factors
- •24.3.2 Derivation of Developmental Toxicant RfD
- •24.3.3 Determination of RfD and RfC of Naphthalene with the NOAEL Approach
- •24.3.4 Benchmark Dose Approach
- •24.3.5 Determination of BMD and BMDL for ETU
- •24.3.6 Quantifying Risk for Noncarcinogenic Effects: Hazard Quotient
- •24.3.7 Chemical Mixtures
- •24.4 Cancer Risk Assessment
- •24.5 PBPK Modeling
- •Suggested Reading
- •25 Analytical Methods in Toxicology
- •25.1 Introduction
- •25.2 Chemical and Physical Methods
- •25.2.1 Sampling
- •25.2.2 Experimental Studies
- •25.2.3 Forensic Studies
- •25.2.4 Sample Preparation
- •25.2.6 Spectroscopy
- •25.2.7 Other Analytical Methods
- •Suggested Reading
- •26 Basics of Environmental Toxicology
- •26.1 Introduction
- •26.2 Environmental Persistence
- •26.2.1 Abiotic Degradation
- •26.2.2 Biotic Degradation
- •26.2.3 Nondegradative Elimination Processes
- •26.3 Bioaccumulation
- •26.4 Toxicity
- •26.4.1 Acute Toxicity
- •26.4.2 Mechanisms of Acute Toxicity
- •26.4.3 Chronic Toxicity
- •26.4.5 Abiotic and Biotic Interactions
- •26.5 Conclusion
- •Suggested Reading
- •27.1 Introduction
- •27.2 Sources of Toxicants to the Environment
- •27.3 Transport Processes
- •27.3.1 Advection
- •27.3.2 Diffusion
- •27.4 Equilibrium Partitioning
- •27.5 Transformation Processes
- •27.5.1 Reversible Reactions
- •27.5.2 Irreversible Reactions
- •27.6 Environmental Fate Models
- •Suggested Reading
- •28 Environmental Risk Assessment
- •28.1 Introduction
- •28.2 Formulating the Problem
- •28.2.1 Selecting Assessment End Points
- •28.2.2 Developing Conceptual Models
- •28.2.3 Selecting Measures
- •28.3 Analyzing Exposure and Effects Information
- •28.3.1 Characterizing Exposure
- •28.3.2 Characterizing Ecological Effects
- •28.4 Characterizing Risk
- •28.4.1 Estimating Risk
- •28.4.2 Describing Risk
- •28.5 Managing Risk
- •Suggested Reading
- •29 Future Considerations for Environmental and Human Health
- •29.1 Introduction
- •29.2 Risk Management
- •29.3 Risk Assessment
- •29.4 Hazard and Exposure Assessment
- •29.5 In vivo Toxicity
- •29.6 In vitro Toxicity
- •29.7 Biochemical and Molecular Toxicology
- •29.8 Development of Selective Toxicants
- •Glossary
- •Index

336 IMMUNOTOXICITY
antigen. Type I reactions include immediate asthmatic responses to allergen, allergic rhinitis (hay fever), atopic dermatitis (eczema), and acute urticaria (hives). The most severe form is systemic anaphylaxis (e.g., in response to a bee sting), which results in anaphylactic shock, and potentially death.
Type II hypersensitivity is the result of antibody-mediated cytotoxicity that occurs when antibodies respond to cell surface antigens. Antibodies bound to antigen on the cell surface activate the complement system and/or macrophages, leading to lysis of the target cell. Frequently blood cells are the targets, as in the case of an incompatible blood transfusion or Rh blood incompatibility between mother and child. The basement membrane of the kidney or lung may also be a target. Autoimmune diseases can result from drug treatments with penicillin, quinidine, quinine, or acetaminophen. Apparently these drugs interact with the cell membrane such that the immune system detects “foreign” antigens on the cell surface. This type of autoimmune disease may also have unknown etiologies.
Type III reactions are the result of antigen-antibody (IgG) complexes that accumulate in tissues or the circulation, activate macrophages and the complement system, and trigger the influx of granulocytes and lymphocytes (inflammation). This is sometimes referred to as the Arthrus reaction and includes postinfection sequelae such as rheumatic heart disease. Farmer’s lung, a pneumonitis caused by molds has been attributed to both type III and type IV, and some of the late phase response (4–6 hours after exposure) in asthmatics may be the result of Arthrus-type reactions.
Unlike the preceding three types, type IV, or delayed-type hypersensitivity (DTH), involves T cells and macrophages, not antibodies. Activated T cells release cytokines that cause accumulation and activation of macrophages, which in turn cause local damage. This type of reaction is very important in defense against intracellular infections such as tuberculosis, but is also responsible for contact hypersensitivity responses (allergic contact dermatitis) such as the response to poison ivy. Inhalation of beryllium can result in a range of pathologies, including acute pneumonitis, tracheobronchitis, and chronic beryllium disease, all of which appear to be due to type IV berylliumspecific immune responses. The expression of type IV responses following challenge is delayed, occurring 24 to 48 hours after exposure.
The different types of immune-mediated injury are not mutually exclusive. More than one hypersensitivity mechanism may be involved in the response to a particular antigen. Also the resulting pathology, particularly that caused by type III and IV, reactions may appear very similar, although the mechanisms leading to the effect are different.
19.5EFFECTS OF CHEMICALS ON ALLERGIC DISEASE
Xenobiotics can affect allergic disease in one of two ways. They can themselves act as antigens and elicit hypersensitivity responses, or they can enhance the development or expression of allergic responses to commonly encountered allergens, such as dust mite. Chemicals that act as allergens include certain proteins that can by themselves induce an immune response and low molecular weight chemicals (known as haptens) that are too small to induce a specific immune response but may react with a protein to induce an immune response that is then hapten specific. Haptens have been associated with both allergic contact dermatitis (ACD), sometimes called contact hypersensitivity

EFFECTS OF CHEMICALS ON ALLERGIC DISEASE |
337 |
(CHS), and respiratory hypersensitivity. Proteins have been associated with respiratory hypersensitivity and food allergies. When a chemical is an allergen or a hapten, there are two doses of concern, the sensitizing dose and the elicitation dose. In general, the dose required for sensitization is greater than that required to elicit a response in a sensitized individual. Chemicals that enhance the development of allergic sensitization are referred to as adjuvants. Air pollutants have been associated both with enhanced sensitization and exacerbation of allergic respiratory symptoms.
19.5.1Allergic Contact Dermatitis
Allergic contact dermatitis (ACD) or contact hypersensitivity (CHS) is one of the most common occupational health problems and hence is one of the most common problems associated with immunotoxicity. It is a type IV response that occurs as a result of dermal exposure to chemicals that are haptens. Following dermal exposure, the chemical reacts with host cell protein at the surface of the skin and is picked up by epidermal dendritic cells, known as Langerhans cells. Cytokines released from the epidermal keratinocytes and from Langerhans cells cause maturation and mobilization of the Langerhans cells, which travel to the draining local lymph node and present antigen to lymphocytes. Clonal expansion occurs, enlarging the number of T lymphocytes specific for that allergen and generating memory cells that, in addition to specificity for the allergen, have the propensity to home to the skin. On re-exposure to the chemical, these specific T cells are activated, proliferate, home rapidly to the site of exposure, and produce erythema and edema typical of a type IV response. The reaction to poison ivy is the classic example.
Methods to assess chemicals (drugs, pesticides, dyes, cosmetics, and household products, etc.) for potential to induce CHS are well established, and several protocols using guinea pigs have been in use since the 1950s. These protocols assess the actual disease end point, skin erythema, and edema, following sensitization and challenge with the test agent. Two commonly used tests are the guinea pig maximization test and the Buehler occluded patch test. The sensitization procedure for the maximization tests includes intradermal injection of the test chemical with an adjuvant (intended to enhance the sensitization process) as well as topical application. The Buehler test relies on topical sensitization alone. In both cases, after approximately 2 weeks, animals are challenged at a different site on the skin and erythema and edema are assessed 24 to 48 hours later. This assessment is somewhat subjective and these tests are fairly expensive.
A chemical is considered to be a sensitizer if 30% (maximization) or 15% (Buehler) of the animals respond. Recently a more economical, less subjective, test for CHS has been developed using mice. This test, the local lymph node assay (LLNA), assesses the proliferative response of lymphocytes in the draining lymph node following application of the agent to the ear and is based on our understanding of the immunologic mechanisms underlying CHS; that is, clonal expansion has to occur in the draining lymph node if there is to be allergic sensitization (Figure 19.5). The LLNA is gaining acceptance as a stand-alone alternative to the guinea pig tests and is likely to become the assay of choice.
Finally structure activity approaches have recently been developed to identify contact sensitizers. This approach is based on the concept that the biologic mechanisms that determine a chemical’s effect are related to its structure and hence chemicals with similar structures will have similar effects. Computer models have been developed to compare the structure of an unknown chemical to structures in a database for known
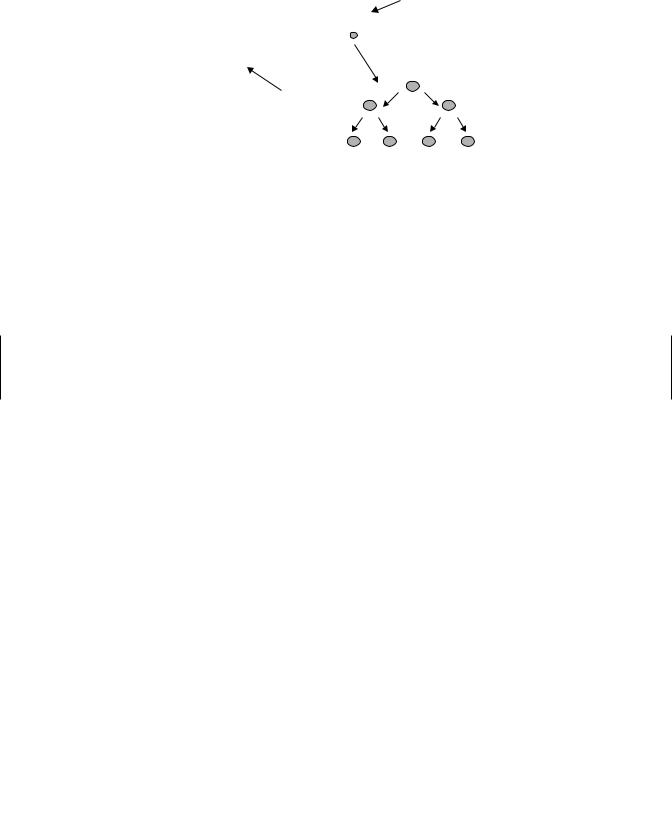
338 IMMUNOTOXICITY
Local Lymph |
1. Apply chemical to |
Node Assay |
both ears Days 1,2,3 |
2. Inject 3H- thymidine IV Day 6
3. Remove lymph node and measure proliferation:
5 hour after IV injection
Figure 19.5 Assessing chemicals for potential contact sensitivity. In the local lymph node assay the chemical in question is applied to both ears on three consecutive days. Control mice are treated with vehicle. Radioisotope is injected intravenously on day 6. The draining lymph nodes are removed 5 hours later and the proliferative response is measured by the incorporation of radio isotope. Results are frequently presented as a stimulation index (counts per min (cpm) for the test chemical/cpm for control). (Picture adapted from D. Sailstad, Lab Animal 31: 36, 2002.)
contact sensitizers. CHS lends itself to this approach because there is a large database of chemicals known to cause it, and there is a reasonable understanding of chemical characteristics that facilitate skin penetration, chemical reactivity with host proteins, and immune reactivity.
Because nonspecific inflammatory responses also can occur following chemical exposure to the skin, a distinction must be made between an irritant and a sensitizer. An irritant is an agent that causes local inflammatory effects but induces no immunological memory. Therefore, on subsequent exposures, local inflammation will again result, but there is no enhancement of the magnitude of the response and no change in the dose required to induce the response. In immunologically mediated inflammation (hypersensitivity) there may be no response to a sensitizer during the induction stage, but responses to subsequent exposures are exacerbated. The dose required for elicitation is usually less than that required to achieve sensitization.
19.5.2Respiratory Allergens
There is evidence that both occupational and environmental exposures to chemicals (both proteins and haptens) can result in the induction or exacerbation of respiratory allergies (Table 19.6). Of particular concern is the induction of allergic asthma. In sensitized asthmatic individuals the antigen challenge generally causes a type I (IgEmediated) immediate hypersensitivity response with release of mediators responsible for bronchoconstriction. Between 2 and 8 hours after the immediate response, asthmatics experience a more severe and prolonged (late phase) reaction that is characterized by mucus hypersecretion, bronchoconstriction, airway hyperresponsiveness to a variety of nonspecific stimuli (e.g., histamine, methacholine), and airway inflammation characterized by eosinophils. This later response is not mediated by IgE.

EFFECTS OF CHEMICALS ON ALLERGIC DISEASE |
339 |
Table 19.6 Example of Chemicals Associated with
Respiratory Allergy
Proteins
Enzymes
Latex
Animal dander
Dust mite
Molds
Cockroach
Microbial pesticides
Low molecular weight (<3000 )-haptens
Toluene diisocyanate
Diphenylmethane diisocyanate
Phthalic anhydride
Trimellitic anhydride
Platinum salts
Reactive dyes
Adjuvants
Ozone
Nitrogen dioxide
Diesel exhaust
Residual oil fly ash
Although proteins are generally immunogens, not all proteins are allergens and there is a range of potencies for those that are. There is also a strong genetic component associated with susceptibility to develop allergic reactions to proteins. Susceptible individuals are called atopic. There is at present no structural motif that can be used to characterize a protein as an allergen for hazard identification. Examples of occupational protein exposures associated with respiratory allergy and asthma include enzymes, latex, flour (both the grain itself and fungal contaminants), and animal dander. Environmental (mostly indoor) exposure including molds, spores, dust mite, animal dander, and cockroach have also been associated with this type of respiratory disease. Because this is a type 1 response, cytophilic antibodies (IgE) specific for the allergen are frequently used to identify proteins that may cause this effect. For example, in order to determine the etiology of occupational asthma in human subjects, the skin prick test is often used. Different proteins are injected under the skin to test for the presence of cytophilic antibodies in order to identify which proteins are causing a response in an individual. Serum may also be tested for protein specific IgE. Because IgE can sometimes be detected in the absence of respiratory responses, a positive IgE test may be followed by an assessment of respiratory responses.
Under very controlled situations patients may be exposed via the respiratory route to suspect allergens (bronchoprovocation test) and respiratory function monitored to pinpoint the offending allergen. Guinea pigs and mice have been used to test proteins for potential allergenicity. Animals are usually sensitized by the respiratory route and monitored for the development of cytophilic antibody (IgG1 in guinea pigs, IgE in mice) as well as increased respiratory rate and other changes in pulmonary function.
- #15.08.20134.04 Mб17Hastie T., Tibshirani R., Friedman J. - The Elements of Statistical Learning Data Mining, Inference and Prediction (2002)(en).djvu
- #
- #
- #
- #
- #
- #
- #
- #15.08.201315.44 Mб27Hudlicky M, Pavlath A.E. (eds.) - Chemistry of Organic Fluorine Compounds 2[c] A critical Review (1995)(en).djvu
- #
- #
