
- •CONTENTS
- •Preface
- •Contributors
- •1 Introduction to Toxicology
- •1.1 Definition and Scope, Relationship to Other Sciences, and History
- •1.1.2 Relationship to Other Sciences
- •1.1.3 A Brief History of Toxicology
- •1.3 Sources of Toxic Compounds
- •1.3.1 Exposure Classes
- •1.3.2 Use Classes
- •1.4 Movement of Toxicants in the Environment
- •Suggested Reading
- •2.1 Introduction
- •2.2 Cell Culture Techniques
- •2.2.1 Suspension Cell Culture
- •2.2.2 Monolayer Cell Culture
- •2.2.3 Indicators of Toxicity in Cultured Cells
- •2.3 Molecular Techniques
- •2.3.1 Molecular Cloning
- •2.3.2 cDNA and Genomic Libraries
- •2.3.3 Northern and Southern Blot Analyses
- •2.3.4 Polymerase Chain Reaction (PCR)
- •2.3.5 Evaluation of Gene Expression, Regulation, and Function
- •2.4 Immunochemical Techniques
- •Suggested Reading
- •3.1 Introduction
- •3.2 General Policies Related to Analytical Laboratories
- •3.2.1 Standard Operating Procedures (SOPs)
- •3.2.2 QA/QC Manuals
- •3.2.3 Procedural Manuals
- •3.2.4 Analytical Methods Files
- •3.2.5 Laboratory Information Management System (LIMS)
- •3.3 Analytical Measurement System
- •3.3.1 Analytical Instrument Calibration
- •3.3.2 Quantitation Approaches and Techniques
- •3.4 Quality Assurance (QA) Procedures
- •3.5 Quality Control (QC) Procedures
- •3.6 Summary
- •Suggested Reading
- •4 Exposure Classes, Toxicants in Air, Water, Soil, Domestic and Occupational Settings
- •4.1 Air Pollutants
- •4.1.1 History
- •4.1.2 Types of Air Pollutants
- •4.1.3 Sources of Air Pollutants
- •4.1.4 Examples of Air Pollutants
- •4.1.5 Environmental Effects
- •4.2 Water and Soil Pollutants
- •4.2.1 Sources of Water and Soil Pollutants
- •4.2.2 Examples of Pollutants
- •4.3 Occupational Toxicants
- •4.3.1 Regulation of Exposure Levels
- •4.3.2 Routes of Exposure
- •4.3.3 Examples of Industrial Toxicants
- •Suggested Reading
- •5 Classes of Toxicants: Use Classes
- •5.1 Introduction
- •5.2 Metals
- •5.2.1 History
- •5.2.2 Common Toxic Mechanisms and Sites of Action
- •5.2.3 Lead
- •5.2.4 Mercury
- •5.2.5 Cadmium
- •5.2.6 Chromium
- •5.2.7 Arsenic
- •5.2.8 Treatment of Metal Poisoning
- •5.3 Agricultural Chemicals (Pesticides)
- •5.3.1 Introduction
- •5.3.3 Organochlorine Insecticides
- •5.3.4 Organophosphorus Insecticides
- •5.3.5 Carbamate Insecticides
- •5.3.6 Botanical Insecticides
- •5.3.7 Pyrethroid Insecticides
- •5.3.8 New Insecticide Classes
- •5.3.9 Herbicides
- •5.3.10 Fungicides
- •5.3.11 Rodenticides
- •5.3.12 Fumigants
- •5.3.13 Conclusions
- •5.4 Food Additives and Contaminants
- •5.5 Toxins
- •5.5.1 History
- •5.5.2 Microbial Toxins
- •5.5.3 Mycotoxins
- •5.5.4 Algal Toxins
- •5.5.5 Plant Toxins
- •5.5.6 Animal Toxins
- •5.6 Solvents
- •5.7 Therapeutic Drugs
- •5.8 Drugs of Abuse
- •5.9 Combustion Products
- •5.10 Cosmetics
- •Suggested Reading
- •6 Absorption and Distribution of Toxicants
- •6.1 Introduction
- •6.2 Cell Membranes
- •6.3 Mechanisms of Transport
- •6.3.1 Passive Diffusion
- •6.4 Physicochemical Properties Relevant to Diffusion
- •6.4.1 Ionization
- •6.5 Routes of Absorption
- •6.5.1 Extent of Absorption
- •6.5.2 Gastrointestinal Absorption
- •6.5.3 Dermal Absorption
- •6.5.4 Respiratory Penetration
- •6.6 Toxicant Distribution
- •6.6.1 Physicochemical Properties and Protein Binding
- •6.7 Toxicokinetics
- •Suggested Reading
- •7 Metabolism of Toxicants
- •7.1 Introduction
- •7.2 Phase I Reactions
- •7.2.4 Nonmicrosomal Oxidations
- •7.2.5 Cooxidation by Cyclooxygenases
- •7.2.6 Reduction Reactions
- •7.2.7 Hydrolysis
- •7.2.8 Epoxide Hydration
- •7.2.9 DDT Dehydrochlorinase
- •7.3 Phase II Reactions
- •7.3.1 Glucuronide Conjugation
- •7.3.2 Glucoside Conjugation
- •7.3.3 Sulfate Conjugation
- •7.3.4 Methyltransferases
- •7.3.7 Acylation
- •7.3.8 Phosphate Conjugation
- •Suggested Reading
- •8 Reactive Metabolites
- •8.1 Introduction
- •8.2 Activation Enzymes
- •8.3 Nature and Stability of Reactive Metabolites
- •8.4 Fate of Reactive Metabolites
- •8.4.1 Binding to Cellular Macromolecules
- •8.4.2 Lipid Peroxidation
- •8.4.3 Trapping and Removal: Role of Glutathione
- •8.5 Factors Affecting Toxicity of Reactive Metabolites
- •8.5.1 Levels of Activating Enzymes
- •8.5.2 Levels of Conjugating Enzymes
- •8.5.3 Levels of Cofactors or Conjugating Chemicals
- •8.6 Examples of Activating Reactions
- •8.6.1 Parathion
- •8.6.2 Vinyl Chloride
- •8.6.3 Methanol
- •8.6.5 Carbon Tetrachloride
- •8.6.8 Acetaminophen
- •8.6.9 Cycasin
- •8.7 Future Developments
- •Suggested Reading
- •9.1 Introduction
- •9.2 Nutritional Effects
- •9.2.1 Protein
- •9.2.2 Carbohydrates
- •9.2.3 Lipids
- •9.2.4 Micronutrients
- •9.2.5 Starvation and Dehydration
- •9.2.6 Nutritional Requirements in Xenobiotic Metabolism
- •9.3 Physiological Effects
- •9.3.1 Development
- •9.3.2 Gender Differences
- •9.3.3 Hormones
- •9.3.4 Pregnancy
- •9.3.5 Disease
- •9.3.6 Diurnal Rhythms
- •9.4 Comparative and Genetic Effects
- •9.4.1 Variations Among Taxonomic Groups
- •9.4.2 Selectivity
- •9.4.3 Genetic Differences
- •9.5 Chemical Effects
- •9.5.1 Inhibition
- •9.5.2 Induction
- •9.5.3 Biphasic Effects: Inhibition and Induction
- •9.6 Environmental Effects
- •9.7 General Summary and Conclusions
- •Suggested Reading
- •10 Elimination of Toxicants
- •10.1 Introduction
- •10.2 Transport
- •10.3 Renal Elimination
- •10.4 Hepatic Elimination
- •10.4.2 Active Transporters of the Bile Canaliculus
- •10.5 Respiratory Elimination
- •10.6 Conclusion
- •Suggested Reading
- •11 Acute Toxicity
- •11.1 Introduction
- •11.2 Acute Exposure and Effect
- •11.3 Dose-response Relationships
- •11.4 Nonconventional Dose-response Relationships
- •11.5 Mechanisms of Acute Toxicity
- •11.5.1 Narcosis
- •11.5.2 Acetylcholinesterase Inhibition
- •11.5.3 Ion Channel Modulators
- •11.5.4 Inhibitors of Cellular Respiration
- •Suggested Reading
- •12 Chemical Carcinogenesis
- •12.1 General Aspects of Cancer
- •12.2 Human Cancer
- •12.2.1 Causes, Incidence, and Mortality Rates of Human Cancer
- •12.2.2 Known Human Carcinogens
- •12.3 Classes of Agents Associated with Carcinogenesis
- •12.3.2 Epigenetic Agents
- •12.4 General Aspects of Chemical Carcinogenesis
- •12.5 Initiation-Promotion Model for Chemical Carcinogenesis
- •12.6 Metabolic Activation of Chemical Carcinogens and DNA Adduct Formation
- •12.7 Oncogenes
- •12.8 Tumor Suppressor Genes
- •12.8.1 Inactivation of Tumor Suppressor Genes
- •12.8.2 p53 Tumor Suppressor Gene
- •12.9 General Aspects of Mutagenicity
- •12.10 Usefulness and Limitations of Mutagenicity Assays for the Identification of Carcinogens
- •Suggested Reading
- •13 Teratogenesis
- •13.1 Introduction
- •13.2 Principles of Teratology
- •13.3 Mammalian Embryology Overview
- •13.4 Critical Periods
- •13.5 Historical Teratogens
- •13.5.1 Thalidomide
- •13.5.2 Accutane (Isotetrinoin)
- •13.5.3 Diethylstilbestrol (DES)
- •13.5.4 Alcohol
- •13.6 Testing Protocols
- •13.6.1 FDA Guidelines for Reproduction Studies for Safety Evaluation of Drugs for Human Use
- •13.6.3 Alternative Test Methods
- •13.7 Conclusions
- •Suggested Reading
- •14 Hepatotoxicity
- •14.1 Introduction
- •14.1.1 Liver Structure
- •14.1.2 Liver Function
- •14.2 Susceptibility of the Liver
- •14.3 Types of Liver Injury
- •14.3.1 Fatty Liver
- •14.3.2 Necrosis
- •14.3.3 Apoptosis
- •14.3.4 Cholestasis
- •14.3.5 Cirrhosis
- •14.3.6 Hepatitis
- •14.3.7 Oxidative Stress
- •14.3.8 Carcinogenesis
- •14.4 Mechanisms of Hepatotoxicity
- •14.5 Examples of Hepatotoxicants
- •14.5.1 Carbon Tetrachloride
- •14.5.2 Ethanol
- •14.5.3 Bromobenzene
- •14.5.4 Acetaminophen
- •14.6 Metabolic Activation of Hepatotoxicants
- •Suggested Reading
- •15 Nephrotoxicity
- •15.1 Introduction
- •15.1.1 Structure of the Renal System
- •15.1.2 Function of the Renal System
- •15.2 Susceptibility of the Renal System
- •15.3 Examples of Nephrotoxicants
- •15.3.1 Metals
- •15.3.2 Aminoglycosides
- •15.3.3 Amphotericin B
- •15.3.4 Chloroform
- •15.3.5 Hexachlorobutadiene
- •Suggested Reading
- •16 Toxicology of the Nervous System
- •16.1 Introduction
- •16.2 The Nervous system
- •16.2.1 The Neuron
- •16.2.2 Neurotransmitters and their Receptors
- •16.2.3 Glial Cells
- •16.3 Toxicant Effects on the Nervous System
- •16.3.1 Structural Effects of Toxicants on Neurons
- •16.3.2 Effects of Toxicants on Other Cells
- •16.4 Neurotoxicity Testing
- •16.4.1 In vivo Tests of Human Exposure
- •16.4.2 In vivo Tests of Animal Exposure
- •16.4.3 In vitro Neurochemical and Histopathological End Points
- •16.5 Summary
- •Suggested Reading
- •17 Endocrine System
- •17.1 Introduction
- •17.2 Endocrine System
- •17.2.1 Nuclear Receptors
- •17.3 Endocrine Disruption
- •17.3.1 Hormone Receptor Agonists
- •17.3.2 Hormone Receptor Antagonists
- •17.3.3 Organizational versus Activational Effects of Endocrine Toxicants
- •17.3.4 Inhibitors of Hormone Synthesis
- •17.3.5 Inducers of Hormone Clearance
- •17.3.6 Hormone Displacement from Binding Proteins
- •17.4 Incidents of Endocrine Toxicity
- •17.4.1 Organizational Toxicity
- •17.4.2 Activational Toxicity
- •17.4.3 Hypothyroidism
- •17.5 Conclusion
- •Suggested Reading
- •18 Respiratory Toxicity
- •18.1 Introduction
- •18.1.1 Anatomy
- •18.1.2 Cell Types
- •18.1.3 Function
- •18.2 Susceptibility of the Respiratory System
- •18.2.1 Nasal
- •18.2.2 Lung
- •18.3 Types of Toxic Response
- •18.3.1 Irritation
- •18.3.2 Cell Necrosis
- •18.3.3 Fibrosis
- •18.3.4 Emphysema
- •18.3.5 Allergic Responses
- •18.3.6 Cancer
- •18.3.7 Mediators of Toxic Responses
- •18.4 Examples of Lung Toxicants Requiring Activation
- •18.4.1 Introduction
- •18.4.2 Monocrotaline
- •18.4.3 Ipomeanol
- •18.4.4 Paraquat
- •18.5 Defense Mechanisms
- •Suggested Reading
- •19 Immunotoxicity
- •19.1 Introduction
- •19.2 The Immune System
- •19.3 Immune Suppression
- •19.4 Classification of Immune-Mediated Injury (Hypersensitivity)
- •19.5 Effects of Chemicals on Allergic Disease
- •19.5.1 Allergic Contact Dermatitis
- •19.5.2 Respiratory Allergens
- •19.5.3 Adjuvants
- •19.6 Emerging Issues: Food Allergies, Autoimmunity, and the Developing Immune System
- •Suggested Reading
- •20 Reproductive System
- •20.1 Introduction
- •20.2 Male Reproductive Physiology
- •20.3 Mechanisms and Targets of Male Reproductive Toxicants
- •20.3.1 General Mechanisms
- •20.3.2 Effects on Germ Cells
- •20.3.3 Effects on Spermatogenesis and Sperm Quality
- •20.3.4 Effects on Sexual Behavior
- •20.3.5 Effects on Endocrine Function
- •20.4 Female Reproductive Physiology
- •20.5 Mechanisms and Targets of Female Reproductive Toxicants
- •20.5.1 Tranquilizers, Narcotics, and Social Drugs
- •20.5.2 Endocrine Disruptors (EDs)
- •20.5.3 Effects on Germ Cells
- •20.5.4 Effects on the Ovaries and Uterus
- •20.5.5 Effects on Sexual Behavior
- •Suggested Reading
- •21 Toxicity Testing
- •21.1 Introduction
- •21.2 Experimental Administration of Toxicants
- •21.2.1 Introduction
- •21.2.2 Routes of Administration
- •21.3 Chemical and Physical Properties
- •21.4 Exposure and Environmental Fate
- •21.5 In vivo Tests
- •21.5.1 Acute and Subchronic Toxicity Tests
- •21.5.2 Chronic Tests
- •21.5.3 Reproductive Toxicity and Teratogenicity
- •21.5.4 Special Tests
- •21.6 In vitro and Other Short-Term Tests
- •21.6.1 Introduction
- •21.6.2 Prokaryote Mutagenicity
- •21.6.3 Eukaryote Mutagenicity
- •21.6.4 DNA Damage and Repair
- •21.6.5 Chromosome Aberrations
- •21.6.6 Mammalian Cell Transformation
- •21.6.7 General Considerations and Testing Sequences
- •21.7 Ecological Effects
- •21.7.1 Laboratory Tests
- •21.7.2 Simulated Field Tests
- •21.7.3 Field Tests
- •21.8 Risk Analysis
- •21.9 The Future of Toxicity Testing
- •Suggested Reading
- •22 Forensic and Clinical Toxicology
- •22.1 Introduction
- •22.2 Foundations of Forensic Toxicology
- •22.3 Courtroom Testimony
- •22.4.1 Documentation Practices
- •22.4.2 Considerations for Forensic Toxicological Analysis
- •22.4.3 Drug Concentrations and Distribution
- •22.5 Laboratory Analyses
- •22.5.1 Colorimetric Screening Tests
- •22.5.2 Thermal Desorption
- •22.5.6 Enzymatic Immunoassay
- •22.6 Analytical Schemes for Toxicant Detection
- •22.7 Clinical Toxicology
- •22.7.1 History Taking
- •22.7.2 Basic Operating Rules in the Treatment of Toxicosis
- •22.7.3 Approaches to Selected Toxicoses
- •Suggested Reading
- •23 Prevention of Toxicity
- •23.1 Introduction
- •23.2 Legislation and Regulation
- •23.2.1 Federal Government
- •23.2.2 State Governments
- •23.2.3 Legislation and Regulation in Other Countries
- •23.3 Prevention in Different Environments
- •23.3.1 Home
- •23.3.2 Workplace
- •23.3.3 Pollution of Air, Water, and Land
- •23.4 Education
- •Suggested Reading
- •24 Human Health Risk Assessment
- •24.1 Introduction
- •24.2 Risk Assessment Methods
- •24.2.2 Exposure Assessment
- •24.2.3 Dose Response and Risk Characterization
- •24.3 Noncancer Risk Assessment
- •24.3.1 Default Uncertainty and Modifying Factors
- •24.3.2 Derivation of Developmental Toxicant RfD
- •24.3.3 Determination of RfD and RfC of Naphthalene with the NOAEL Approach
- •24.3.4 Benchmark Dose Approach
- •24.3.5 Determination of BMD and BMDL for ETU
- •24.3.6 Quantifying Risk for Noncarcinogenic Effects: Hazard Quotient
- •24.3.7 Chemical Mixtures
- •24.4 Cancer Risk Assessment
- •24.5 PBPK Modeling
- •Suggested Reading
- •25 Analytical Methods in Toxicology
- •25.1 Introduction
- •25.2 Chemical and Physical Methods
- •25.2.1 Sampling
- •25.2.2 Experimental Studies
- •25.2.3 Forensic Studies
- •25.2.4 Sample Preparation
- •25.2.6 Spectroscopy
- •25.2.7 Other Analytical Methods
- •Suggested Reading
- •26 Basics of Environmental Toxicology
- •26.1 Introduction
- •26.2 Environmental Persistence
- •26.2.1 Abiotic Degradation
- •26.2.2 Biotic Degradation
- •26.2.3 Nondegradative Elimination Processes
- •26.3 Bioaccumulation
- •26.4 Toxicity
- •26.4.1 Acute Toxicity
- •26.4.2 Mechanisms of Acute Toxicity
- •26.4.3 Chronic Toxicity
- •26.4.5 Abiotic and Biotic Interactions
- •26.5 Conclusion
- •Suggested Reading
- •27.1 Introduction
- •27.2 Sources of Toxicants to the Environment
- •27.3 Transport Processes
- •27.3.1 Advection
- •27.3.2 Diffusion
- •27.4 Equilibrium Partitioning
- •27.5 Transformation Processes
- •27.5.1 Reversible Reactions
- •27.5.2 Irreversible Reactions
- •27.6 Environmental Fate Models
- •Suggested Reading
- •28 Environmental Risk Assessment
- •28.1 Introduction
- •28.2 Formulating the Problem
- •28.2.1 Selecting Assessment End Points
- •28.2.2 Developing Conceptual Models
- •28.2.3 Selecting Measures
- •28.3 Analyzing Exposure and Effects Information
- •28.3.1 Characterizing Exposure
- •28.3.2 Characterizing Ecological Effects
- •28.4 Characterizing Risk
- •28.4.1 Estimating Risk
- •28.4.2 Describing Risk
- •28.5 Managing Risk
- •Suggested Reading
- •29 Future Considerations for Environmental and Human Health
- •29.1 Introduction
- •29.2 Risk Management
- •29.3 Risk Assessment
- •29.4 Hazard and Exposure Assessment
- •29.5 In vivo Toxicity
- •29.6 In vitro Toxicity
- •29.7 Biochemical and Molecular Toxicology
- •29.8 Development of Selective Toxicants
- •Glossary
- •Index

 CHAPTER 17
CHAPTER 17
Endocrine System
GERALD A. LEBLANC
17.1INTRODUCTION
Among the various organ systems of the body, the endocrine system is somewhat unique. While most systems are associated with a specific physiological task (respiration, reproduction, excretion, etc.), the endocrine system functions to regulate many of the activities associated with these other systems. Accordingly the endocrine system is integral to the maintenance of total normal bodily function (homeostasis), and disruption of normal endocrine function by exogenous chemicals can result in multiple, diverse, and dire consequences. Toxicity to the endocrine system is most commonly associated with altered development, growth, maturation, and reproduction (Table 17.1). However, endocrine toxicity also can present as gastro-intestinal dysfunction, malaise, neurological and other disorders (Table 17.1). This is why endocrine toxicity often can be misconstrued as toxicity to some other endocrine-regulated system of the body.
The endocrine system, as an authentic target of chemical toxicity, tragically entered the limelight as a consequence of the widespread use of the drug diethylstilbestrol (DES). DES, a nonsteroidal synthetic estrogen, was prescribed to pregnant woman from the 1940s to the 1960s as a prophylactic against miscarriage (see Section 17.4.1). Following the discovery of the endocrine toxicity of this drug, many additional drugs and environmental chemicals have been shown to mimic the action of hormones or interfere with their hormonal function. These activities often have been clearly shown, in laboratory studies, to result in endocrine-related toxicity. In some instances drug use or exposure to ambient environmental chemicals has been shown to result in endocrine toxicity. Such examples will be presented at the end of this chapter.
17.2ENDOCRINE SYSTEM
The endocrine system can be broadly described as an assemblage of organs (glands) that produce chemical messengers (hormones) that regulate various bodily functions. The bodily functions regulated by the endocrine system can be categorized as those
A Textbook of Modern Toxicology, Third Edition, edited by Ernest Hodgson
ISBN 0-471-26508-X Copyright 2004 John Wiley & Sons, Inc.
299

300 ENDOCRINE SYSTEM
Table 17.1 Processes Regulated by Some Hormones of the Endocrine System Susceptible to Disruption by Endocrine Toxicants
Hormone |
|
|
|
Group |
Example |
Origin |
Regulated Process |
|
|
|
|
Androgens |
Testosterone |
Testes, adrenals |
Sexual differentiation, fertility, |
|
|
|
secondary sex characteristics, |
|
|
|
sexual function, libido |
Estrogens |
17β-Estradiol |
Ovaries, testes |
Sexual differentiation, fertility, |
|
|
|
secondary sex characteristics, |
|
|
|
bone density maintenance, |
|
|
|
blood coagulation |
Glucocorticoids |
Cortisol |
Adrenals |
Bone formation, wound healing, |
|
|
|
growth, development |
Thyroid hormones |
Thyroxine |
Thyroid gland |
Fetal brain and bone development, |
|
|
|
oxygen consumption, gut |
|
|
|
motility |
|
|
|
|
involved in the maintenance of homeostasis and those involved in physiological progression. Functions regulated by the endocrine system resulting in homeostasis include maintenance of the reproductive system, energy production, and metabolism. Functions regulated by the endocrine system resulting in physiological progression include fetal development, growth, and maturation. Endocrine processes related to physiological progression historically have received the greatest attention in endocrine toxicology and will be emphasized in this chapter.
Both the maintenance of homeostasis and the regulation of physiological progression require that the endocrine system detect signals, either external or internal, and transduce these signals to the appropriate target sites within the body. These target sites then respond in the appropriate manner to maintain homeostasis or institute change related to development, maturation, and so on. In many species these initial signals are of external origin. For example, many species initiate reproductive maturation in response to changes in environmental temperature and day length. Reproductively mature organisms often respond to external visual or olfactory stimuli produced by sexually receptive individual to initiate sexual behavior.
The signal to be transduced by the endocrine system initiates in the central nervous system. In mammals, the hypothalamus commonly initiates the endocrine signaling pathway by secreting peptide hormones. These neuro-endocrine hormones can be rapidly synthesized, secreted, and degraded to allow near-instantaneous, short-lived responses to the stimulatory signal. Accordingly they can be present in the body in pulses and secretory rhythms that often contribute to their signaling function. For example, the hypothalamic peptide hormones “growth hormone releasing hormone” (GHRH) and somatostatin are secreted in an alternating pulsatile fashion. Both hormones target the pituitary gland, though GHRH stimulates and somatostatin inhibits growth hormone secretion by the pituitary. As a result the secretory pattern of the secondary hormone messenger in this cascade, growth hormone, is highly controlled. Disruption of this rhythm in rodent models can alter hepatic enzyme expression and other dynamic processes. Disruption of the growth hormone secretory rhythm associated with sleep has been shown to interfere with normal growth in children. Hormone

ENDOCRINE SYSTEM |
301 |
secretory rhythms have been associated with other physiological processes including sleep, sexual behavior, and ovulation.
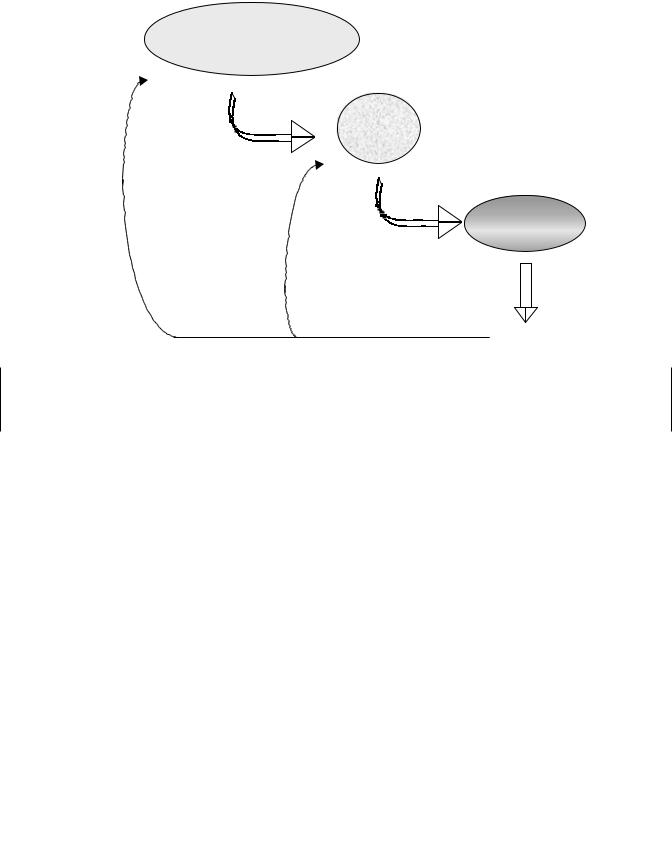
Endocrine signaling pathways from the central nervous system to the target organ typically occur along axes (Figure 17.1). An axis is defined by the endocrine glands that produce signaling hormones along the cascade (i.e., hypothalamic–pituitary–gonadal axis), and sometimes, a terminal target organ of the signaling pathway (i.e., hypotha- lamic–pituitary–gonadal–hepatic axis).
Endocrine signaling cascades offer several advantages over a single hormone signaling strategy. Cascades provide several sites at which the signal can be regulated thus ensuring maintenance of the appropriate endocrine signal (Figure 17.2). For example, testosterone is secreted by the testis but regulates its own secretion by acting upstream in the axis at the pituitary gland and hypothalamic gland. Signaling cascades also
Hypothalamus
|
Thyrotropin- |
|
|
Gonadotropin- |
Releasing |
Adrenocorticotropic- |
|
Hormone |
|||
Releasing |
|||
Releasing |
|||
|
|||
|
Hormone |
||
Hormone |
|
||
|
|
||
|
Pituitary |
|
|
|
gland |
|
|
Gonadotropin |
|
Adrenocorticotropin |
|
|
Thyrotropin |
|
|
Gonads |
|
Adrenal gland |
Thyroid gland
Corticosteroids
Sex steroids
Thyroxine
Figure 17.1 Some major neuro–endocrine axes that transduce endocrine signals to target organs. Neuro–endocrine signaling is initiated by the secretion of releasing hormones, or in some instances inhibiting hormones, that regulate secretion of the secondary hormone signal by the pituitary. Pituitary hormones then regulate secretion of the tertiary hormone, often a steroid hormone, by the appropriate endocrine gland. The tertiary hormones then stimulate gene transcription at target organs.
302 ENDOCRINE SYSTEM
Hypothalamus
Pituitary
gland
Luteinizing Hormone
Releasing Hormone
Level II |
Gonads |
|
Luteinizing
Hormone
Level I
Sex Steroid Hormones
Feedback regulation of sex steroid hormone synthesis
Figure 17.2 The hypothalamic–pituitary–gonadal axis. Endocrine signaling cascades provide multiple sites for regulation and ensure optimum signaling.
utilize multiple hormones with differing properties to contribute to the process. Peptide hormones are commonly the intermediate messengers along a signaling cascade, while the terminal hormone is often of nonpeptide origin (i.e., steroids). Peptide hormones offer advantages as intermediate messengers in that they can be rapidly synthesized and degraded (i.e., turned “on” and “off”). Peptide hormones also do not require cell entry to elicit activity but rather bind to cell surface receptors. This facilitates a rapid physiological response to the hormone. Steroid and other nonpeptide hormones are typically more stable, they are maintained in circulation at a relatively constant, physiologically appropriate level, they can be stored as precursor molecules or apolar conjugates, they can be mobilized as polar conjugates, and most often, they require cell entry to interact with its receptor and elicit a response. Accordingly the nonpeptide terminal hormones offer the advantages of constant availability but lack the advantages of rapid modulation.
17.2.1Nuclear Receptors
Toxicologically the function of the terminal hormones of endocrine cascades (i.e., steroid, retinoid, thyroid hormones) appear to be most susceptible to disruption by chemicals. This is because many foreign molecules share sufficient characteristics with these hormone molecules to allow binding to the nuclear receptors of these hormones in either an agonistic or antagonistic fashion. The binding of the xenobiotic to the

ENDOCRINE SYSTEM |
303 |
receptor results in aberrant receptor function with associated toxicological outcome. The nuclear receptors are so called since these receptors initiate their classical physiological responses within the cell nucleus. Cell surface receptors to peptide hormones, on the other hand, can likely discriminate between peptide molecules and nonpeptide xenobiotics thus minimizing the likelihood of interaction and associated disruption of function.
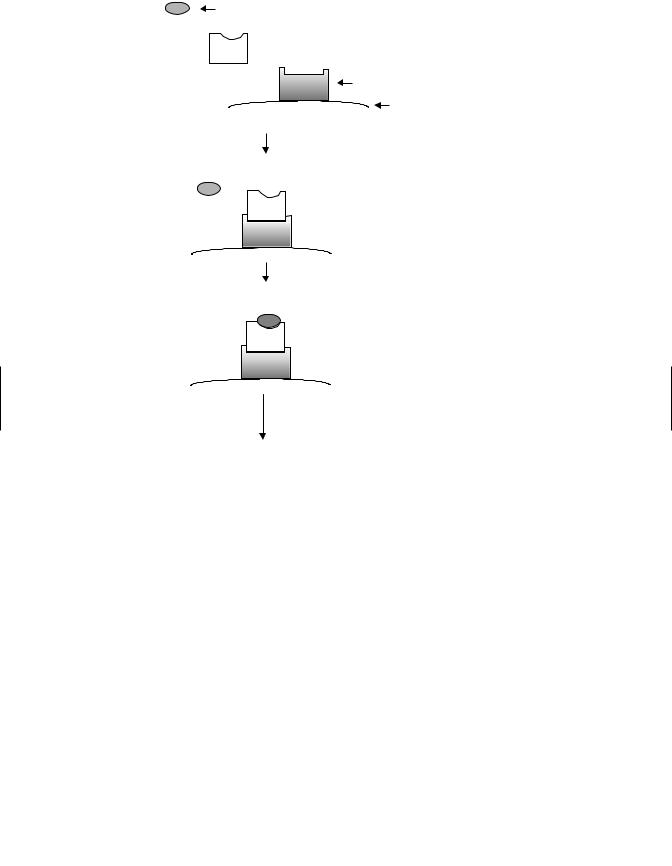
The nuclear receptor superfamily consists of members of the steroid receptor family and the thyroid receptor family (Figure 17.3). Members of these two receptor families are distinct in many structural and functional attributes. Steroid receptor family members typically exist in the extranuclear matrix of the cell in association with various accessory proteins (hsp90, hsp70, hsp56). These accessory proteins stabilize the receptor molecule and help maintain the molecule’s integrity. Binding of hormone ligand to the receptor protein stimulates dissociation with the accessory proteins, homodimerization of two receptor molecules, and nuclear localization (Figure 17.4). Here the receptor complex interacts with hormone response elements (Table 17.2) that are associated with hormone responsive genes, and transcription of these genes is regulated.
-androgen receptor |
-thyroid hormone receptor |
|
-progesterone receptor |
||
-retinoic acid receptor |
||
-glucocorticoid receptor |
||
-retinoid X receptor |
||
-mineralocorticoid receptor |
||
-vitamin D receptor |
||
-others |
||
-others |
||
|
Figure 17.3 The nuclear receptor superfamily. Steroid receptor family members and thyroid receptor family members differ in several structural and functional properties. The estrogen receptors share properties with both steroid and thyroid receptor families and are likely an evolutionary precursor to both families.
Table 17.2 DNA Recognition Sequences Utilized by
Steroid, Estrogen, and Thyroid Receptor Family
Members
Receptor |
|
Family |
Recognition Sequence |
|
|
Steroid |
AGAACA . . . TGTTCT |
Estrogen |
AGGTCA . . . TGACCT |
Thyroid |
AGGTCA . . . AGGTCA |
Note: Recognition sequences are split by spacer nucleotides, which are denoted by the dots.

304 ENDOCRINE SYSTEM
(a) |
|
(b) |
|
|
|
(c) |
|
|
|
(d) |
|
|
(e) |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Nuclear Membrane
Cell Surface Membrane
Figure 17.4 Intracellular steroid receptor activation by hormone ligands. (a) Steroid hormones diffuse across the cell membrane into the cell. (b) Steroid hormone receptors in the basal state bound to accessory proteins. (c) Steroid hormones bind to receptors and accessory proteins are dissociated from the receptors. (d) Hormone : receptor complexes dimerize. (e) Dimer complexes enter the nucleus and initiate transcription of responsive genes.
In contrast to the steroid receptor family, members of the thyroid receptor family typically do not associate with accessory proteins and are not localized to the extranucleus matrix. Rather, these receptors exist in the basal state associated with chromatin in the cell nucleus. When bound by hormone ligand, thyroid receptor family members dissociate from the chromatin and typically form heterodimeric combinations with the retinoid-X receptor (RXR). RXR also is capable of homodimerization in association with its ligand 9-cis retinoic acid. Thus high 9-cis retinoic acid levels apparently promote homodimerization, and low levels are permissive of heterodimerization of RXR with activation by the partner ligand.
The estrogen receptor shares structural and functional attributes of both nuclear receptor families. For example, the estrogen receptor resides in the cell nucleus, but it associates with accessory proteins rather than chromatin. The estrogen receptor shares a high degree of sequence homology with the thyroid receptor family members but does not heterodimerize with RXR. Rather, active estrogen receptor exists as a homodimer. The observation that the estrogen receptor shares attributes of both nuclear receptor families supports nucleotide sequence evidence that the estrogen receptor is an ancestral precursor to both receptor families.
17.2.2Membrane-Bound Steroid Hormone Receptors
Some cellular responses occur too rapidly following steroid hormone exposure to involve the multi-step process of nuclear receptor activation. For example, 17β-estradiol can rapidly stimulate adenylate cyclase and cause a near-instantaneous increase in intracellular cAMP in cultured prostate cells. These effects are mediated by the interaction of steroid hormones with cell surface proteins.
ENDOCRINE SYSTEM |
305 |
Steroid Hormone
 SHBG
SHBG
SHBG Receptor
Cell Surface
Membrane
Signal Transduction
Figure 17.5 Endocrine signaling pathway involving steroid hormone, sex hormone binding globulin (SHBG), and the SHBG receptor.
Due to their lipophilic nature, steroid hormones are mobilized in the circulatory system by transfer proteins. Sex hormone-binding globulin (SHBG) is one such transfer protein that binds testosterone, 17β-estradiol, and other sex steroids. Roughly half of circulating testosterone and 17β-estradiol is bound to SHBG. Receptors exist on the surface of some cells that are capable of binding unliganded SHBG (Figure 17.5). Unliganded SHBG, which is bound to the cell surface receptor, can subsequently bind steroid hormone. Binding of an appropriate hormone to the SHBG then stimulates a signal-transduction pathway within the cell. Some steroid hormones (17β-estradiol, 5α-androstan-3α,17β-diol) function as SHBG : SHBG-receptor agonists, while others (testosterone, 5α-dihydrotestosterone) function as antagonists. Interestingly 5α- androstan-3α,17β-diol had previously been considered an inactivation product of the potent androgen 5α-dihydrotestosterone (DHT). Studies in human prostate cells have shown that activation of this SHBG-dependent pathway stimulates DNA synthesis and cell growth. These observations, in combination with studies in dogs that have shown 5α-androstan-3α,17β-diol to stimulate benign prostatic hyperplasia, have led to
- #15.08.20134.04 Mб17Hastie T., Tibshirani R., Friedman J. - The Elements of Statistical Learning Data Mining, Inference and Prediction (2002)(en).djvu
- #
- #
- #
- #
- #
- #
- #
- #15.08.201315.44 Mб27Hudlicky M, Pavlath A.E. (eds.) - Chemistry of Organic Fluorine Compounds 2[c] A critical Review (1995)(en).djvu
- #
- #
