
- •Chemotherapy
- •Cytotoxic antibiotics and antimetabolites
- •The purine pathway to chemotherapy
- •Good drugs and bad
- •Combination chemotherapy
- •Alternative targets for cancer therapy: towards a scientific rationale
- •Inhibiting the EGF family of receptor kinases
- •The antibody approach: trastuzumab
- •The tyrosine kinase inhibitor approach
- •Erlotinib
- •Gefitinib
- •Imatinib: chronic myeloid leukaemia and the Bcr-Abl fusion story
- •Development of imatinib, inhibitor of c-Abl
- •Why is treatment of CML successful?
- •Molecular mechanism of inhibition by imatinib
- •Resistance due to mutations at different sites in the Abl moiety of Bcr-Abl
- •Other signal transduction components targeted for therapeutic intervention
- •Towards a different approach in testing cancer drugs?
- •References
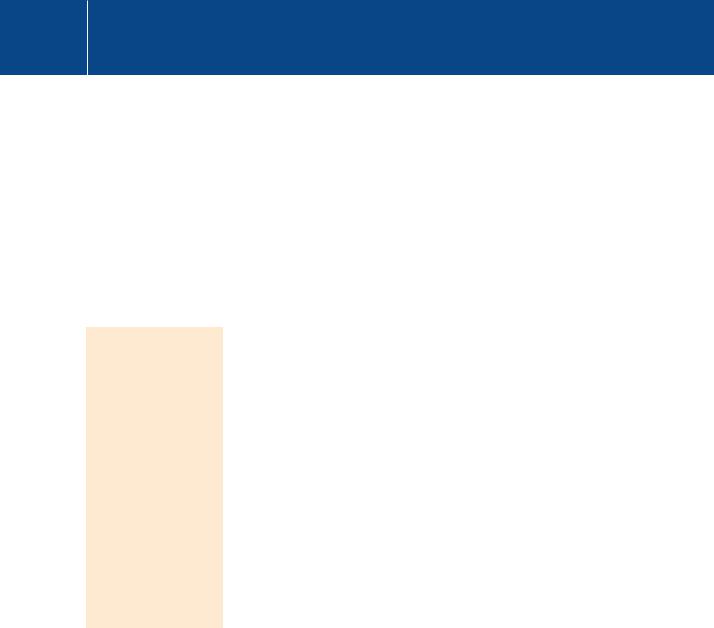
Signal Transduction
If the statins (lovastatin, simvastatin, atorvastatin etc., inhibitors of HMGCoA reductase) have offered little benefit in the treatment of cancer, they are regarded almost as wonder drugs in the prevention of atherosclerosis and stroke. Polyisoprene (squalene) formation
is a key step in the formation of cholesterol and the membrane anchors (farnesyl and geranylgeranyl) that link the Ras GTPases to the plasma membrane (see page 105).
advantage that the kinase is less likely to escape inhibition. The general trend now is to shift attention away from single, highly selective inhibitors towards broader spectrum compounds or to cocktails of specific inhibitors acting
in different ways. These approaches open up a range of new therapeutic promise but also the possibility of unacceptable adverse effects.
Other signal transduction components targeted for therapeutic intervention
There are many other components of signal transduction pathways that might be regarded as possible targets for drug therapy. The Ras-MAP kinase pathway has been studied extensively and its components, H-, K- and N-Ras, B-Raf, and MEK1, have been identified as human oncogenes. In particular, it has been suggested that because Ras-mediated cell transformation demands membrane attachment (through polyisoprenoid chains: see page 105), prevention of this post translational modification might stop cancer growth.52 However, none of the drugs developed for this purpose have shown any benefit in the cancer field, and in many cases mutated Ras escapes their effect. Inhibitors of B-Raf share the same misfortune though the jury is still out for the inhibitors of MEK. Three highly selective MEK inhibitors have recently reached the stage of clinical trials.53,54 These differ from many other kinase inhibitors in that they do not compete with ATP but bind close to the catalytic cleft, thereby impeding access of the activation segment of ERK (the unique substrate of MEK) (Figure 23.10).
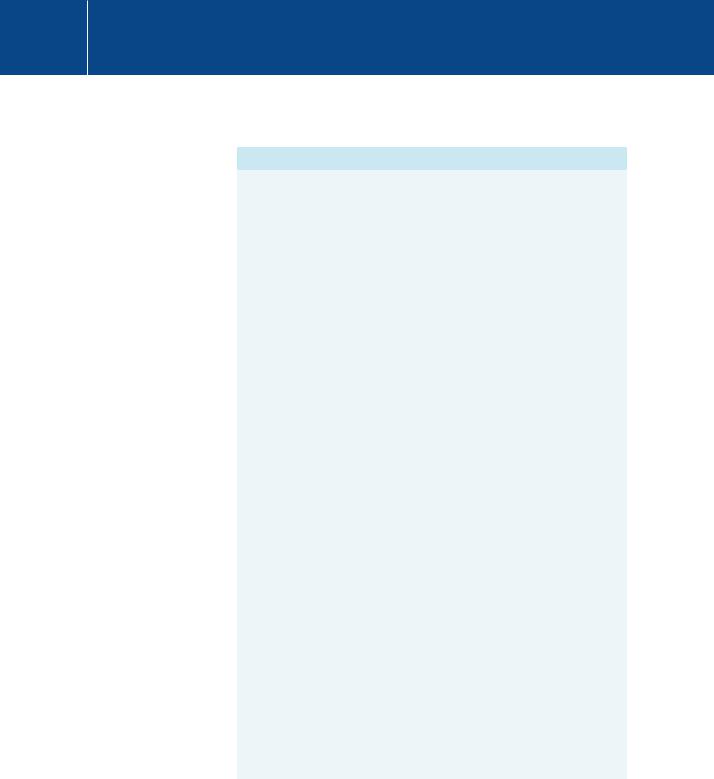
Inhibitors targeting enzymes of the PI 3-kinase pathway (PI 3-kinase, PKB, and mTOR) that controls protein synthesis, cell cycle progression and, importantly, cell survival, also show some promise.55 Another possible target is Hsp90,56 involved in protein folding. It is highly expressed in tumours and appears to play a role in the activation and localization of kinases such as Raf, Cdk4/6, v-Src, or PKB. Some of the compounds developed to inhibit protein kinases are listed in Table 23.3.
Towards a different approach in testing cancer drugs?
The general opinion is that with the single exception of imatinib, signal transduction blockers have yet to realize a real breakthrough in cancer treatment. Whereas the limiting factor to advances in this field used to be the definition of drug targets, today it seems to be the design of appropriate and effective clinical trials. We have many compounds, all based on good scientific rationales, all seemingly safe and which offer promising effects in in vitro (cell culture) and in vivo (mouse xenograft) studies.
754
Targeting Transduction Pathways for Research and Medical Intervention
Fig 23.10 Inhibition of MEK2.
PD3334581 inhibits MEK2 by occupying the substrate entry site. It also positions the activation segment in front of the catalytic cleft, making access still more difficult. Moreover, the C-helix is turned away so that the interaction of E114 with K97, essential for the correct positioning of ATP, is prevented (see also Figure 18.6, page 553 for the activation of PKB). NB: The disordered segment of the protein is represented as red dotted line, with only one of the two ‘activation’ serines (S218) visible (1s9i54).
There are two important obstacles to be faced in the clinical testing of cancer drugs. The first concerns the choice of patients. The old motto ‘one drug suits all’ is perhaps not appropriate for cancer therapy, not even within carefully defined classes of tumours. Precise predictive markers are needed in order to improve the choice of patients to be included in the trialling of novel compounds. To illustrate this, the two most useful predictors of the responsiveness of breast cancers are the expression of oestrogen receptors (ER) for anti-oestrogen therapy and HER-2 amplification for trastuzumab
(Herceptin) therapy. But these only have positive predictive values of 30–50%. At best, only one in two of the selected patients shows a good response to the therapy. Their negative predictive value, at 95%, is more convincing.57 It is not unthinkable that the compounds currently being rejected have yet to find the right subjects in which to express their therapeutic potential.
The second obstacle concerns the time span of testing. At an early stage it is essential to have good prognostic markers so as to be able to get a realistic impression of the effectiveness of a drug. Once a drug has been accepted
755
Signal Transduction
Table 23.3 Some compounds targeting signal transduction pathways under scrutiny in clinical trials
Kinome branch |
compound |
Known targets |
|
|
|
TK |
AZD-2171 |
VEGFR |
|
vandetanib ZD6474 |
VEGR/EGFR |
|
vatalanib PTK787 |
VEGFR, PDGFR, c-Kit |
|
axitnib AG-013736 |
VEGFR, PDGFR |
|
lapatinib (Tykerb) |
EGFR, ERBB2 |
|
GW572016 |
|
|
CP-751871 (antibody) |
IGFR |
|
dastinib BMS-354825 |
c-Src, c-Abl |
|
AMN-107 |
c-Abl |
|
dasatinib (Sprycel) |
c-Abl, PDFR, c-Kit, c-Src |
|
|
members |
|
nilotinib (Tasigna) |
c-Abl, PDGFR, c-Kit |
|
sunitinib (Sutent) |
VEGFR, c-KIT, PDGFR, |
|
|
Ret, CSFR, Flt3 |
|
sorafenib (Nexavar) |
VEGFR2, PDGFR, c-Kit, |
|
|
Flt3, A-Raf, B-Raf |
|
bevacizumab (Avastin) |
VEGFA |
|
antibody |
|
|
panitumumab |
EGFR |
|
(antibody) |
|
|
|
|
CMGC (CDK, MAPK, GSK |
flavopiridol |
Pan-CDK |
and Cdc-like kinases) |
PD332991 |
CDK4 |
|
||
|
|
|
STE (MAP2K, MAP3K, |
BAY 43-9006 |
C-Raf |
MAP4K) |
PD0325901 |
MEK |
|
||
|
PD184352 (CI-1040) |
MEK |
|
PD318088 |
MEK1 |
|
PD334581 |
MEK2 |
|
|
|
AGC |
rapamycin |
mTOR |
|
|
|
756
Targeting Transduction Pathways for Research and Medical Intervention
for general clinical application, these markers remain useful in order to verify whether or not tumours are responding.57 The design of clinical trials should provide the opportunity to assess retrospectively why a particular compound failed to give the expected result in a particular patient.53 Unfortunately,
the pressure exerted both by patients and by the industry to advance new blockbuster drugs limits the scale and value of such retrospective analytical approaches.58
For more information about clinical trials we suggest: http://www. cancer.gov/clinicaltrials/ learning
List of Abbreviations
Abbreviation |
Full name/description |
SwissProt entry |
Other names, OMIM link |
|
|
|
|
Bcr |
breakpoint cluster region |
P11274 |
|
|
|
|
|
B-Raf |
B-type rat fibrosarcoma |
P15056 |
BRaf1, MAP3K |
|
|
|
|
c-Abl |
cellular homologue of Abelson viral |
P00519 |
|
|
product |
|
|
|
|
|
|
c-KIT |
kitten (oncogene from Hardy- |
P10721 |
mast/stem cell growth factor |
|
Zuckerman 4 feline sarcoma virus) |
|
receptor |
|
|
|
|
CML |
chronic myelogenous leukaemia |
|
MIM:608232 |
|
|
|
|
EGFR |
epidermal growth factor receptor |
P00533 |
|
|
(ERBB1) |
|
|
|
|
|
|
ERBB2 |
erythroblastosis-B2 |
P04626 |
Her-2, Neu, MIM:164870 |
|
|
|
|
ERK1 |
extracellular signal-regulated |
P27361 |
p44-MAPK, MK03 |
|
kinase-1 |
|
|
|
|
|
|
Flt3 |
fetal liver tyrosine protein kinase-3 |
P36888 |
CD135, stem cell tyrosine kinase |
|
|
|
(STK1) |
|
|
|
|
GIST |
gastrointestinal stromal tumour |
|
MIM;606764 |
|
|
|
|
HER2 |
human EGF receptor-2 |
P04626 |
ERBB2, neu, MIM:164870 |
|
|
|
|
Hsp90 |
heat shock protein of 90 kDa |
P07900 |
renal carcinoma-induced antigen |
|
|
|
|
MEK1 |
MAPK ERK kinase-1 |
Q02750 |
MAP2K |
|
|
|
|
neu |
oncogene from neuro/glioblastoma |
P04626 |
HER-2, ERBB2, MIM:164870 |
|
cells |
|
|
|
|
|
|
PDGFR-A |
platelet-derived growth factor |
P16234 |
|
|
receptor-A |
|
|
|
|
|
|
Src |
sarcoma |
|
|
|
|
|
|
VEGFR |
vascular endothelial growth factor |
P17948 |
Fems-like tyrosine kinase-1 (FLT1) |
|
receptor type 1 |
|
|
|
|
|
|
757
