
- •Preface
- •Acknowledgments
- •Contents
- •1.1 Introduction
- •1.2 Normal Embryology
- •1.3 Abnormalities of the Kidney
- •1.3.1 Renal Agenesis
- •1.3.2 Renal Hypoplasia
- •1.3.3 Supernumerary Kidneys
- •1.3.5 Polycystic Kidney Disease
- •1.3.6 Simple (Solitary) Renal Cyst
- •1.3.7 Renal Fusion and Renal Ectopia
- •1.3.8 Horseshoe Kidney
- •1.3.9 Crossed Fused Renal Ectopia
- •1.4 Abnormalities of the Ureter
- •1.5 Abnormalities of the Bladder
- •1.6 Abnormalities of the Penis and Urethra in Males
- •1.7 Abnormalities of Female External Genitalia
- •Further Reading
- •2.1 Introduction
- •2.2 Pathophysiology
- •2.3 Etiology of Hydronephrosis
- •2.5 Clinical Features
- •2.6 Investigations and Diagnosis
- •2.7 Treatment
- •2.8 Antenatal Hydronephrosis
- •Further Reading
- •3.1 Introduction
- •3.2 Embryology
- •3.3 Pathophysiology
- •3.4 Etiology of PUJ Obstruction
- •3.5 Clinical Features
- •3.6 Diagnosis and Investigations
- •3.7 Management of Newborns with PUJ Obstruction
- •3.8 Treatment
- •3.9 Post-operative Complications and Follow-Up
- •Further Reading
- •4: Renal Tumors in Children
- •4.1 Introduction
- •4.2 Wilms’ Tumor
- •4.2.1 Introduction
- •4.2.2 Etiology
- •4.2.3 Histopathology
- •4.2.4 Nephroblastomatosis
- •4.2.5 Clinical Features
- •4.2.6 Risk Factors for Wilms’ Tumor
- •4.2.7 Staging of Wilms Tumor
- •4.2.8 Investigations
- •4.2.9 Prognosis and Complications of Wilms Tumor
- •4.2.10 Surgical Considerations
- •4.2.11 Surgical Complications
- •4.2.12 Prognosis and Outcome
- •4.2.13 Extrarenal Wilms’ Tumors
- •4.3 Mesoblastic Nephroma
- •4.3.1 Introduction
- •4.3.3 Epidemiology
- •4.3.5 Clinical Features
- •4.3.6 Investigations
- •4.3.7 Treatment and Prognosis
- •4.4 Clear Cell Sarcoma of the Kidney (CCSK)
- •4.4.1 Introduction
- •4.4.2 Pathophysiology
- •4.4.3 Clinical Features
- •4.4.4 Investigations
- •4.4.5 Histopathology
- •4.4.6 Treatment
- •4.4.7 Prognosis
- •4.5 Malignant Rhabdoid Tumor of the Kidney
- •4.5.1 Introduction
- •4.5.2 Etiology and Pathophysiology
- •4.5.3 Histologic Findings
- •4.5.4 Clinical Features
- •4.5.5 Investigations and Diagnosis
- •4.5.6 Treatment and Outcome
- •4.5.7 Mortality/Morbidity
- •4.6 Renal Cell Carcinoma in Children
- •4.6.1 Introduction
- •4.6.2 Histopathology
- •4.6.4 Staging
- •4.6.5 Clinical Features
- •4.6.6 Investigations
- •4.6.7 Management
- •4.6.8 Prognosis
- •4.7 Angiomyolipoma of the Kidney
- •4.7.1 Introduction
- •4.7.2 Histopathology
- •4.7.4 Clinical Features
- •4.7.5 Investigations
- •4.7.6 Treatment and Prognosis
- •4.8 Renal Lymphoma
- •4.8.1 Introduction
- •4.8.2 Etiology and Pathogenesis
- •4.8.3 Diagnosis
- •4.8.4 Clinical Features
- •4.8.5 Treatment and Prognosis
- •4.9 Ossifying Renal Tumor of Infancy
- •4.10 Metanephric Adenoma
- •4.10.1 Introduction
- •4.10.2 Histopathology
- •4.10.3 Diagnosis
- •4.10.4 Clinical Features
- •4.10.5 Treatment
- •4.11 Multilocular Cystic Renal Tumor
- •Further Reading
- •Wilms’ Tumor
- •Mesoblastic Nephroma
- •Renal Cell Carcinoma in Children
- •Angiomyolipoma of the Kidney
- •Renal Lymphoma
- •Ossifying Renal Tumor of Infancy
- •Metanephric Adenoma
- •Multilocular Cystic Renal Tumor
- •5.1 Introduction
- •5.2 Embryology
- •5.4 Histologic Findings
- •5.7 Associated Anomalies
- •5.8 Clinical Features
- •5.9 Investigations
- •5.10 Treatment
- •Further Reading
- •6: Congenital Ureteral Anomalies
- •6.1 Etiology
- •6.2 Clinical Features
- •6.3 Investigations and Diagnosis
- •6.4 Duplex (Duplicated) System
- •6.4.1 Introduction
- •6.4.3 Clinical Features
- •6.4.4 Investigations
- •6.4.5 Treatment and Prognosis
- •6.5 Ectopic Ureter
- •6.5.1 Introduction
- •6.5.3 Clinical Features
- •6.5.4 Diagnosis
- •6.5.5 Surgical Treatment
- •6.6 Ureterocele
- •6.6.1 Introduction
- •6.6.3 Clinical Features
- •6.6.4 Investigations and Diagnosis
- •6.6.5 Treatment
- •6.6.5.1 Surgical Interventions
- •6.8 Mega Ureter
- •Further Reading
- •7: Congenital Megaureter
- •7.1 Introduction
- •7.3 Etiology and Pathophysiology
- •7.4 Clinical Presentation
- •7.5 Investigations and Diagnosis
- •7.6 Treatment and Prognosis
- •7.7 Complications
- •Further Reading
- •8.1 Introduction
- •8.2 Pathophysiology
- •8.4 Etiology of VUR
- •8.5 Clinical Features
- •8.6 Investigations
- •8.7 Management
- •8.7.1 Medical Treatment of VUR
- •8.7.2 Antibiotics Used for Prophylaxis
- •8.7.3 Anticholinergics
- •8.7.4 Surveillance
- •8.8 Surgical Therapy of VUR
- •8.8.1 Indications for Surgical Interventions
- •8.8.2 Indications for Surgical Interventions Based on Age at Diagnosis and the Presence or Absence of Renal Lesions
- •8.8.3 Endoscopic Injection
- •8.8.4 Surgical Management
- •8.9 Mortality/Morbidity
- •Further Reading
- •9: Pediatric Urolithiasis
- •9.1 Introduction
- •9.2 Etiology
- •9.4 Clinical Features
- •9.5 Investigations
- •9.6 Complications of Urolithiasis
- •9.7 Management
- •Further Reading
- •10.1 Introduction
- •10.2 Embryology of Persistent Müllerian Duct Syndrome
- •10.3 Etiology and Inheritance of PMDS
- •10.5 Clinical Features
- •10.6 Treatment
- •10.7 Prognosis
- •Further Reading
- •11.1 Introduction
- •11.2 Physiology and Bladder Function
- •11.2.1 Micturition
- •11.3 Pathophysiological Changes of NBSD
- •11.4 Etiology and Clinical Features
- •11.5 Investigations and Diagnosis
- •11.7 Management
- •11.8 Clean Intermittent Catheterization
- •11.9 Anticholinergics
- •11.10 Botulinum Toxin Type A
- •11.11 Tricyclic Antidepressant Drugs
- •11.12 Surgical Management
- •Further Reading
- •12.1 Introduction
- •12.2 Etiology
- •12.3 Pathophysiology
- •12.4 Clinical Features
- •12.5 Investigations and Diagnosis
- •12.6 Management
- •Further Reading
- •13.1 Introduction
- •13.2 Embryology
- •13.3 Epispadias
- •13.3.1 Introduction
- •13.3.2 Etiology
- •13.3.4 Treatment
- •13.3.6 Female Epispadias
- •13.3.7 Surgical Repair of Female Epispadias
- •13.3.8 Prognosis
- •13.4 Bladder Exstrophy
- •13.4.1 Introduction
- •13.4.2 Associated Anomalies
- •13.4.3 Principles of Surgical Management of Bladder Exstrophy
- •13.4.4 Evaluation and Management
- •13.5 Cloacal Exstrophy
- •13.5.1 Introduction
- •13.5.2 Skeletal Changes in Cloacal Exstrophy
- •13.5.3 Etiology and Pathogenesis
- •13.5.4 Prenatal Diagnosis
- •13.5.5 Associated Anomalies
- •13.5.8 Surgical Reconstruction
- •13.5.9 Management of Urinary Incontinence
- •13.5.10 Prognosis
- •13.5.11 Complications
- •Further Reading
- •14.1 Introduction
- •14.2 Etiology
- •14.3 Clinical Features
- •14.4 Associated Anomalies
- •14.5 Diagnosis
- •14.6 Treatment and Prognosis
- •Further Reading
- •15: Cloacal Anomalies
- •15.1 Introduction
- •15.2 Associated Anomalies
- •15.4 Clinical Features
- •15.5 Investigations
- •Further Reading
- •16: Urachal Remnants
- •16.1 Introduction
- •16.2 Embryology
- •16.4 Clinical Features
- •16.5 Tumors and Urachal Remnants
- •16.6 Management
- •Further Reading
- •17: Inguinal Hernias and Hydroceles
- •17.1 Introduction
- •17.2 Inguinal Hernia
- •17.2.1 Incidence
- •17.2.2 Etiology
- •17.2.3 Clinical Features
- •17.2.4 Variants of Hernia
- •17.2.6 Treatment
- •17.2.7 Complications of Inguinal Herniotomy
- •17.3 Hydrocele
- •17.3.1 Embryology
- •17.3.3 Treatment
- •Further Reading
- •18: Cloacal Exstrophy
- •18.1 Introduction
- •18.2 Etiology and Pathogenesis
- •18.3 Associated Anomalies
- •18.4 Clinical Features and Management
- •Further Reading
- •19: Posterior Urethral Valve
- •19.1 Introduction
- •19.2 Embryology
- •19.3 Pathophysiology
- •19.5 Clinical Features
- •19.6 Investigations and Diagnosis
- •19.7 Management
- •19.8 Medications Used in Patients with PUV
- •19.10 Long-Term Outcomes
- •19.10.3 Bladder Dysfunction
- •19.10.4 Renal Transplantation
- •19.10.5 Fertility
- •Further Reading
- •20.1 Introduction
- •20.2 Embryology
- •20.4 Clinical Features
- •20.5 Investigations
- •20.6 Treatment
- •20.7 The Müllerian Duct Cyst
- •Further Reading
- •21: Hypospadias
- •21.1 Introduction
- •21.2 Effects of Hypospadias
- •21.3 Embryology
- •21.4 Etiology of Hypospadias
- •21.5 Associated Anomalies
- •21.7 Clinical Features of Hypospadias
- •21.8 Treatment
- •21.9 Urinary Diversion
- •21.10 Postoperative Complications
- •Further Reading
- •22: Male Circumcision
- •22.1 Introduction
- •22.2 Anatomy and Pathophysiology
- •22.3 History of Circumcision
- •22.4 Pain Management
- •22.5 Indications for Circumcision
- •22.6 Contraindications to Circumcision
- •22.7 Surgical Procedure
- •22.8 Complications of Circumcision
- •Further Reading
- •23: Priapism in Children
- •23.1 Introduction
- •23.2 Pathophysiology
- •23.3 Etiology
- •23.5 Clinical Features
- •23.6 Investigations
- •23.7 Management
- •23.8 Prognosis
- •23.9 Priapism and Sickle Cell Disease
- •23.9.1 Introduction
- •23.9.2 Epidemiology
- •23.9.4 Pathophysiology
- •23.9.5 Clinical Features
- •23.9.6 Treatment
- •23.9.7 Prevention of Stuttering Priapism
- •23.9.8 Complications of Priapism and Prognosis
- •Further Reading
- •24.1 Introduction
- •24.2 Embryology and Normal Testicular Development and Descent
- •24.4 Causes of Undescended Testes and Risk Factors
- •24.5 Histopathology
- •24.7 Clinical Features and Diagnosis
- •24.8 Treatment
- •24.8.1 Success of Surgical Treatment
- •24.9 Complications of Orchidopexy
- •24.10 Infertility and Undescended Testes
- •24.11 Undescended Testes and the Risk of Cancer
- •Further Reading
- •25: Varicocele
- •25.1 Introduction
- •25.2 Etiology
- •25.3 Pathophysiology
- •25.4 Grading of Varicoceles
- •25.5 Clinical Features
- •25.6 Diagnosis
- •25.7 Treatment
- •25.8 Postoperative Complications
- •25.9 Prognosis
- •Further Reading
- •26.1 Introduction
- •26.2 Etiology and Risk Factors
- •26.3 Diagnosis
- •26.4 Intermittent Testicular Torsion
- •26.6 Effects of Testicular Torsion
- •26.7 Clinical Features
- •26.8 Treatment
- •26.9.1 Introduction
- •26.9.2 Etiology of Extravaginal Torsion
- •26.9.3 Clinical Features
- •26.9.4 Treatment
- •26.10 Torsion of the Testicular or Epididymal Appendage
- •26.10.1 Introduction
- •26.10.2 Embryology
- •26.10.3 Clinical Features
- •26.10.4 Investigations and Treatment
- •Further Reading
- •27: Testicular Tumors in Children
- •27.1 Introduction
- •27.4 Etiology of Testicular Tumors
- •27.5 Clinical Features
- •27.6 Staging
- •27.6.1 Regional Lymph Node Staging
- •27.7 Investigations
- •27.8 Treatment
- •27.9 Yolk Sac Tumor
- •27.10 Teratoma
- •27.11 Mixed Germ Cell Tumor
- •27.12 Stromal Tumors
- •27.13 Simple Testicular Cyst
- •27.14 Epidermoid Cysts
- •27.15 Testicular Microlithiasis (TM)
- •27.16 Gonadoblastoma
- •27.17 Cystic Dysplasia of the Testes
- •27.18 Leukemia and Lymphoma
- •27.19 Paratesticular Rhabdomyosarcoma
- •27.20 Prognosis and Outcome
- •Further Reading
- •28: Splenogonadal Fusion
- •28.1 Introduction
- •28.2 Etiology
- •28.4 Associated Anomalies
- •28.5 Clinical Features
- •28.6 Investigations
- •28.7 Treatment
- •Further Reading
- •29: Acute Scrotum
- •29.1 Introduction
- •29.2 Torsion of Testes
- •29.2.1 Introduction
- •29.2.3 Etiology
- •29.2.4 Clinical Features
- •29.2.5 Effects of Torsion of Testes
- •29.2.6 Investigations
- •29.2.7 Treatment
- •29.3 Torsion of the Testicular or Epididymal Appendage
- •29.3.1 Introduction
- •29.3.2 Embryology
- •29.3.3 Clinical Features
- •29.3.4 Investigations and Treatment
- •29.4.1 Introduction
- •29.4.2 Etiology
- •29.4.3 Clinical Features
- •29.4.4 Investigations and Treatment
- •29.5 Idiopathic Scrotal Edema
- •29.6 Testicular Trauma
- •29.7 Other Causes of Acute Scrotum
- •29.8 Splenogonadal Fusion
- •Further Reading
- •30.1 Introduction
- •30.2 Imperforate Hymen
- •30.3 Vaginal Atresia
- •30.5 Associated Anomalies
- •30.6 Embryology
- •30.7 Clinical Features
- •30.8 Investigations
- •30.9 Management
- •Further Reading
- •31: Disorders of Sexual Development
- •31.1 Introduction
- •31.2 Embryology
- •31.3 Sexual and Gonadal Differentiation
- •31.5 Evaluation of a Newborn with DSD
- •31.6 Diagnosis and Investigations
- •31.7 Management of Patients with DSD
- •31.8 Surgical Corrections of DSD
- •31.9 Congenital Adrenal Hyperplasia (CAH)
- •31.10 Androgen Insensitivity Syndrome (Testicular Feminization Syndrome)
- •31.13 Gonadal Dysgenesis
- •31.15 Ovotestis Disorders of Sexual Development
- •31.16 Other Rare Disorders of Sexual Development
- •Further Reading
- •Index
21.3 Embryology |
455 |
|
|
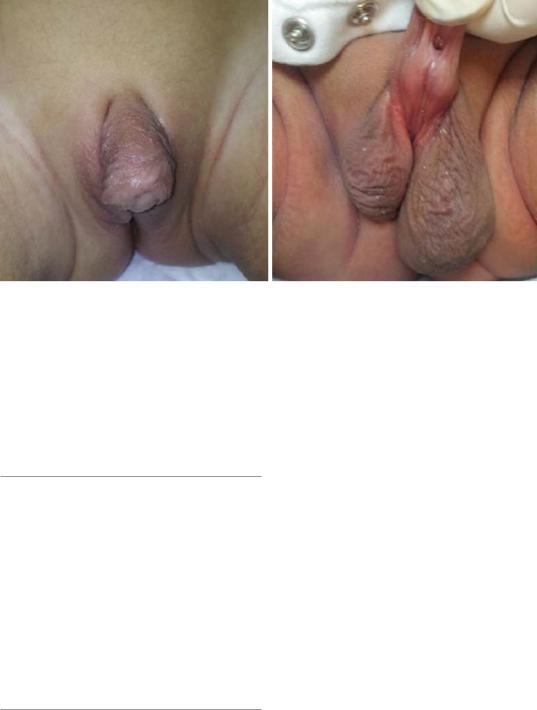
Figs. 21.11 and 21.12 Clinical photographs of two patients with hypospadias and undescended testes. In the first one, there are bilateral undescended testes while in the second one there is unilateral undescended testis
–Better understanding of the anatomy of the penis
–Improved anesthetic techniques
–Fine instrumentations and sutures
–Improved dressing materials, and antibiotics
21.2Effects of Hypospadias
•Abnormal urinary stream. The more proximally ectopic the position of the urethral meatus, the more likely the urinary stream is to be deflected downward. Any element of chordee can exacerbate this abnormality.
•Fertility may be affected as hypospadias may preclude effective insemination.
•Painful erection
•Psychological stress
21.3Embryology
•Hypospadias is a congenital defect that is thought to occur during urethral development.
•This occurs between 8 and 20 weeks’ gestation.
• Embyologically, the external genitalia are identical in males and females until about 8 weeks’ gestation.
–In males, the external genitalia develop a masculine phenotype under the influence of testosterone.
–Testesterone is converted to dihydrotestesterone under the influence of 5-alpha reductase.
–Dihydrotestesterone acts locally to change the external genitalia into a masculine phenotype.
–As the phallus grows, the open urethral groove extends from its base to the level of the corona.
–The urethral folds coalesce in the midline from base to tip, forming a tubularized penile urethra and median scrotal raphe.
–This accounts for the posterior and middle parts of the urethra.
–The anterior or glanular urethra is thought to develop in a proximal direction, with an ectodermal core forming at the tip of the glans penis, which canalizes to join with the more proximal urethra at the level of the corona.
–The higher incidence of subcoronal hypospadias supports the vulnerable final step in this embryological theory of development.

456 |
21 Hypospadias |
|
|
•In 2000, Baskin proposed a modification of this theory in which the urethral folds fuse to form a seam of epithelium, which is then transformed into mesenchyme and subsequently canalizes by apoptosis or programmed cell resorption. Similarly, this seam theoretically also develops at the glanular level, and the endoderm differentiates to ectoderm with subsequent canalization by apoptosis.
•The prepuce normally forms as a ridge of skin from the corona that grows circumferentially, fusing with the glans. Failure of fusion of the urethral folds in hypospadias impedes this process, and a dorsal hooded prepuce results.
•On rare occasions, a glanular cleft with intact prepuce may occur. This is termed the megameatus intact prepuce (MIP) variant.
•Chordee (ventral curvature of the penis) is often associated with hypospadias, especially the more proximal forms of hypospadias.
–This is thought to result from a growth disparity between the normal dorsal tissue of the corporal bodies and the attenuated ventral urethra and associated tissues.
–Rarely, the abortive spongiosal tissue and fascia distal to the urethral meatus forms a tethering fibrous band that contributes to the chordee.
21.4Etiology of Hypospadias
•The etiology of hypospadias is multifactorial but in the majority (65–75 %) the cause remains unknown.
•There has been several etiological factors for hypospadias.
•Several etiologies for hypospadias have been suggested, including genetic, endocrine, and environmental factors.
–Defects in the androgen synthesis or its action during embryogenesis (A defect in testosterone biosynthesis).
–Mutations in the 5-alpha reductase enzyme, which converts testosterone to the more potent dihydrotestosterone (DHT).
–There is a fivefold increased risk of hypospadias in males born through IVF. This
may be related to maternal exposure to progesterone, which is commonly administered in IVF protocols. Progesterone is a substrate for 5-alpha reductase and acts as a competitive inhibitor during the process of conversion of testosterone to DHT.
–Hypospadias is also found in the most common disorder of sex development, the familial male pseudohermaphroditism.
–Ingestion of substances with estrogenic activity, such as insecticides, natural estrogens, organic products from the manufacture of plastics and pesticides, which are included in food.
–A higher incidence of hypospadias in winter conceptions has also been proposed.
–Increased estradiol concentration in placental basal syncytiotrophoblasts of boys with undescended testes.
–A genetic predisposition to hypospadias.
•There is a genetic predisposition to hypospadias.
•The inheritance is likely polygenic.
•There is an eight times increased in incidence of hypospadias among monozygotic twins.
•Hypospadias was also shown to be familial.
•The probability to have a second child with hypospadias is 14 % if the first child has hypospadias.
•If the father has hypospadias, the probability that one of his sons suffers from the same pathology is 8 %.
•Hypospadias is present in many syndromes, such as Smith-Lemli-Opitz syndrome or Robinow syndrome.
•Hypospadias is associated with increasing parity, increasing maternal age, and low birth weight.
21.5Associated Anomalies
•A patent processus vaginalis (9 %)
•Undescended testes and inguinal hernias are the most common anomalies associated with hypospadias.
21.5 Associated Anomalies |
457 |
|
|
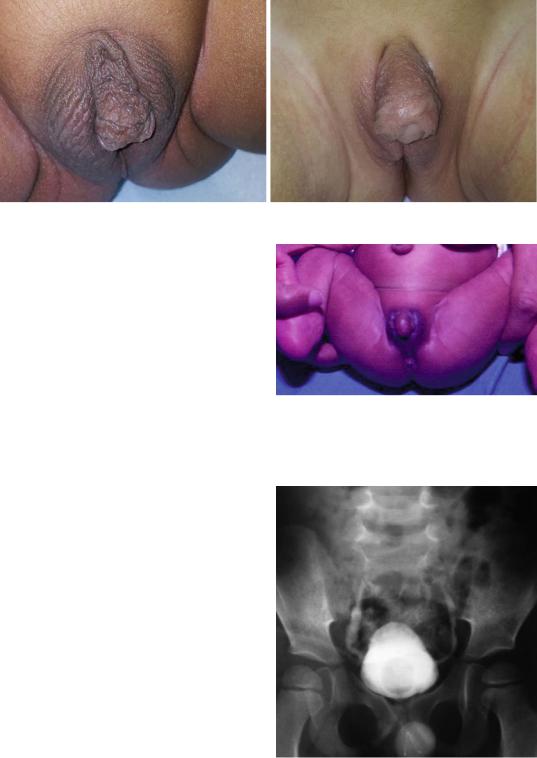
Figs. 21.13 and 21.14 Clinical photographs showing severe hypospadias and bilateral undescended testes
•The incidence of undescended testes and inguinal hernias with hypospadias is about 9 % for each.
•Undescended testis is seen in about 5 % of mild forms of hypospadias (Figs. 21.13 and 21.14).
•The incidence of undescended testes is more than 30 % in those with more proximal hypospadias.
•The incidence of inguinal hernias is about 20 % in those with more proximal hypospadias.
•The combination of hypospadias and undescended testis should raise the possibility of an underlying disorder of sexual development (DSD) (Fig. 21.15).
•DSD were identified in approximately 30 % of patients with unilateral or bilateral undescended testes and hypospadias. The incidence approaches 50 % in those with nonpalpable testes but if the testes are palpable, the incidence is only 15 %.
•The more proximal hypospadias have a higher association with DSD.
•A prostatic utricle is another association with hypospadias. This is more likely so in those with more proximal hypospadias.
•Sometimes hypospadias is associated with a low-grade vesicoureteral reflux (Fig. 21.16).
•Upper urinary tract anomalies are rarely associated with hypospadias and do not justify routine imaging in these patients.
Fig. 21.15 A clinical photograph showing a newborn with disorder of sexual development resembling hypospadias clinically
Fig. 21.16 A micturating cystourethrogram showing mild vesicoureteral reflux in a patient with hypospadias
458 |
21 Hypospadias |
|
|
21.6Classification of Hypospadias
•There are several classifications for hypospadias depending on the site of the meatus.
•One of the oldest classification was that proposed by Browne in 1938.
–Glandular hypospadias (Figs. 21.17 and 21.18)
–Coronal hypospadias (Figs. 21.19 and 21.20)
–Subcoronal hypospadias (Figs. 21.21 and 21.22)
–Distal penile hypospadias (Figs. 21.23 and 21.24)
–Midshaft hypospadias (Figs. 21.25 and 21.26)
–Proximal penile hypospadias (Figs. 21.27 and 21.28)
–Penoscrotal (Figs. 21.29 and 21.30)
–Perineal (Figs. 21.31 and 21.32)
•The most accepted and currently used classification is the one proposed by Barcat and modified by Duckett in 1996. He divided the different types of hypospadias into anterior
(50 %), middle (30 %) and posterior (20 %) hypospadias.
•These are also sub classified according to the location of the meatus:
–The anterior type:
•Glandular
•Coronal
•Distal penile
–The middle type:
•Midshaft
•Proximal penile
–The posterior type:
•Penoscrotal
•Scrotal
•Perineal
•The latest classification was proposed in 2003 and divides hypospadias into three types.
–Glaular
–Distal
–Proximal
•The subcoronal hypospadias is the most common type.
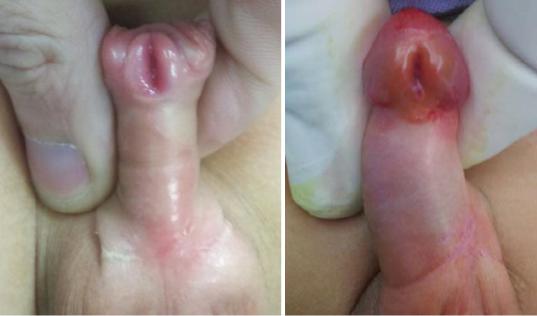
Figs. 21.17 and 21.18 Clinical photographs showing glanular hypospadias
21.6 Classification of Hypospadias |
459 |
|
|
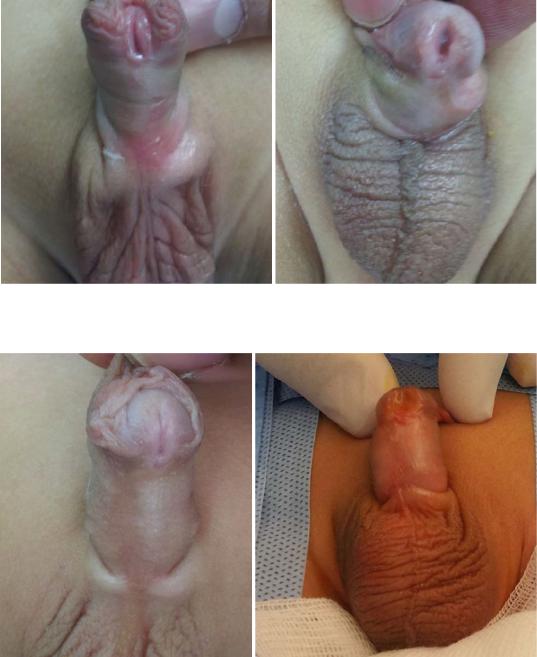
Figs. 21.19 and 20 Clinical photographs showing coronal hypospadias
Figs. 21.21 and 21.22 A clinical photograph showing subcoronal hypospadias
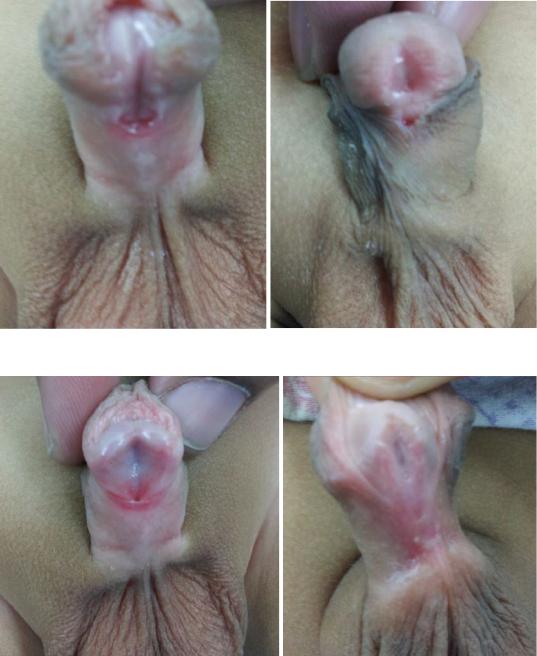
460 |
21 Hypospadias |
|
|
Figs. 21.23 and 21.24 Clinical photographs showing distal penile hypospadias
Figs. 21.25 and 21.26 Clinical photographs showing midshaft hypospadias

21.6 Classification of Hypospadias |
461 |
|
|
Figs. 21.27 and 21.28 Clinical photographs showing proximal hypospadias
Figs. 21.29 and 21.30 Clinical photographs showing penoscrotal hypospadias
