
- •Preface
- •Acknowledgments
- •Basic Physics
- •Transducers and Resolution
- •Doppler Physics
- •Artifacts
- •Summary
- •Introduction
- •Patient Preparation
- •Patient Positioning
- •Transducer Selection
- •Two-Dimensional Images
- •Two-Dimensional Imaging Controls
- •Introduction
- •M-Mode Echocardiography
- •Color-Flow Doppler
- •Spectral Doppler
- •Tissue Doppler Imaging
- •Measurement and Assessment of Two-Dimensional Images
- •Measurement and Assessment of M-Mode Images
- •Measurement and Assessment of Spectral Doppler Flow
- •Measurement and Assessment of Tissue Doppler Imaging
- •Evaluation of Color-Flow Doppler
- •Evaluation of Ventricular Function
- •Mitral Regurgitation
- •Aortic Regurgitation
- •Tricuspid Regurgitation
- •Pulmonary Regurgitation
- •Endocarditis
- •Pulmonary Hypertension
- •Systemic Hypertension
- •Hypertrophic Cardiomyopathy
- •Dynamic Right Ventricular Outflow Obstruction
- •Moderator Bands
- •Dilated Cardiomyopathy
- •Right Ventricular Cardiomyopathy
- •Restrictive Cardiomyopathy
- •Endocardial Fibroelastosis
- •Arrhythmogenic Right Ventricular Cardiomyopathy
- •Myocardial Infarction
- •Myocardial Contusions
- •Pericardial Effusion
- •Neoplasia as a Cause of Pericardial Effusion
- •Pericardial Disease
- •Abscesses
- •Pericardial Cysts
- •Thrombus
- •Ventricular Septal Defect
- •Patent Ductus Arteriosus
- •Aorticopulmonary Window
- •Right to Left Shunting PDA
- •Atrial Septal Defects
- •Endocardial Cushion Defects
- •Bubble Studies
- •Atrioventricular Valve Dysplasia
- •Outflow Obstructions
- •Inflow Obstructions
- •Tetralogy of Fallot
- •APPENDIX ONE Bovine
- •APPENDIX TWO Canine
- •APPENDIX THREE Equine
- •APPENDIX FOUR Feline
- •APPENDIX FIVE Miscellaneous Species
- •Index
associated with pulmonary hypertension are seen including severe right ventricular hypertrophy and a very prominent pulmonary artery. Chapters 4 and 6 describe the findings of pulmonary hypertension. Doppler flow will be negative when the shunt is interrogated on right parasternal four-chamber views. Aortic flow velocities will be elevated since the shunt now involves that vessel.
Endocardial Cushion Defects
When both a very large ostium primum atrial septal defect and a large ventricular septal defect are present, the abnormality is called an endocardial cushion defect. It may also be called complete atrioventricular canal or atrioventricular septal defect (62,100,101). Endocardial cushion defects fall into two categories: incomplete (or partial) and complete. Complete defects include an ostium primum atrial septal defect and a ventricular septal defect where a large portion of the inferior atrial septum and a large portion of the superior portion of the ventricular septum are missing. These two septal defects connect and the entire central portion of the heart is missing. There is typically one large atrioventricular valve since the septal portion of the endocardial cushion did not develop sufficiently to form the septal portions of the AV annuluses. Incomplete endocardial cushion defects involve a very large ostium primum atrial septal defect resulting in an almost common atrium and abnormalities of the atrioventricular valves (1,100) (Figure 9.29). The mitral valve is often cleft (1,100).
Two-dimensional echocardiographic images are very dramatic. The right parasternal four-chamber view shows a large atrial septal defect as well as the ventricular septal defect (Figure 9.34). The mitral and tricuspid valves originate from the endocardial cushion and are usually dysplastic. There often appears to be just one large atrioventricular valve with only the parietal leaflets of the tricuspid and mitral valves present. When septal mitral and tricuspid leaflets are present, they are usually continuous through the endocardial cushion defect. The right atrium, right ventricle, and left atrium are always dilated. The left ventricle may be dilated. The valves are usually insufficient, and the defects are so large that both color-flow and spectral Doppler are of no help in determining the direction of flow.
Figure 9.34 Endocardial cushion defects have a large ostium primum atrial septal defect and a ventricular septal defect. (A) Long-axis four-chamber images show large portions of both the atrial and ventricular septums missing. Only the parietal leaflets of the mitral and tricuspid valves are present. (B) Transverse views of the heart base show the large ostium primum defect of this endocardial cushion defect (arrow). (C) Transverse images at the level of the left ventricle show the large ventricular septal defect associated with the endocardial cushion defect (arrow). AO = aorta, RA = right atrium, LA = left atrium, RV = right ventricle, LV = left ventricle, IAS = interatrial septum.

Bubble Studies

Bubble studies help confirm the existence of right to left shunting. Normal saline is drawn into a syringe, shaken vigorously, all large bubbles of air are pushed from the syringe leaving only microbubbles within the solution. Several milliliters of saline are used in larger dogs, but about 1 milliliter is used in cats and other small animals. The saline is injected into a peripheral vein while the heart is being imaged. A right parasternal four-chamber view is used for the detection of atrial septal defects, and a right parasternal long-axis left ventricular outflow view should be used for ventricular septal defects. Dense echoes associated with the air-filled fluid can be seen crossing the atrial or ventricular septums into the left-sided chambers (62,102,103).
Bubble studies
help identify R → L shunts
use the abdominal aorta for PDA
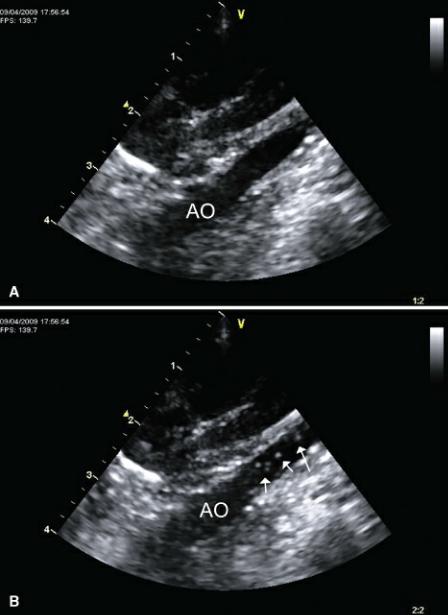
Because the shunt in patent ductus arteriosus is extracardiac and blood flows from the pulmonary artery into the descending aorta, a bubble study using cardiac images will not help define the existence of a reverse PDA. The abdominal aorta should be used instead (Figure 9.35). The dense echoes associated with the microbubbles will show up in the abdominal aorta if a reverse PDA exists (92). It is important to rule out VSD and ASD since these reverse flows will also be seen in the abdominal aorta (103).
Figure 9.35 (A) The abdominal aorta is examined when performing a bubble study to confirm the presence of reverse patent ductus arteriosus. (B) Dense echoes associated with reverse flow (arrows) fill the aorta if a reverse shunt of any kind is present. A bubble study within the heart rules out reversed atrial or ventricular septal defects. These will also cause bubbles to appear in the abdominal aorta. AO = aorta.
Atrioventricular Valve Dysplasia
Congenital dysplasia of the atrioventricular valves usually causes valvular insufficiency. The degree of dysplasia and regurgitation is very variable. The cardiac changes associated with these defects are similar to those seen when the valvular insufficiencies are secondary to acquired valvular lesions. Cardiac changes associated with mitral dysplasia include and eccentric left ventricular hypertrophy, elevated parameters of left ventricular function, left atrial dilation, and excessive wall and septal motion (62). Right-sided changes associated with tricuspid dysplasia include right ventricular volume overload, possible paradoxical septal motion, and right atrial dilation (62). For details and further description of these changes, refer to Chapter 5 and the discussions on acquired mitral and tricuspid valve disease. Descriptions of the two-dimensional appearance of the dysplastic atrioventricular valves follow.
The appearance of the valvular apparatus is extremely variable, and any combination of abnormally shaped leaflets, abnormal chordal attachments, and abnormal papillary muscles may be seen. The leaflets may be thick, long, short, contain clefts, or have some degree of commissural fusion. The papillary muscles may be abnormally shaped or elongated or large (54,57,60).
Tricuspid Dysplasia
This defect of the tricuspid valve is particularly common in Labrador retrievers, but other large breed dogs are also predisposed to the defect (54). The septal leaflet is typically tethered to the right side of the septum. Short chordae may be visible as the leaflet tries to move toward its normal closed position during systole (Figure 9.36). The middle of the leaflet often buckles away from the septum while the tips remain closely apposed to the septum. The anterior leaflet is typically elongated and may come close to achieving closure of the tricuspid valves during systole. It is sometimes difficult to define where the anterior leaflet of the tricuspid valve ends and where the chordae tendinae start (21).
Figure 9.36 Tricuspid dysplasia may involve any aspect of the tricuspid apparatus. Typically, the septal leaflet of the valve is tethered to the interventricular septum by short chordae. Often the parietal tricuspid valve leaflet is elongated with abnormal attachment to a web of papillary muscles. (A) The septal leaflet is seen tethered to the septum (small arrow). Notice that the mitral leaflets are closed while the tricuspid leaflets are wide open. There is abnormal papillary muscle architecture (long arrows) at the apex of the right ventricle. (B) The web of papillary muscles at the apex of the right ventricle is seen here (short arrow), and there is a glimpse of the septal leaflet as it remains tethered to the septum (long arrow). (C) The tricuspid valve leaflets in this dog have chordae that attach abnormally to the leaflets (arrows). Planes = right parasternal four-chamber views, RV = right ventricle, RA = right atrium, LV = left ventricle, LA = left atrium.
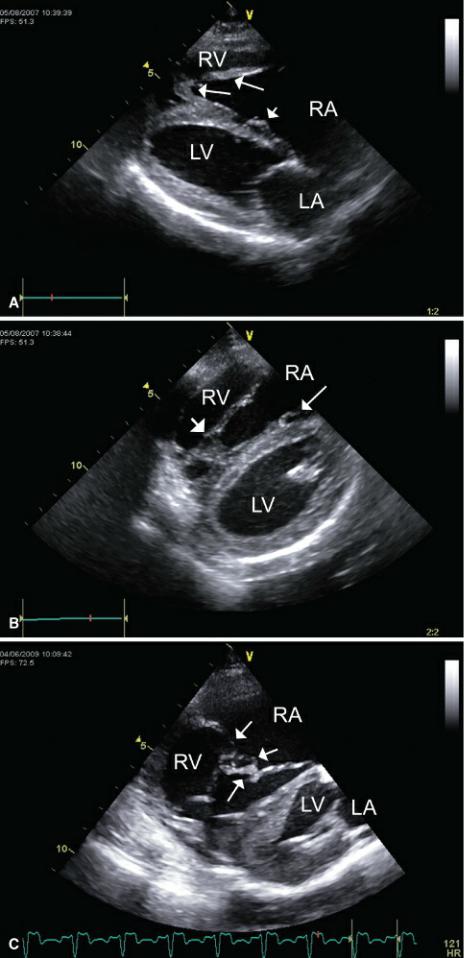
An unusual variation of tricuspid dysplasia is Ebstein’s Anomaly. The tricuspid annulus is actually displaced toward the apex of the right ventricle (104). The right atrial chamber is very large in this

defect, and the ventricle is called atrialized. The annulus can be identified on right parasternal longaxis four-chamber views of the heart. The mitral and tricuspid annulus should be aligned almost directly across from each other in normal hearts. The tricuspid valve is typically a millimeter or 2 closer to the apex of the normal heart than the mitral annulus. With Ebstein’s anomaly, the tricuspid annulus is clearly displaced toward the apex (Figure 9.37). Be sure to examine the septal cusp carefully since a severely tethered leaflet may only move away from the septum at a more apical location and yield the appearance of a displaced annulus. Regardless, the valve is dysplastic (21).
Figure 9.37 With Ebstein’s anomaly, the tricuspid annulus is displaced toward the apex of the right ventricle. (A) An atrialized right ventricle is seen with tricuspid annular displacement to the apex (small arrow). The normal point of attachment should be at the level of mitral annulus (large arrow). (B) The tricuspid annulus (long arrow) is situated closer to the apex of the right ventricle compared to the mitral annulus (short arrow). This image also shows evidence of a patent foramen ovale (thick arrow).
Even though the right atrium is enlarged because the leaflets close in a more apical location than normal, tricuspid insufficiency is not always severe. Often valves with very abnormal architecture on
echocardiographic images are only mild to moderately incompetent.
Mitral Dysplasia
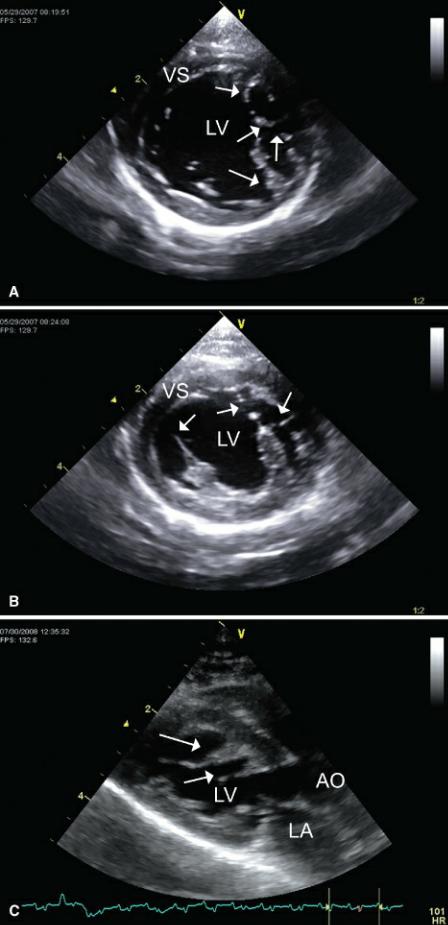
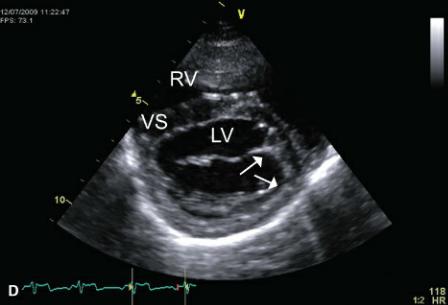
Mitral dysplasia is particularly common in cats (60). As with tricuspid dysplasia, the leaflets may be short and thick or elongated. They may be associated with abnormally shaped, elongated or large papillary muscles, and aberrant chordae tendinae with abnormal attachments to the ventricular wall or septum (Figure 9.38). Although it does occur, the septal leaflet is not usually tethered to the septum as in tricuspid dysplasia. The valves may display increased echogenicity and abnormal motion secondary to malformation of the mitral apparatus. There may be prolapse and some degree of restricted motion secondary to commissural fusion or abnormal chordae tendinae (54,57).
Figure 9.38 Mitral dysplasia is associated with abnormally shaped, elongated, or large papillary muscles and aberrant chordae tendinae with abnormal attachments to the ventricular wall or septum. (A) Aberrant chordae tendinae (arrows) with abnormal attachments to the ventricular wall are present in this cat. Plane = right parasternal transverse mitral valve. (B) Chordae tendinae extend from the papillary muscles to the septum and lateral wall (arrows) of the left ventricular chamber in this cat. Plane = right parasternal transverse left ventricle. (C) Abnormal muscular protrusions from the septum and aberrant chordae tendinae with attachments to the ventricular wall are present in this cat. Plane = right parasternal left ventricular outflow view. (D) This dog has abnormal points of mitral chordal attachment (arrows) to the lateral wall of the ventricular chamber. Plane = right parasternal transverse mitral valve, VS = ventricular septum, LV = left ventricle, AO = aorta, LA = left atrium, RV = right ventricle.
References
1.Driscoll DJ. Left-to-right shunt lesions. Pediatr Clin North Am 1999;46:355–368, x.
2.Tidholm A. Retrospective study of congenital heart defects in 151 dogs. J Small Anim Pract 1997;38:94–98.
3.Sisson D, Thomas W, Bonagura J. Congenital heart disease. In: Ettinger S, ed. Textbook of veterinary internal medicine. 5th ed. Philadelphia: WB Saunders Co, 2000;737–787.
4.Shimizu M, Tanaka R, Hirao H, et al. Percutaneous transcatheter coil embolization of a ventricular septal defect in a dog. J Am Vet Med Assoc 2005;226:69–72, 52–63.
5.Buchanan J. Prevalence of cardiovascular disorders. In: Fox P, Sisson D, Moise N, eds. Textbook of Canine and Feline Cardiology. 2nd ed. Philadelphia: WB Saunders, 1999;457–470.
6.Buczinski S, Fecteau G, DiFruscia R. Ventricular septal defects in cattle: a retrospective study of 25 cases. Can Vet J 2006;47:246–252.
7.Bayly WM, Reed SM, Leathers CW, et al. Multiple congenital heart anomalies in five Arabian foals. J Am Vet Med Assoc 1982;181:684–689.
8.Lombard CW, Scarratt WK, Buergelt CD. Ventricular septal defects in the horse. J Am Vet Med Assoc 1983; 183:562–565.
9.Boon JA, Knight AP, Moore DH. Llama cardiology. Vet Clin North Am Food Anim Pract 1994;10:353– 370.
10.Gopal T, Leipold HW, Dennis SM. Congenital cardiac defects in calves. Am J Vet Res 1986;47:1120– 1121.
11.Reef VB. Evaluation of ventricular septal defects in horses using two-dimensional and Doppler echocardiography. Equine Vet J Suppl 1995:86–95.
12.Shimizu M, Tanaka R, Hoshi K, et al. Surgical correction of ventricular septal defect with aortic regurgitation in a dog. Aust Vet J 2006;84:117–121.
13.Sisson D, Luethy M, Thomas W. Ventricular septal defect accompanied by aortic regurgitation in
five dogs. J Am Anim Hosp Assoc 1991;27:441–448.
14.Bonagura J, Darke P. Congenital heart disease. In: Ettinger S, Feldman E, eds. Textbook of veterinary internal medicine Diseases of the dog and cat. 5th ed. Philadelphia: WB Saunders, 1995;892–943.
15.Reef V, McGuirk S. Diseases of the cardiovascular system. In: Smith B, ed. Large animal internal medicine. 3rd ed. St Louis: Mosby, 2002;443–478.
16.Pipers FS, Reef V, Wilson J. Echocardiographic detection of ventricular septal defects in large animals. J Am Vet Med Assoc 1985;187:810–816.
17.Lombard C, Scaratt W, Buergelt C. Ventricular septal defects in the horse. J AM Vet Med Assoc 1983;183: 562–565.
18.Bonagura J, Lehmkuhl L. Congenital heart disease In: PR Fox, D Sisson, NS Moise, eds. Textbook of canine and feline cardiology: principles and clinical practice. Philadelphia: WB Saunders, 1999;471–535.
19.Moise NS. Doppler echocardiographic evaluation of congenital cardiac disease. An introduction. J Vet Intern Med 1989;3:195–207.
20.Moise N. Echocardiography and Doppler imaging. In: PR Fox, D Sisson, NS Moise, eds. Textbook of canine and feline cardiology. 2nd ed. New York: Churchill Livingstone, 1999;130–171.
21.Wingfield WE, Boon JA. Echocardiography for the diagnosis of congenital heart defects in the dog. Vet Clin North Am Small Anim Pract 1987;17:735–753.
22.Tohyama MDK, Satomi MDG, Momma MDK. Aortic valve prolapse and aortic regurgitation associated with subpulmonic ventricular septal defect. Am J Cardiol 1997;79:1285–1289.
23.Layangool T, Kirawittaya T, Sangtawesin C. Aortic valve prolapse in subpulmonic ventricular septal defect. J Med Assoc Thai 2003;86 Suppl 3:S549–555.
24.Eapen RS, Lemler MS, Scott WA, et al. Echocardiographic characteristics of perimembranous ventricular septal defects associated with aortic regurgitation. J Am Soc Echocardiogr 2003;16:209–
25.Graham T, Kavanaugh-McHugh A. Ventricular septal defect and aortic regurgitation. Prog Ped Cardiol 2001;14:163–173.
26.Eroglu AG, Öztunç F, Saltik L, et al. Aortic valve prolapse and aortic regurgitation in patients with ventricular septal defect. Pediatr Cardiol 2003;24: 36–39.
27.Bütter A, Duncan W, Weatherdon D, et al. Aortic cusp prolapse in ventricular septal defect and its association with aortic regurgitation—appropriate timing of surgical repair and outcomes. Can J Cardiol 1998;14:833–840.
28.Anderson RH, de Leval MR. The morphology of ventricular septal defects as related to the mechanics associated with aortic regurgitation. Seminars in Thoracic and Cardiovascular Surgery: Pediatric Cardiac Surgery Annual 2006;9:140–146.
29.Tatsuno K, Konno S, Sakakibara S. Ventricular septal defect with aortic insufficiency. Angiocardiographic aspects and a new classification. Am Heart J 1973;85:13–21.
30.Tatsuno K, Konno S, Ando M, et al. Pathogenetic mechanisms of prolapsing aortic valve and aortic regurgitation associated with ventricular septal defect. Anatomical, angiographic, and surgical considerations. Circulation 1973;48:1028–1037.
31.Tomita H, Arakaki Y, Ono Y, et al. Imbalance of cusp width and aortic regurgitation associated with aortic cusp prolapse in ventricular septal defect. Jpn Circ J 2001;65:500–504.
32.Thomas WP. Echocardiographic diagnosis of congenital membranous ventricular septal aneurysm in the dog and cat. J Am Anim Hosp Assoc 2005;41:215–220.
33.Onat T, Ahunbay G, Batmaz G, et al. The natural course of isolated ventricular septal defect during adolescence. Pediatr Cardiol 1998;19:230–234.
34.Beerman LB, Park SC, Fischer DR, et al. Ventricular septal defect associated with aneurysm of the membranous septum. J Am Coll Cardiol 1985;5:118–123.
35.Anderson RH, Lenox CC, Zuberbuhler JR. Mechanisms of closure of perimembranous ventricular septal defect. Am J Cardiol 1983;52:341–345.
36.Wu MH, Wu JM, Chang CI, et al. Implication of aneurysmal transformation in isolated perimembranous ventricular septal defect. Am J Cardiol 1993;72: 596–601.
37.Freedom RM, White RD, Pieroni DR, et al. The natural history of the so-called aneurysm of the membranous ventricular septum in childhood. Circulation 1974;49:375–384.
38.Rausch WP, Keene BW. Spontaneous resolution of an isolated ventricular septal defect in a dog. J Am Vet Med Assoc 2003;223:219–220, 197.
39.Breznock EM. Spontaneous closure of ventricular septal defects in the dog. J Am Vet Med Assoc 1973;162: 399–403.
40.Magee AG, Fenn L, Vellekoop J, et al. Left ventricular function in adolescents and adults with restrictive ventricular septal defect and moderate left-to-right shunting. Cardiol Young 2000;10:126–
41.Eren M, Dagdeviren B, Bolca O, et al. Proximal isovelocity surface area (PISA) as a noninvasive method for the estimation of the shunt quantification in perimembranous ventricular septal defects.
Echocardiography 2001; 18:137–147.
42.Webb G, Smallhorn J, Therrien J, et al. Congenital heart disease. In: Libby P, Bonow R, Mann D, et al., eds. Braunwald’s Heart Disease: a textbook of cardiovascular medicine. Philadelphia: Saunders Elsevier, 2008;1583–1586.
43.Schamberger MS, Farrell AG, Darragh RK, et al. Use of peak Doppler gradient across ventricular septal defects leads to underestimation of right-sided pressures in patients with “sloped” Doppler signals. J Am Soc Echocardiogr 2001;14:1197–1202.
44.Appleton CP, Hatle LK, Popp RL. Demonstration of restrictive ventricular physiology by Doppler echocardiography. J Am Coll Cardiol 1988;11:757–768.
45.Marx GR, Allen HD, Goldberg SJ. Doppler echocardiographic estimation of systolic pulmonary artery pressure in pediatric patients with interventricular communications. J Am Coll Cardiol 1985;6:1132–1137.
46.Pieroni DR, Nishimura RA, Bierman FZ, et al. Second natural history study of congenital heart defects. Ventricular septal defect: echocardiography. Circulation 1993;87(2) Suppl:I80–88.
47.Murphy DJ, Jr., Ludomirsky A, Huhta JC. Continuous-wave Doppler in children with ventricular septal defect: noninvasive estimation of interventricular pressure gradient. Am J Cardiol 1986;57:428–432.
48.Ge Z, Zhang Y, Kang W, et al. Noninvasive evaluation of interventricular pressure gradient across
ventricular septal defect: a simultaneous study of Doppler echocardiography and cardiac catheterization. Am Heart J 1992;124:176–182.
49.Lindblade CL, Schamberger MS, Darragh RK, et al. Use of peak Doppler gradient across ventricular septal defect leads to underestimation of right-sided pressures in a patient with M-shaped Doppler signal: a case report. J Am Soc Echocardiogr 2004;17:1207–1209.
50.Warren AE, Sharratt GP. Right bundle branch block: a source of error in the Doppler assessment of right ventricular pressure in patients with a ventricular septal defect. Can J Cardiol 1998;14:1165– 1167.
51.Moise V, Maciel B, Hornberger L, et al. A new method for noninvasive estimation of ventricular septal defect shunt flow by Doppler color flow mapping: imaging of the laminar flow convergence region on the left septal surface. J Am Coll Cardiol 1991;18:824–832.
52.Kurotobi S, Sano T, Matsushita T, et al. Quantitative, non-invasive assessment of ventricular septal defect shunt flow by measuring proximal isovelocity surface area on colour Doppler mapping. Heart 1997;78: 305–309.
53.Kosecik M, Sagin-Saylam G, Unal N, et al. Noninvasive assessment of left-to-right shunting in ventricular septal defects by the proximal isovelocity surface area method on Doppler colour flow mapping. Can J Cardiol 2007; 23:1049–1053.
54.Bonagura JD, Herring DS. Echocardiography. Congenital heart disease. Vet Clin North Am Small Anim Pract 1985;15:1195–1208.
55.Bonagura J. Contemporary issues in small animal practice: cardiology. New York: Churchill Livingston, 1987.
56.Buchanan J. Causes and prevalence of cardiovascular disease. In: Kirk RW, Bonagura JD eds, Current Vet Therapy XI. Philadelphia, PA: WB Saunders 1992: 647–655.
57.Darke P. Congenital heart disease in dogs and cats. J Sm Anim Prac 1989;30:599–607.
58.Fingland RB, Bonagura JD, Myer CW. Pulmonic stenosis in the dog: 29 cases (1975–1984). J Am Vet Med Assoc 1986;189:218–226.
59.Van Israël N, French AT, Dukes-McEwan J, et al. Review of left-to-right shunting patent ductus arteriosus and short term outcome in 98 dogs. J Small Anim Pract 2002;43:395–400.
60.Bolton GR, Liu SK. Congenital heart diseases of the cat. Vet Clin North Am 1977;7:341–353.
61.Reef V. Echocardiographic findings in horses with congenital cardiac disease. Comp Med 1991;13:109–117.
62.Kaplan PM. Congenital heart disease. Probl Vet Med 1991;3:500–519.
63.Caguioa ES, Reimold SC, Velez S, et al. Influence of aortic pressure on effective regurgitant orifice area in aortic regurgitation. Circulation 1992;85:1565–1571.
64.Van Israël N, Dukes-McEwan J, French AT. Long-term follow-up of dogs with patent ductus arteriosus. J Small Anim Pract 2003;44:480–490.
65.Luis-Fuentes V, Schober K, Bonagura J. Current issues in echocardiography. Proc 18th ACVIM 2000:88.
66.Barlow AJ, Ward C, Webber SA, et al. Myocardial contractility in premature neonates with and without patent ductus arteriosus. Pediatr Cardiol 2004;25: 102–107.
67.Saunders AB, Miller MW, Gordon SG, et al. Echocardiographic and angiographic comparison of
ductal dimensions in dogs with patent ductus arteriosus. Journal of Veterinary Internal Medicine 2007; 21:68–75.
68.Corti LB, Merkley D, Nelson OL, et al. Retrospective evaluation of occlusion of patent ductus arteriosus with hemoclips in 20 dogs. J Am Anim Hosp Assoc 2000;36:548–555.
69.Kittleson M. Patent ductus arteriosus. In: Kittleson M, Kienle R, eds. Small animal Cardiovascular medicine. St. Louis: Mosby, 1998;218–230.
70.Jeong Y-H, Yun T-J, Song J-M, et al. Left ventricular remodeling and change of systolic function after closure of patent ductus arteriosus in adults: Device and surgical closure. American Heart Journal 2007;154:436–440.
71.Schober KE, Luis Fuentes V, McEwan JD, et al. Pulmonary venous flow characteristics as assessed by transthoracic pulsed Doppler echocardiography in normal dogs. Vet Radiol Ultrasound 1998;39:33–41.
72.Jacquet J, Nicolle AP, Chetboul V, et al. Echocardiographic and Doppler characteristics of postoperative ductal aneurysm in a dog. Vet Radiol Ultrasound 2005;46: 518–520.
73.Schneider M, Hildebrandt N, Schweigl T, et al. Transthoracic echocardiographic measurement of patent ductus arteriosus in dogs. J Vet Intern Med 2007;21:251–257.
74.Pariaut R, Moise N, Kraus M, et al. Use of transesophageal echocardiography for visualization of the patent ductus arteriosus during transcatheter coil embolization. J Vet Cardiol 2004;6:32–39.
75.Wong JA, Shim D, Khoury PR, et al. Validation of color Doppler measurements of minimum patent ductus arteriosus diameters: significance for coil embolization. Am Heart J 1998;136(4) Pt 1:714–717.
76.O’Grady M, Cockshutt J, Khanna A, et al. Patent ductus arteriosus in a Holstein calf: a twodimensional and Doppler echocardiographic study of the ductus arteriosus and validation. Can Vet J 1991;32:303–304.
77.Skinner J. Diagnosis of patent ductus arteriosus. Seminars in Neonatology 2001;6:49–61.
78.Pouchelon J, Chetboul V, Garnier E. Proposition d’une classification echocardiographique de la persistence du canal arterial chez le chien a partir de 24 cas. Receuil de Medicine Veterinaire 1997;172:623–642.
79.Buchanan J. Patent ductus arteriosus: morphology, pathogenesis, types and treatment. J Vet Cardiol 2001; 3:7–16.
80.Bureau S, Monnet E, Orton EC. Evaluation of survival rate and prognostic indicators for surgical treatment of left-to-right patent ductus arteriosus in dogs: 52 cases (1995–2003). Journal of the American Veterinary Medical Association 2005;227:1794–1799.
81.Goodwin J, Lombard C. Patent ductus arteriosus in adult dogs: clinical features of 14 cases. JAVMA 1992; 28:349–354.
82.Kono T, Sabbah HN, Rosman H, et al. Left ventricular shape is the primary determinant of functional mitral regurgitation in heart failure. J Am Coll Cardiol 1992; 20:1594–1598.
83.Soares AM, Atik E, Cortêz TM, et al. Aortopulmonary window. Clinical and surgical assessment of 18 cases. Arq Bras Cardiol 1999;73:59–74.
84.Grossman W, Jones D, McLaurin L. Wall stress and patterns of hypertrophy. J Clin Invest 1975;56:56–64.
85.Turk JR, Miller LM, Miller JB, et al. Necrotizing pulmonary arteritis in a dog with patent ductus arteriosus. J Small Anim Pract 1981;22:603–608.
86.Feldman T. Rheumatic mitral stenosis. On the rise again. Postgrad Med 1993;93:93–94, 99–104.
87.Weir E, Reeves J. Pulmonary hypertension. New York: Futura Publishing Co Inc, 1984.
88.Nimmo-Wilkie J, Feldman E. Pulmonary vascular lesions associated with congenital heart defects in three dogs. J Am Anim Hosp Assoc 1981;17:485–490.
89.Patterson DF. Congenital defects of the cardiovascular system of dogs: studies in comparative cardiology. Adv Vet Sci Comp Med 1976;20:1–37.
90.Patterson DF. Canine congenital heart disease: epidemiology and etiological hypotheses. J Small Anim Pract 1971;12:263–287.
91.Weirich W, Blevins W, Rebar A. Late consequences of patent ductus arteriosus in the dog: a report of six cases. J Am Anim Hosp Assoc 1978;14:40–51.
92.Ferasin L, Rizzo F, Darke PGG. Original investigation of right-to-left shunting patent ductus arteriosus in an Irish setter puppy. The Veterinary Journal 2007;173: 443–448.
93.Kirberger RM, Berry WL. Atrial septal defect in a dog: the value of Doppler echocardiography. J S Afr Vet Assoc 1992;63:43–47.
94.Olivier N. Congenital heart disease in dogs. In: Fox P, ed. Canine and Feline Cardiology. New York: Churchill Livingston, 1988;357–389.
95.Taylor FG, Wotton PR, Hillyer MH, et al. Atrial septal defect and atrial fibrillation in a foal. Vet Rec 1991;128:80–81.
96.Church D, Allan G. Atrial septal defect and Eisenmenger’s syndrome in a mature cat. Aus Vet J 1990;67:380.
97.Chetboul V, Trollé JM, Nicolle A, et al. Congenital heart diseases in the boxer dog: A retrospective study of 105 cases (1998–2005). J Vet Med A Physiol Pathol Clin Med 2006;53:346–351.
98.McCann WD, Harbold NB, Jr., Giuliani ER. The echocardiogram in right ventricular overload. JAMA 1972;221:1243–1245.
99.Marx GR, Allen HD, Goldberg SJ, et al. Transatrial septal velocity measurement by Doppler echocardiography in atrial septal defect: correlation with Qp:Qs ratio. Am J Cardiol 1985;55:1162– 1167.
100.Sulafa AK, Tamimi O, Najm HK, et al. Echocardiographic differentiation of atrioventricular septal defects from inlet ventricular septal defects and mitral valve clefts. Am J Cardiol 2005;95:607–
101.Ecke P, Malik R, Kannegieter NJ. Common atrioventricular canal in a foal. N Z Vet J 1991;39:97–98.
102.Bonagura J, Herring D. Echocardiography—acquired heart disease. Vet Clin N Am: Sm Anim Prac 1985;15:1209–1224.
103.Goodwin J, Holland M. Contrast echoaortography as an aid in the diagnosis of right to left shunting patent ductus arteriosus. Vet Rad Ultra 1995;36:157–159.
104.Andelfinger G, Wright KN, Lee HS, et al. Canine tricuspid valve malformation, a model of human Ebstein anomaly, maps to dog chromosome 9. J Med Genet 2003;40:320–324.
CHAPTER TEN
Stenotic Lesions
Outflow obstructions created by aortic or pulmonary stenosis create excessive work for the ventricles and lead to concentric hypertrophy of the respective chamber. The hypertrophy develops in order to normalize systolic wall stress (1,2). Pulmonary stenosis leads to hypertrophy of both the right ventricular wall and the septum, while aortic stenosis leads to increased free wall and septal thicknesses. The chamber size may be smaller than normal secondary to the hypertrophy. Mild obstructions to outflow may not result in any visible changes within the heart, and Doppler studies are necessary to confirm the presence of mild stenosis on either side of the heart.
Inflow obstruction secondary to mitral and tricuspid stenosis is much less common. The resistance to filling of the ventricular chambers results in dilated atria. The atrioventricular valves are often incompetent as well. Obstructions can also exist within the atrial chambers and will create the same hemodynamic problems as valvular stenosis.
The many varying morphological manifestations of these stenotic lesions are presented here. An algorithm has been created that provides the echocardiographer with specific parameters that logically lead to a diagnosis and assessment of severity in animals with isolated congenital heart defects (Figure 10.1) (3). This algorithm works well when the congenital heart disease is not complicated with other concurrent cardiac problems such as valvular insufficiency, pulmonary hypertension, or combinations of stenotic or shunt lesions.
Figure 10.1 Echocardiographic algorithm for the evaluation of canine congenital heart disease.
Reprinted with permission of Oyama MA, Sisson DD. Evaluation of canine congenital heart disease using an echocardiographic algorithm. J Am Anim Hosp Assoc 2001;37:519–535.
