
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index
212 EVOLUTION OF THE CHLOROPLAST
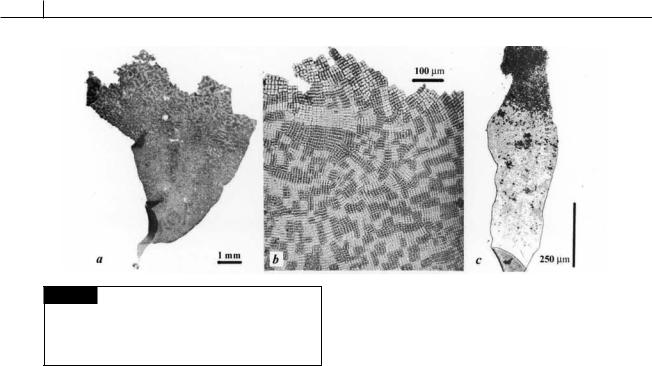
Fig. 5.79 Prasiola stipitata. (a) Sporophyte producing male and female gametes. (b) Apical part of sporophyte thallus with male (dark areas) and female (light areas) gametes.
(c) Sporophyte producing spores from the apical part of the thallus. (From Friedmann, 1959.)
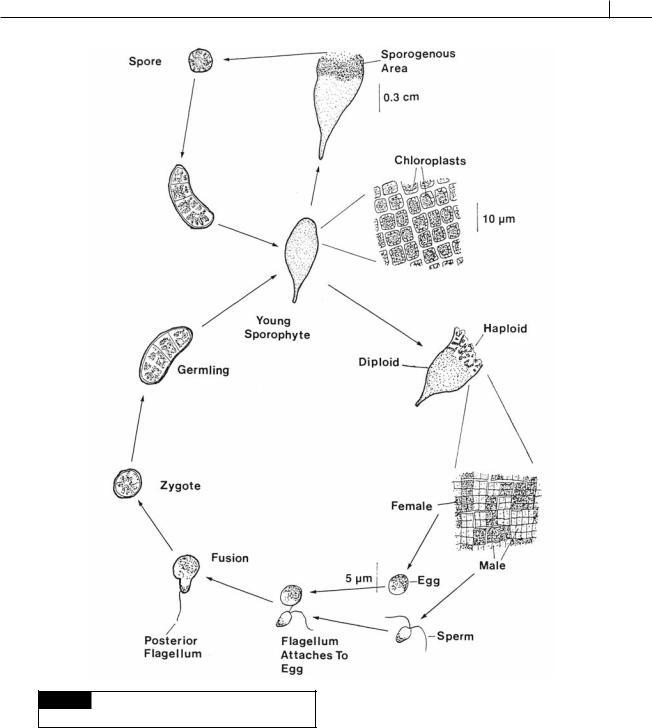
zygote swims for several hours, retracts the flagellum, sinks, attaches firmly to the substratum, and develops a cell wall. The zygotes germinate into diploid thalli, completing the life cycle (Friedmann, 1959).
Chlorellales
In this order are the algae that have a non-motile vegetative thallus where the thallus comprises only a single cell or a coenobium composed of a definite number of cells arranged in a specific manner. Asexual reproduction occurs by zoospores or aplanospores that are commonly autospores. These autospores are probably no more than daughter cells of the parent thallus. Sexual reproduction can be isogamous, anisogamous, or oogamous. The order is exclusively freshwater.
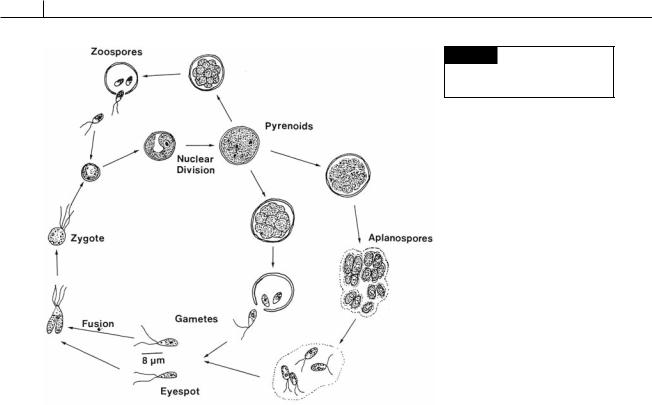
Chlorococcum (Fig. 5.81) is an alga frequently isolated from beneath the surface of the soil. It sometimes occurs in abundance on damp soil or brickwork. The cells can survive for a long time in the soil. In one study they grew after being in desiccated soil for 59 years (Trainor, 1970). The cells of the same species vary considerably in size, young cells having a thin cell wall and older cells a thick one. The chloroplast in young cells is a massive parietal cup with a single pyrenoid. In older
cells the chloroplast becomes diffuse. Asexual reproduction is by zoospores, there never being any vegetative division. The biflagellate zoospores have a cup-shaped chloroplast and an eyespot. Sexual reproduction occurs by the formation of isogametes. Under certain conditions, aplanospores can be produced, which eventually liberate two to four biflagellate gametes. Nearly dormant cells of Chlorococcum echinozygotum that have been deprived of nitrogen can be induced to undergo gametogenesis by being placed in fresh medium in darkness without nitrogen (O’Kelley, 1984). If nitrogen is supplied 6 hours after the cells are placed in darkness, zoosporogenesis occurs instead of gametogenesis.
Hydrodictyon reticulatum (Fig. 5.82), or the water net, is a free-floating, relatively rare freshwater alga that forms colonies that are net-like with polygonal or hexagonal meshes. The net is formed by multinucleate elongated cells joined end to end, probably by lectins such as concanavalin A (Hatano and Ueda, 2000). Each cell has a large central vacuole and a reticulate chloroplast with pyrenoids. In asexual reproduction a daughter net is produced inside, and subsequently released from, the parent cell (Pocock, 1960; Marchant and Pickett-Heaps, 1971, 1972a–d). Asexual reproduction begins with the disappearance of pyrenoids, coinciding with the accumulation of starch grains in the fragmenting chloroplasts. Regularly spaced nuclei are surrounded by fragments of chloroplasts. A vesicle forms around and outside of the tonoplast, restricting most of the proto-
CHLOROPHYTA 213
Fig. 5.80 The life cycle of Prasiola stipitata. (Adapted from
Friedmann, 1959.)
plasm to a small area around the periphery of the cell. The protoplasm divides to produce uninucleate, biflagellate zoospores that actively swim about inside the parent cell. The zoospores stop swimming, become joined in certain areas, and retract their flagella. A daughter net has now been
formed within the parent cell, which is released to enlarge to a mature colony.
Chlorella cells are spherical with a cup-shaped chloroplast (Fig. 5.83). The only method of reproduction is by daughter cells that resemble the parent cell. Chlorella often forms intracellular symbioses with aquatic invertebrates and protozoa (such as Paramecium). In these symbioses, Chlorella is held in host vacuoles where the
214 EVOLUTION OF THE CHLOROPLAST
Fig. 5.81 The life cycle of
Chlorococcum humicola. (Adapted
from Bristol, 1919.)
Chlorella synthesizes and releases maltose into the host vacuole. Chlorella is held at a low pH in the host vacuoles. Studies on isolated Chlorella cells have shown that maximum synthesis and release of maltose occurs at pH 4–5 (Dorling et al., 1997).
Prototheca lacks chlorophyll and resembles a colorless Chlorella although the Prototheca cells do have starch-containing amyloplasts (Webster et al., 1968). Prototheca causes protothecosis in animals and humans. This disease is more common than is supposed, but because the small round colorless cells resemble yeasts, the disease, at least in animals, is often incorrectly diagnosed. Prototheca cells are fairly common in the soil, where they presumably live by digesting organic matter, and it is from here that most infections occur. In animals most of the reported cases have been severe systemic infections, such as massive invasion of the bloodstream, that have resulted in the death of the animal within a short time (van Kruiningen et al., 1969). Such massive infections have been reported in humans but usually only as secondary invaders after the primary invader has seriously lowered the body’s defenses. More common in humans is the subcutaneous type of infection
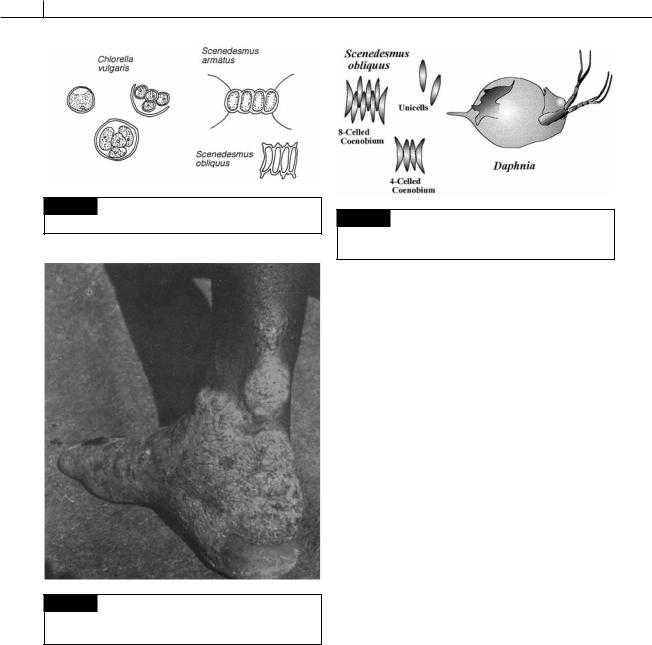
that starts initially as a small lesion and spreads slowly through the lymph glands, covering large areas of the body and preventing the sufferer from performing normal work duties (Fig. 5.84) (Mars et al., 1971).
Scenedesmus is a common alga (Fig. 5.83), often occurring as almost a pure culture in plankton. Cells in the colony occur in multiples of two, with four or eight cells being most common. The species differ mostly in the number and type of spines on the cells and the texture of the wall. The uninucleate cells have a single laminate chloroplast. The morphology of the colony can be varied considerably by varying the medium in which the cells are growing (Egan and Trainor, 1989). In a medium with low phosphorus or low salt concentration, the Scenedesmus is induced to grow as unicells, resembling the genera Chodatella and Franceia (Trainor, 1992) and the same species can also be induced to grow with or without spines. Composed of aggregated proteinaceous tubules, the spines probably aid in the flotation of the colonies (Staehelin and Pickett-Heaps, 1975). Scenedesmus occasionally forms zoospores when deprived of nitrogen (Trainor, 1963).

CHLOROPHYTA 215
Fig. 5.82 The life cycle of Hydrodictyon reticulatum.
(Adapted from Pocock, 1960; Marchant and Pickett-Heaps,
1971, 1972a–d.)
216 EVOLUTION OF THE CHLOROPLAST
Fig. 5.83 Drawing of Chlorella vulgaris (with autospores),
Scenedesmus armatus, and S. obliquus.
Fig. 5.85 Scenedesmus obliquus without grazers occurs primarily as unicells. When grazers such as Daphnia are present, S. obliquus grows primarily as coenobial colonies.
Fig. 5.84 Lesions of protothecosis on the foot and lower leg of a man from Sierra Leone. (From Davies and Wilkinson, 1967.)
Many species of Scenedesmus grow as unicells in an environment without grazers. In the presence of grazers, such as Daphnia (Fig. 5.85), Scenedesmus forms multicellular colonies that are not grazed as heavily. Infochemicals produced by the grazers induce the formation of coenobia. Infochemicals are chemicals that, in the natural context, convey information in an interaction between two organisms, evoking a behavioral or physiological response in the receiving organism, which is adaptively favorable to one or both organisms in the interaction. In the case of
Scenedesmus, the infochemicals originate from the digestive tract of the grazer. Starved grazers do not produce infochemicals. In the multicellular state, Scenedesmus is not grazed as heavily by Daphnia. However, the advantages to Scenedesmus in the unicellular state include smaller sinking velocity and advantageous surface to volume ratio in terms of nutrient uptake and light harvesting (Lurling, 1998, 1999).
Unicellular green algae, such as Chlorella, have been extensively investigated as possible new sources of food for an increasing world population (Yamaguchi, 1997). Chlorella produces little cellulose or other carbohydrate wall material; thus more of the cell is digestible in Chlorella than in an alga with a large amount of cellulose. Exponentially growing Chlorella cells contain about 50% protein, 5% chlorophyll, and a large number of vitamins. Much greater quantities of biomass can be obtained per unit area with algae than with higher plants. When the growth of algae is linked to the purification of sewage in oxidation ponds, yields of 112 000 kg hectare 1 year 1 of dried algae can be obtained. At the same time, the algae are taking up elements such as nitrogen and phosphorus, and reducing the biological oxygen demand (an indication of the organic material in the water) by 85% (McGarry and Tongkasame, 1971). The algae obtained make a very good stock feed, and attempts have been made to utilize it as human food, although the algae in the human digestive tract lead to undesirable intestinal floras that often result in gas in the intestine. A second line of investigation in this
