
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index

496 CHLOROPLAST E.R.: EVOLUTION OF TWO MEMBRANES
The external coccoliths can be removed by lowering the pH of the culture medium. Cells of Coccolithus huxleyi decalcified in this manner may acquire a complete coccolith envelope (about 15 coccoliths) within 15 hours of their being transferred back to a normal medium in the light. Complete recalcification in Cricosphaera sp. may require 40 hours. In both instances, cell division is not a prerequisite for the formation of new coccoliths. If the organisms are grown in artificial seawater, the coccoliths dissolve if the product of the concentrations of calcium and carbonate is appreciably smaller than the solubility product of calcite. In C. huxleyi, coccoliths are still formed inside cells even when the calcium content of the medium is reduced to levels where external coccoliths dissolve, although coccolith production is retarded at calcium concentrations less than half that of normal seawater. A photochemical process is apparently directly associated with coccolith production because when light is turned off, there is a sharp drop in coccolith production.
Pleurochrysis carterae has biflagellate cells surrounded by a coccosphere consisting of a single layer of 100 to 200 coccoliths. The cells incorporate calcium into extracellular coccoliths at a more or less constant rate throughout a 16-hour light : 8-hour dark cycle. The cells divide during the dark periods with a concomitant decrease in cell size during the dark period followed by an increase in cell size during the light period. The cells form coccoliths in the light as well as in the dark at a similar rate (van der Wal et al., 1987) (although Emiliania huxleyi (Fig. 22.16(b)) produces coccoliths only during the light period (Linschooten et al., 1991).
Although coccolithophorids constitute a minor part of recent calcareous oozes (bottom sediments composed of calcified remains of organisms) in the ocean, in the Cretaceous they dominated the calcareous nanoplankton (Tasch, 1973). This domination paralleled an increase in Ca2 in seawater at the time (Brennan et al., 2004). Coccolithophorids provided the major constituent of Mesozoic (Jurassic and Cretaceous) and Tertiary chalks and marls. The abundance of coccolithophorids in these chalks can be demonstrated by taking a piece of ordinary blackboard chalk, pulverizing it, mixing it with distilled
water in a test tube, and letting it stand for 20 minutes. Draw some of the solution into a pipette, dispose of the first four to five drops, and place the next few drops on a slide. Place a cover slip on the slide, and view it at a magnification of 400 to 500 . Many coccoliths and other remains will be seen.
Coccoliths in sedimentary rocks can be used as markers in the discovery and mode of deposition of oil deposits. For example, the oil shales of the Kimmeridge Clays in England are sandwiched between limestone bands that are composed mostly of coccoliths of one species,
Ellipsagelosphaera britannica (Gallois, 1976). Other oil-bearing rocks have similar characteristic coccoliths. Therefore petroleum geologists know that when a drill core shows certain coccoliths that are associated with petroleum, there is a good chance of finding oil in that stratum of rock.
Toxins
The prymnesiophycean alga Prymnesium parvum (Fig. 22.7) secretes the potent exotoxin prymnesin (Fig. 22.17). The toxin causes fish mortalities by increasing cell membrane permeability and disturbing cellular ion balance (Fistarol et al., 2003). The toxin is most effective against aquatic gillbreathing animals, such as fish and molluscs. In Amphibia, only the gill-containing tadpole stage is sensitive to immersion in solutions containing the ichthytoxin. The rapidity of the action of Prymnesium toxin on immersed fish suggests that the immediate target must be an exposed organ, probably the gill. Experiments have shown that the toxins affect the permeability of the gill, resulting in the increased sensitivity of the fish. In fish removed promptly from such toxin solutions, the gill damage is repaired within hours. In Israel, Shilo (1967) has found that it is possible to control P. parvum in fish breeding ponds by adding small amounts of ammonium salt, which causes the algal cells to lyse.
Secretion of prymnesin by Prymnesium parvum immobilizes prey organisms and enables P. parvum to more easily seize its prey (Skovgaard and Hansen, 2003). Prymnesium produces higher quantities of toxin when phosphorus is limiting
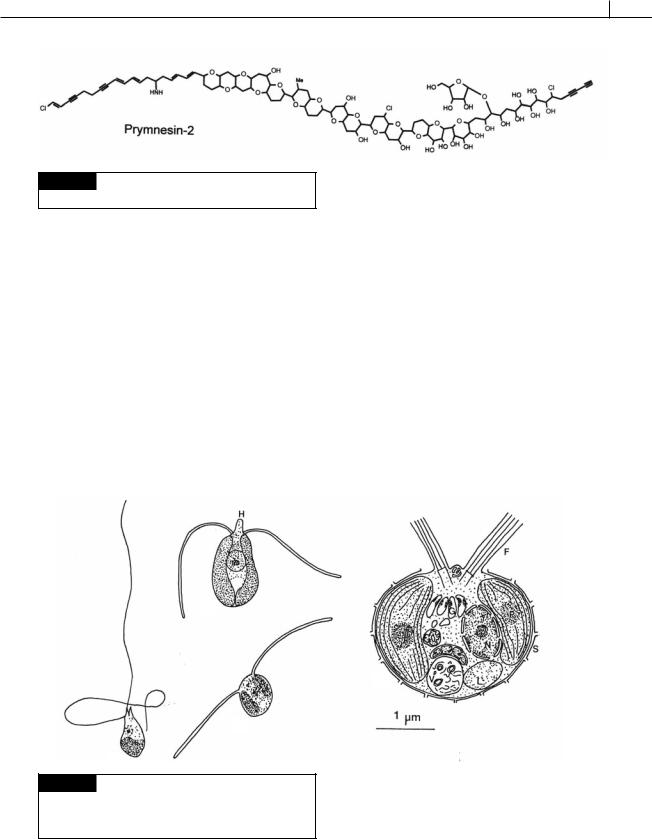
PRYMNESIOPHYTA 497
Fig. 22.17 The chemical structure of prymnesin-2. (After
Igarashi et al., 1996.)
(Legrand et al., 2001). Secretion of the toxin results in greater kill of prey organisms which are subsequently ingested by the Prymnesium cells. The phosphorus in the prey organisms alleviates the phosphorus deficiency in the Prymnesium cells, resulting in decreased production of toxin.
Some species of Chrysochromulina produce toxins that kill fish, mussels, and ascidians (Hansen et al., 1995; Moestrup, 1994; Simonsen and Moestrup, 1997). The best documented fish kills have occurred off the coast of Norway and Sweden. The large blooms of Chrysochromulina causing the fish kills have been associated with a lack of predation by the normal ciliate grazers of
Chrysochromulina. It appears that the long spines on the surface of the Chrysochromulina cells make them too large to be taken up by the ciliates (Hansen et al., 1995).
In the North Sea of Europe, and in the seas off Antarctica, blooms of the prymnesiophyte Phaeocystis (Figs. 22.4, 22.18(a)) occur as macroscopic lobed colonies or “bladders” in the spring and fall. Phaeocystis colonies are hollow, balloonlike structures with individual cells lying beneath a thin mucous skin (Hamm et al., 1999; Solomon et al., 2003). Grazing by invertebrates results in colonies of larger size, the larger size induced by chemicals released into the water by the grazing (Tang, 2003). Colony formation and enlargement is a defense mechanism that results in clogging of the filtration apparatus of the grazers (Haberman et al., 2002).
(b)
(d)
(c)
(a)
Fig. 22.18 (a) Motile cell of Phaeocystis poucheti.
(b) Isochrysis galbana. (c), (d) Imantonia rotunda. (F) Flagellum;
(H) haptonema; (L) leucosin vesicle; (N) nucleus;
(P) pyrenoid; (S) scale. ((d) after Reynolds, 1974.)
