
The Nitro Group in Organic Synthesis
.pdf
6.1 |
NEF REACTION (ALDEHYDES, KETONES, AND CARBOXYLIC ACIDS) 161 |
||||||
|
O |
|
|
|
|
|
O |
|
O |
|
O |
||||
|
|
|
|
||||
|
|
|
|
1) LiOCH3 |
|
|
(6.5) |
|
|
|
|
2) KMnO4 |
O |
||
|
|
|
|
|
|||
|
NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
95% |
|
|
|
|
|
|
|
|
|
O Me Me |
|
|
|
|
|
|
O Me Me |
|
|
1) t-BuOK |
ButO |
CHO |
|||
ButO |
NO |
|
|
|
|
(6.6) |
|
|
2) KMnO4, 0 ºC, 10 min |
||||||
|
|
||||||
Me Me |
2 |
|
|
Me Me |
|||
|
|
|
|
|
|
95% |
|
|
|
|
|
|
|
|
|
When the substrate does not contain a reactive carbon–carbon double bond, the ozonolysis procedure appears to provide a convenient and efficient method for the conversion of primary and secondary nitro compounds into carbonyl compounds (Eq. 6.7).12
O |
O |
|
|
||
|
1) CH3ONa |
(6.7) |
|
2) O3 |
|
|
O |
|
NO2 |
73% |
|
|
|
|
Anodic oxidation of nitronate anions provides an attractive method for the Nef reaction (Eq. 6.8).20
O |
O |
|
|
(CH2)6CH3 |
|
||
(CH2)6CH3 |
electrolysis |
|
|
CH3OH, HCO2Na |
O |
|
|
|
|
||
NO2 |
90% |
(6.8) |
|
|
|
||
Dimethyldioxirane oxidation of nitronates anions affords the corresponding carbonyl products. Highest yields are obtained when one equivalent of water is added before the oxidation (Eq. 6.9).17
O |
1) t-BuOK |
O |
|
|
|||
|
2) H2O, 2 min |
(6.9) |
|
|
3) O |
||
NO2 |
O |
||
O |
|||
80% |
|||
|
|
Sodium chlorite under phase-transfer catalysis conditions (CH2Cl2-NaOH-Bu4NHSO4) is also a good choice for the Nef reaction of primary and secondary nitro compounds (Eq. 6.10).16
O |
|
|
|
O |
|
|
|
|
|
|
|
|
NaClO2, CH2Cl2 |
(6.10) |
|
|
|
|
|
|
|
NaOH, Bu4N+HSO4– |
||
|
NO2 |
CHO |
||
|
|
|
||
|
|
|
67% |
|
|
|
|
|
|

162 CONVERSION OF NITRO COMPOUNDS INTO OTHER COMPOUNDS
A very mild oxidative transformation of nitro compounds into ketones using tetrapropylammonium perruthenate (TPAP) has been developed. A stoichiometric amount of TPAP in the presence of N-methylmorpholine N-oxide (NMO) and 4 A molecular sieves (MS).18a As the reaction conditions are neutral and mild, this method is compatible with the presence of other sensitive functionalities (Eq. 6.11). This transformation can be carried out with 10 mol% of TPAP and 1.5 equiv of NMO in the presence of potassium carbonate, 4 A MS, and silver acetate (Eq. 6.12).18b
HO |
O |
|
|
||
TPAP, NMO |
(6.11) |
|
4 Å MS |
O |
|
NO2 |
83% |
|
NO2 |
O |
|
OCH2Ph |
||
OCH2Ph TPAP (10 mol%) |
||
(6.12) |
||
NMO, AgOAc |
||
|
||
|
62% |
Secondary nitro compounds are converted into ketones under very mild conditions using n-propyl nitrite and sodium nitrite in DMSO.21
The Nef reaction of primary nitro compounds gives aldehydes or carboxylic acids, depending on the reaction conditions. Each transformation provides an important tool in organic synthesis. Primary nitro compounds are converted into carboxylic acids with concentrated mineral acids.22 Because such harsh conditions also lead to side reactions, a milder method is required in organic synthesis. Basic phosphate-buffered KMnO4 rapidly converts primary nitroalkanes into carboxylic acids in 90–99% yield (Eq. 6.13).23
KMnO4, KOH
HO(CH2)11CH2NO2  HO(CH2)11CO2H (6.13)
HO(CH2)11CO2H (6.13)
K2HPO4, t-BuOH
Scheme 6.1 shows a simple preparation of (+)-isomintlactone, isolated from a sample of peppermint oil. The addition of pyrrolidino enamine of (R)-3-methylcyclohexanone to ni-
O |
|
|
NR2 |
O |
pyrrolidine |
|
|
NO2 |
|
|
|
|
||
|
|
85% |
NO2 |
|
|
|
85% |
||
|
|
OH |
|
H |
|
|
|
O |
|
Baker's yeast |
|
1) KMnO4 |
||
|
O |
|||
|
|
|
NO2 2) p-TsOH |
|
|
|
|
|
H |
|
|
72% |
|
68% |
|
|
H |
|
H |
LDA, MeI |
|
O |
1) Ph2Se2 |
O |
|
O |
O |
||
|
|
H |
2) H2O2 |
|
|
|
|
|
|
|
|
91% |
|
55% |
Scheme 6.1.

6.1 NEF REACTION (ALDEHYDES, KETONES, AND CARBOXYLIC ACIDS) 163
troethylene followed by reduction with baker’s yeast and the Nef reaction gives the condensed lactone. Methylation followed by olefination using α-phenylselenation and oxidation yields the desired lactone (Scheme 6.1).24
Strong oxidizing agents such as KMnO4 are not always useful for synthesis of highly sensitive compounds. In 1956, Kornblum found that a mixture of nitrite ester and sodium nitrite oxidizes primary nitro paraffins to the corresponding carboxylic acids.25 However, the reaction was prone to poor yields and long reaction time. Forty years later, a very efficient related reaction using a mixture of sodium nitrite and acetic acid in DMSO has been reported (Eqs. 6.14 and 6.15).26 Because carbon–carbon double and triple bonds and other various functional groups are compatible with this reaction condition, this method is useful for natural product synthesis. For example, one-synthesis of alkaloid, (–)-horsfiline is shown in Scheme 6.2, in which the creation of chiral quaternary carbon centers via an addition-elimination process is used as the key reactions.27 This strategy has been used very effectively for the synthesis of various natural products; this is discussed in Chapter 4 on the Michael addition to nitroalkenes.
|
|
|
|
|
|
|
NaNO2, AcOH |
CO H |
|
|
|
|
|
|
|||
|
|
5 NO2 |
|
|
|
|
|
2 |
|
|
|
|
|
(6.14) |
|||
|
|
|
|
DMSO, 30 ºC, 6 h |
5 |
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
95% |
|
|
|
|
|
|
|
TMS |
|
|
|
|
|
|
NO2 |
NaNO2, AcOH |
TMS |
|
|
|
|
|
CO2H |
(6.15) |
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
DMSO, 30 ºC, 6 h |
|
|
98% |
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
Because primary alkyl bromides can be converted into the corresponding nitro compounds by the action of NaNO2 in DMSO, primary alkyl bromides are converted directly into the corresponding carboxylic acids by the reaction with an excess of sodium nitrite in acetic acid (Eq. 6.16).26
MeO |
|
|
|
MeO |
|
NO2 |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
NaBH4 |
|
|
|
|
|
|
|
|
|
|
|
|
Ph |
|
O |
|
O |
N |
O |
|
Ph |
N |
|
, MeOH |
|
N |
|
|
O |
||||
|
R |
|
|
||||
R |
|
|
OMe |
|
|
|
|
|
|
|
95% |
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
MeO |
|
NO2 |
|
MeO |
|
|
|
|
|
|
|
|
|
|
|
|
|
NaNO2 |
|
|
CO2H |
|
|
N |
O |
AcOH, DMSO |
N O |
|
|
||
|
|
|
|
||||
|
|
|
|
|
|||
R |
|
|
|
R |
|
|
|
97% |
|
|
|
80% |
|
|
|
Me
N
MeO
N O R
(–) - horsfiline
Scheme 6.2.

164 CONVERSION OF NITRO COMPOUNDS INTO OTHER COMPOUNDS
|
|
O |
|
BnO |
NaNO2 |
BnO |
|
Br |
AcOH, DMSO |
OH |
|
O |
O |
||
|
96% (6.16)
6.1.3 Reductive Method
The Nef reaction can also be carried out with reducing agents. Aqueous titanium chloride reduces nitro compounds to imines, which are readily hydrolyzed to carbonyl compounds (Eq. 6.17).28 The Michael addition of nitroalkanes to enones followed by reaction with TiCl3 provides an excellent route to 1,4-diketones and hence to cyclopentenones. For example, cis-jasmone is readily obtained,28 as shown in Eq. 6.18.
|
|
|
|
|
CH3(CH2)4CH2NO2 |
|
|
|
TiCl3, H2O |
|
CH3(CH2)4CHO |
(6.17) |
||||||||||||||||||||
|
|
|
|
|
|
|
|
dimethoxyethane |
||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
80% |
|
|
|
|
|
|
|||
|
|
|
|
|
|
CH2NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
|
, CH2Cl2 |
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
O |
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
TiCl3, H2O |
|
|
|
|
|
|
|
|
|
|
|
1) base |
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
dimethoxyethane |
|
|
|
|
|
|
|
|
|
|
2) H2 / Lindler |
|
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
85% |
|
|
|
|
|
|
|
|
|
|
|
|
|
cis-jasmone |
|||||||
(6.18) The TiCl3 solution is very acidic such that acid-sensitive compounds do not survive. In such cases, sodium acetate or ammonium acetate is added to the reaction mixture to control pH at 5–6.29 Highly functionalized substrates are synthesized by the Nef reaction using TiCl3 and
sodium acetate, as shown in Eq. 6.19,30 and Eq. 6.20.31
|
|
|
CH2Ph |
|
|
|
|
|
|
|
|
|
|
CH2Ph |
|||||
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
N |
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
O N |
|
NO2 |
|
TiCl3 |
|
|
|
O |
|||||||||||
|
|
|
|
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
(6.19) |
|||||||||||
|
|
|
|
|
|
|
|
AcONa |
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
63% |
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
OH |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
CH |
CH(NO |
)(CH ) CO CH |
3 |
1) NaOCH3, CH3OH |
||||||||||
2 |
2 |
2 4 |
2 |
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
2) TiCl3, NH4O2CCH3, H2O |
||||||
RO CH=CHCH(OR)n-C5H11 |
|
||||||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
OH |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
CH2CO(CH2)4CO2CH3 |
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
RO CH=CHCH(OR)n-C5H11 |
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
70% |
|
|
|
|
(6.20) |
|||||
Vanadium chloride,32 chromium chloride,33 and the combined use of tributylphosphine and diphenyldisulfide34 are also effective in promoting the reductive Nef reaction.

6.1 NEF REACTION (ALDEHYDES, KETONES, AND CARBOXYLIC ACIDS) 165
6.1.4 Direct Conversion of Nitroalkenes to Carbonyl Compounds
Because nitroalkenes are directly prepared by the condensation of aldehydes with nitroalkanes, the conversion of nitroalkenes into carbonyl compounds provides a powerful tool in organic synthesis. In general, the reduction of nitroalkenes followed by the hydrolysis or oxidation gives carbonyl compounds. Old procedures for this transformation involve the reduction with FeHCl35 or Zn-AcOH.36 There are many modified procedures for the conversion of 1-phenyl-2- nitropropene into phenylacetone (Eq. 6.21); method A37: Raney Ni-NaH2PO2, method B38: Cr(2)Cl, method C39: LiBHR3/HCl, method D40: CdCl2-Mg-H2O, method E41: electrochemical reduction using Pb as an electrode.
|
|
|
|
|
|
|
Method |
|
Yield (%) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A |
Raney Ni/NaH2PO2 |
88 |
NO2 |
|
|
O |
B |
CrCl |
80 |
|||
|
|||||||||
|
|
|
|
|
|
|
|
2 |
|
|
|
|
|
|
|
|
C |
LiBHR3, HCl |
80 |
|
|
|
|
|
|
|
D |
CdCl2 -Mg-H2O |
80 |
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
E |
Electrochemical reduction, Pb |
80 |
|
|
|
|
|
|
|
|
|
|
(6.21)
The direct conversion of nitroalkenes into ketones is especially useful for the preparation of arylacetones. They are readily prepared by the condensation of aromatic aldehydes with nitroethane and by the subsequent Nef reaction.42 Typical examples are presented in Eq. 6.2237 and Eq. 6.23; the product of Eq. 6.23 is used for total synthesis of perylenequinone, calphostin D, which is a potent inhibitor of protein kinase C.42b
|
Me |
|
|
|
O |
|
|
|
|
|
|
|
NO2 |
Raney Ni |
|
|
Me |
|
|
|
(6.22) |
||
|
|
NaH2PO2 |
|
|
|
|
|
|
|
|
|
N |
CF3 |
|
|
N |
CF3 |
|
|
86% |
|||
|
|
|
|
||
OMe OAc |
|
|
|
OMe OAc |
|
|
|
|
|
|
|
|
Me |
CrCl2 |
|
|
O |
|
THF - H2O |
|
|
|
|
|
|
|
MeO |
Me |
|
MeO |
NO2 |
|
|
||
|
|
|
72% |
||
|
|
OH |
O |
Me |
|
|
|
|
|||
|
|
|
|
O |
|
|
|
|
|
OH |
|
|
|
MeO |
|
Me |
(6.23) |
|
|
MeO |
|
Me |
|
|
|
|
|
||
|
|
|
|
OH |
|
|
|
|
|
O |
|
|
|
OH |
O |
Me |
|
Iodotrimethylsilane generated in situ from chlorotrimetylsilane and sodium iodide effects the reduction of nitroalkenes into ketones at 0 °C. This method is useful for the conversion of nitro steroids or nitro terpenoids to the corresponding ketones (Eq. 6.24).43
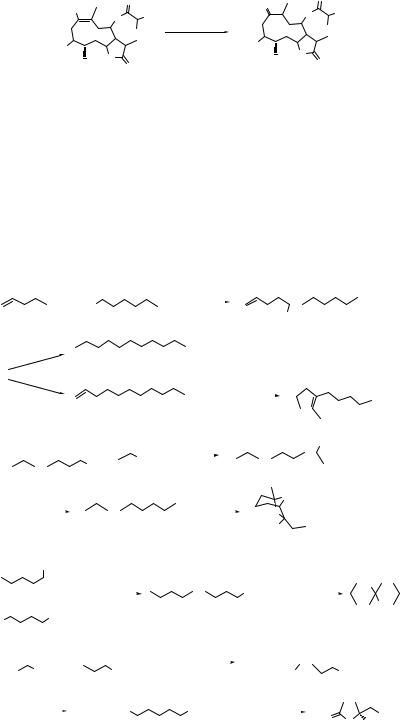
166 CONVERSION OF NITRO COMPOUNDS INTO OTHER COMPOUNDS
|
O |
|
O |
O |
|
O2N |
|
|
Me |
||
|
|
|
|||
|
Me |
O |
|
||
O |
|
Me |
|
||
|
Me |
Me3SiCl-NaI |
|
(6.24) |
|
|
|
AcO |
|
||
AcO |
|
|
|
|
|
|
|
O |
|
|
|
O |
|
|
O |
|
|
O |
|
78% |
|
||
|
|
|
|||
|
|
|
|
Ballini and his coworkers have used the strategy based on the Nef reaction after the carbon–carbon bond formation for the synthesis of various natural products.44 The requisite nitroalkenes are simply prepared by the nitro-aldol condensation using Al2O3. When double bonds are present in nitroalkenes, the reductive Nef reaction gives saturated compounds, as shown in Eq. 6.25.45 A new simple, cheap, and practical procedure for the direct transformation of nitroalkenes into ketones has been obtained by the NaBH4/H2O2 system. By this method, other functional groups such as double bonds, acetals, or aromatic nitro groups are preserved. A convenient synthesis of dihydrojasmone and brevicomine using this transformation is presented in Eqs. 6.25 and 6.26, respectively.46 Other applications of the Nef reaction to the synthesis of natural products such as spiroketalic pheromones and lactonic pheromones are demonstrated in Eqs. 6.27 and 6.28.47
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
Al2O3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
+ |
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O2N |
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
63% |
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
Raney Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
NaH2PO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
60% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
NaBH4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1) PdCl2/ CuCl / O2 |
|
|
|
|
|
|
|
|
|
|
|
|
(6.25) |
||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
|
|
H2O2 |
|
|
|
|
|
|
|
O |
|
|
|
|
|
2) base |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
58% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
Al2O3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|
||||||||||||
|
|
|
|
|
|
|
|
|
|
+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
H |
|
NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
65% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
NaBH4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1) mCPBA |
O |
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||
|
|
|
|
|
|
H2O2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2) TsOH |
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
(6.26) |
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
70% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
88% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
OH |
NO2 |
|
|
|
|
|
|
|
|
|
OH |
|
NO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
+ |
|
|
|
|
|
|
Al2O3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OTHP |
|
1) NaBH4 |
|
|
|
|
|
|
||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
40 ºC |
|
|
|
|
|
|
|
|
|
|
|
|
|
2) H+ |
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
|
|
O |
|
|
|
|
|
|
|
|
|
54% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
OTHP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(6.27) |
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
|
|
|
O |
|
|
|
|
|
|
NO |
2 |
|
|
|
|
|
|
|
|
|
Al |
O |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
|
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
50 ºC |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
n-C11H23 |
H |
|
|
|
|
|
|
|
CO2Me |
|
|
|
n-C |
11 |
H |
CO Me |
||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
23 |
|
|
|
|
|
|
2 |
|
|
|
|
|
||||||
|
|
Raney Ni |
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
Baker's yeast |
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||
|
|
|
|
|
|
n-C11H23 |
|
|
|
|
|
|
OMe |
|
|
|
|
|
|
|
n-C11H23 |
|||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
||||||||||||||||||||
|
|
NaH2PO2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H |
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
(6.28)

6.2 NITRILE OXIDES AND NITRILES 167
The 4-hydroxyheptadecan-7-one, isolated from the root of Chiococca alba, is simply prepared using hydroxy-functionalized nitroalkenes, as shown in Eq. 6.29.48
NO2 |
NO2 |
|
+ CH3(CH2)8CHO |
Al2O3 |
|
OH |
||
OH |
||
45% |
||
|
||
Raney Ni |
O |
|
(6.29) |
||
NaH2PO2 |
||
OH |
||
|
60% |
The Henry reaction of ketones with nitroalkanes in the presence of ethylenediamine gives allylic nitro compounds, which give α,β-unsaturated carbonyl compounds via the Nef reaction (Eq. 6.30).49
CHO
CH2NO2 1) NaOMe
(6.30)
2) TiCl3
66%
6.2 NITRILE OXIDES AND NITRILES
Primary nitro compounds are good precursors for preparing nitriles and nitrile oxides (Eq. 6.31). The conversion of nitro compounds into nitrile oxides affords an important tool for the synthesis of complex natural products. Nitrile oxides are reactive 1,3-dipoles that form isoxazolines or isoxazoles by the reaction with alkenes or alkynes, respectively. The products are also important precursors for various substrates such as β-amino alcohols, β-hydroxy ketones, β-hydroxy nitriles, and β-hydroxy acids (Scheme 6.3). Many good reviews concerning nitrile oxides in organic synthesis exist; some of them are listed here.50–56 Applications of organic synthesis using nitrile oxides are discussed in Section 8.2.2.
|
PhNCO |
R |
|
C |
|
NO |
RCH2NO2 |
Et3N |
|
|
|||
|
|
|||||
|
|
|||||
|
|
|||||
PCl3 |
|
|
|
(6.31) |
||
|
|
|
|
|||
|
|
R |
|
C |
|
N |
|
|
|
|
|||
|
|
|
|
|||
|
|
|
|
|||
The dehydration of primary nitro compounds with phenyl isocyanate (PhNCO) and triethylamine has been widely used for generating nitrile oxides (Mukaiyama-Hoshino method).57a Polymer-bounded nitrile oxide cycloaddition reactions employing phenyl isocyanate have experimental advantage of removing the urea by-product by simply washing the resin with solvent.57b The use of diisocyanates for in situ preparation of nitrile oxides from primary nitroalkanes is of similar advantage, in which the by-product urea polymer is simply removed by filtration.57c An alternative method for generating nitrile oxides is based on the chlorination of oximes. However, the former method is better, because the use of chlorinating agents can be avoided. Recently, some new preparing methods of nitrile oxides from nitro compounds have been achieved, as shown in Eqs. 6.32–6.35; however, the Mukaiyama-Hoshino method is still the most widely used in organic synthesis.

168 CONVERSION OF NITRO COMPOUNDS INTO OTHER COMPOUNDS
R CNO |
|
O |
|
O |
|
1,3-dipolar addition |
|
||
+ |
N |
1) Raney Ni, H2 |
R |
|
|
|
R |
2) Al2O |
|
|
|
|
|
|
|
|
2-isooxazoline |
|
|
LiAlH4 |
O OH |
|
NC |
R |
|
OH |
||
|
||
NH2 OH |
|
|
R |
|
Scheme 6.3.
For example, the reaction of nitroalkanes with di-tert-butyl dicarbonate, (BOC)2O, and 4-dimethylaminopyridine (DMAP) as catalysts in the presence of dipolarophiles at room temperature affords cycloadducts in improved yields compared with the Mukaiyama-Hosino method.58 The conversion of Eq. 6.32 gives a 90% yield by this procedure, whereas the conventional method using PhNCO gives a 79% yield of the same product. An additional advantage of this new method is that the use of (BOC)2O allows the reaction to be carried out with substrates that contain NH or OH groups without prior protection. The cycloaddition leads directly to protected N- or O-Boc products (see Eq. 6.33).
|
|
|
N |
O |
Ph |
|
|
|
|
|
|
|
|
||
|
|
(Boc)2O, DMAP |
|
|
|
|
|
MeCH2NO2 + PhCH=CH2 |
|
|
|
|
(6.32) |
||
20ºC |
Me |
|
|
|
|||
|
|
|
|
|
|
||
|
|
|
|
90% |
|
|
|
Ph |
NO2 |
|
|
|
Ph |
N |
O |
|
|
|
|
|
O |
||
|
|
|
|
|
|
|
|
|
O |
(Boc)2O, DMAP |
|
|
|
|
|
|
+ |
|
|
|
|
OEt |
|
|
20ºC |
|
|
|
|
||
N |
OEt |
|
|
N |
|
||
|
|
|
|
|
|||
H |
|
|
|
|
|
Boc |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
88% |
(6.33) |
The reaction of nitroalkenes or nitroalkanes with TiCl4 and Me3SiN3 gives α-azido functionalized hydroxamoyl chlorides, which act as precursors of nitrile oxides (Eq. 6.34).59
|
|
|
N3 |
O |
N3 |
|
||
|
|
|
Ph |
|
||||
|
|
Me3SiN3 |
Cl |
|
|
|
||
|
|
OEt |
|
|
||||
Ph |
NO2 |
Ph |
|
(6.34) |
||||
|
|
|
|
|
||||
TiCl4 |
N |
Et3N, RT, 8 h |
N |
|||||
|
OEt |
|||||||
|
|
|||||||
|
|
RT 1 h |
OH |
|
|
O |
|
|
|
|
|
78% |
|
|
88% |
|
|
|
|
|
|
|
|
|
||
A new route to nitrile oxides based on the reaction of primary alkyl bromides with NaNO2 in the presence of acetic acid, is also reported (Eq. 6.35).60 This reaction is used for the direct

6.2 NITRILE OXIDES AND NITRILES 169
conversion of alkyl bromides or nitroalkanes to carboxylic acids on treatment with NaNO2 and acetic acid, if the alkenes are absent. The reaction proceeds via nitrolic acids, which can be isolated when the reaction is carried out at 20 °C. When heated in THF, they allow corresponding nitrile oxides to be obtained under neutral conditions.60b
|
NaNO2, AcOH |
N |
O |
CO2Me |
PhCH2Br + |
|
|||
|
|
|
(6.35) |
|
DMSO, RT |
|
|
||
CO2Me |
Ph |
|
|
|
|
|
68% |
|
|
|
|
|
|
Primary nitro ketones, ethyl nitroacetate, and (phenylsulfonyl)nitromethane react with alkenes in the presence of Lewis acids to give nitrile oxide cycloaddition.61a Similarly, the reaction of α-nitro ketones with TeCl4 generates the corresponding nitrile oxides, as shown in Eq. 6.36.61b
O |
TeCl4, Et3N |
O |
Ph |
|
N |
||||
NO2 + Ph |
(6.36) |
|||
–78ºC ~RT |
|
|||
Ph |
Ph |
|
||
|
|
|
||
|
|
O |
|
|
|
|
82% |
|
The conversions of nitro compounds into nitrile oxides have been used extensively in organic synthesis (see Section 8.2.2). On the other hand, nitro compounds have rarely been used as precursors of nitriles in organic synthesis. Some recent procedures for conversion of nitro compounds into nitriles are presented in Eqs. 6.37–6.40. The oxygen transfer from nitrile oxides by isocyanide gives nitriles; thus, treatment of nitro compounds with t-butylisocyanide, n- butylisocyanate, and triethylamine gives nitriles in 70–80% yield (Eq. 6.37).62 A combined process of dehydration and deoxygenation from nitro compounds is also possible by various reagents such as Me3SiI,63 PI3 in the presence of triethylamine,64 PCl3 in pyridine,65 and (Me2N)3P.66 However, they suffer from low yield in some cases. Recently, a more effective method has been obtained, which is based on the reaction using Sn(SPh)4, Bu3P, and diethyl azodicarboxylate (DEAD), as in Eq. 6.38, in which the reaction is complete in 5–10 min to give nitriles in 85–98% yield. Similar results are obtained, although not so rapid, using only Bu3P (2 equiv) and DEAD (1 equiv), as shown in Eq. 6.39.67 Deoxygenation using disilane is also effective for the conversion of nitro compounds into nitriles (Eq. 6.40).68
|
|
N |
|
|
n-BuNCO, t-BuNC |
|
|
N |
|
CN |
(6.37) |
|
Ph |
NO2 |
|
|
Et3N |
|
|
|
|
||
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
Ph |
|
|
|
|
|
|
|
|
|
|
|
|
74% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NO2 |
|
|
|
|
|
|
|
CN |
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
Sn(SPh)4, Bu3P, DEAD |
|
O |
|
|
|
|||
O |
|
CO2t-Bu |
|
|
0º C, 5 min |
|
O |
|
CO2t-Bu |
||
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
95% |
|
(6.38) |
|
|
|
|
|
|
|
|
|
|
|
|
|
O |
NO2 |
|
|
Bu3P, DEAD |
|
O |
|
CN |
|
|
|
|
|
|
|
|
|
|
(6.39) |
|||
|
O |
|
|
|
0º C, 30 min |
|
O |
|
|
||
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
||
92%

170 CONVERSION OF NITRO COMPOUNDS INTO OTHER COMPOUNDS
|
1) |
SLi |
|
Ph |
|
|
Ph |
NO2 |
|
|
|
S |
(6.40) |
|
2) Me3SiSiMe3 |
3) hν |
NC |
|||
|
|
|
||||
|
|
|
|
|
82% |
|
Radical fragmentation process using tin radical is applied to the conversion of nitro compounds into nitriles, as shown in Eq. 6.41.69
|
|
|
OH |
|
O |
|
|
NaNO2 |
|
C Cl |
|
R |
NO2 |
N |
|
|
|
NaOH |
|
|
|
||
|
|
R |
NO2 |
|
|
|
|
O |
|
|
|
|
|
O |
Bu3SnH |
|
|
|
N |
AIBN |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
R |
|
NO2 |
|
|
R C N |
|
OSn |
|
|
|
|
|
C |
Ar |
N |
-NO2• |
90–100% |
|
O |
|
|
||
|
N |
R |
NO2 |
|
|
|
|
|
|
||
R |
NO2 |
|
|
|
(6.41) |
Indirect conversion of nitro compounds into nitriles via dehydration of oximes (see Section 6.3.3) is also a useful method for this purpose.
6.3 REDUCTION OF NITRO COMPOUNDS INTO AMINES
Reduction of aromatic and aliphatic nitro compounds gives various nitrogen compounds, such as amines, imines, and oximes, where the N–O bonds are cleaved, which is one of the basic reactions of nitro compounds. The sequence of nitration and reduction is the most important method for the preparation of aromatic amines. In aliphatic cases, the recent development of the stereoselective nitro-aldol and Michael reaction using aliphatic nitro compounds makes this conversion important as a tool for the stereoselective synthesis of biologically active amino compounds. Although the cleavage of N–O bond is general in the reduction of both aromatic and aliphatic nitro compounds, the C–N bond cleavage is possible in aliphatic nitro compounds (Scheme 6.4). Kornblum and co-workers reported that some kinds of anion radicals derived from aliphatic nitro compounds cleave the carbon-nitrogen bond to give the carbon radicals. In line with this observation, Ono and Tanner have found that aliphatic nitro compounds are reduced to the corresponding hydrocarbons on treatment with tin hydride in the presence of radical initiators. The conversion of R–NO2 to R-H is now widely recognized as a useful tool for organic synthesis (see Section 7.2).
6.3.1 Ar-NH2 From Ar-NO2
Particularly in the aromatic series, many amines have been prepared by the reduction of corresponding nitro compounds. A large number of reducing agents have been used for the reduction of nitro groups.70a–d The catalytic hydrogenation of aromatic nitro compounds to amines has long been recognized as one of the simplest procedures.71
