
Color Atlas of Physiology 2003 thieme
.pdf

|
Potassium Balance |
extracellular K+ concentration rises (especially |
||
|
in non-respiratory acidosis, i.e., by 0.6 mmol/L |
|||
|
|
|||
|
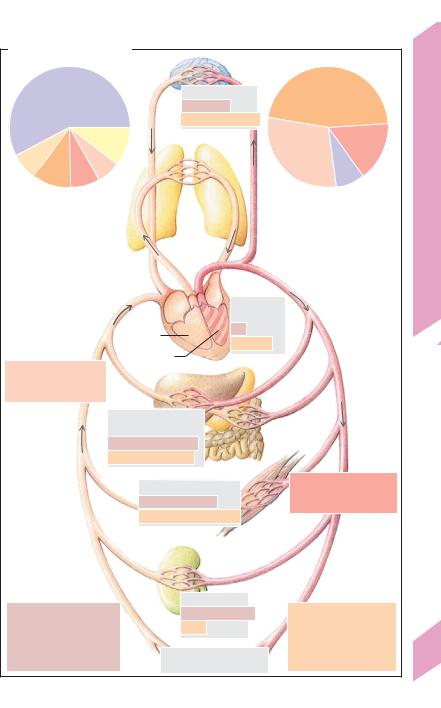
The dietary intake of K+ is about 100 mmol/day |
per 0.1 unit change in pH). Alkalosis results in |
||
|
(minimum requirement: 25 mmol/day). About |
hypokalemia. |
|
|
|
90% of intake is excreted in the urine, and 10% |
Chronic regulation of K+ homeostasis is |
||
|
is excreted in the feces. The plasma K+ conc. |
mainly achieved by the kidney (!B). K+ is sub- |
||
|
normally ranges from 3.5 to 4.8 mmol/L, while |
ject to free glomerular filtration, and most of |
||
|
intracellular K+ conc. can be more than 30 |
the filtered K+ is normally reabsorbed (net re- |
||
Balance |
times as high (due to the activity of Na+-K+- |
absorption). The excreted amount can, in some |
||
in the cells. Although the extracellular K+ conc. |
reabsorbed before reaching the end of the |
|||
|
ATPase; !A). Therefore, about 98% of the ca. |
cases, exceed the filtered amount (net |
secre- |
|
Water |
3000 mmol of K+ ions in the body are present |
tion, see below). About 65% of the filtered K+ is |
||
comprises only about 2% of total body K+, it is |
proximal tubule, regardless of the K+ supply. |
|||
|
||||
|
still very important because (a) it is needed for |
This is comparable to the percentage of Na+ |
||
and |
regulation of K+ homeostasis and (b) relatively |
and H2O reabsorbed (!B1 and p. 157, column |
||
small changes in cellular K+ (influx or efflux) |
2). This type of K+ transport is mainly para- |
|||
Salt, |
can lead to tremendous changes in the plasma |
cellular and therefore passive. Solvent drag |
||
K+ conc. (with an associated risk of cardiac |
(!p. 24) and the lumen-positive |
trans- |
||
Kidneys, |
arrhythmias). Regulation of K+ homeostasis |
epithelial potential, LPTP (!B1 and p. 162), in |
||
and adjustment of K+ excretion according to K+ |
loop of Henle, another 15% of the filtered K+ is |
|||
|
therefore implies distribution of K+ through in- |
the mid and late proximal segments of the |
||
|
tracellular and extracellular compartments |
tubule provide the driving forces for it. In the |
||
7 |
intake. |
reabsorbed by transand paracellular routes |
||
|
||||
|
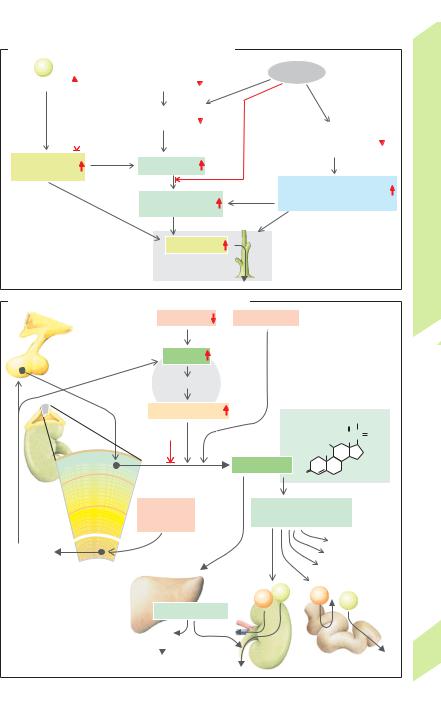
Acute regulation of the extracellular K+ |
(!B2). The amount of K+ excreted is deter- |
||
|
conc. is achieved by internal shifting of K+ be- |
mined in the connecting tubule and collecting |
||
|
tween the extracellular fluid and intracellular |
duct. Larger or smaller quantities of K+ are then |
||
|
||||
|
fluid (!A). This relatively rapid process pre- |
either reabsorbed or secreted according to |
||
|
vents or mitigates dangerous rises in extra- |
need. In extreme cases, the fractional excretion |
||
|
cellular K+ (hyperkalemia) in cases where large |
of K+ (FEK) can rise to more than 100% in re- |
||
|
quantities of K+ are present due to high dietary |
sponse to a high K+ intake, or drop to about |
||
|
intake or internal K+ liberation (e.g., in sudden |
3–5% when there is a K+ deficit (!B). |
|
|
|
hemolysis). The associated K+ shifting is |
Cellular mechanisms of renal K+ transport. |
||
|
mainly subject to hormonal control. The insulin |
The connecting tubule and collecting duct con- |
||
|
secreted after a meal stimulates Na+-K+- |
tain principal cells (!B3) that reabsorb Na+ |
||
|
ATPase and distributes the K+ supplied in the |
and secrete K+. Accumulated intracellular K+ |
||
|
animal and vegetable cells of the food to the |
can exit the cell through K+ channels on either |
||
|
cells of the body. This is also the case in diet-in- |
side of the cell. The electrochemical K+ gradient |
||
|
dependent hyperkalemia, which stimulates |
across the membrane in question is decisive for |
||
|
insulin secretion per se. Epinephrine likewise |
the efflux of K+. The luminal membrane of |
||
|
increases cellular K+ uptake, which is particu- |
principal cells also contains Na+ channels |
||
|
larly important in muscle work and trauma— |
through which Na+ enters the cell (!p. 162). |
||
|
two situations that lead to a rise in plasma K+. |
This depolarizes the luminal membrane, |
||
|
In both cases, the increased epinephrine levels |
which reaches a potential of about –20 mV, |
||
|
allow the re-uptake of K+ in this and other cells. |
while the basolateral membrane maintains its |
||
|
Aldosterone also increases the intracellular K + |
normal potential of ca. –70 mV (!B3). The |
||
|
conc. (see below). |
driving force for K+ efflux (Em – EK, !p. 32) is |
||
|
Changes in pH affect the intraand extra- |
therefore higher on the luminal side than on |
||
|
cellular distribution of K+ (!A). This is mainly |
the opposite side. Hence, K+ preferentially exits |
||
|
because the ubiquitous Na+/H+ antiporter |
the cell toward the lumen (secretion). This is |
||
180 |
works faster in alkalosis and more slowly in |
mainly why K+ secretion is coupled with Na+ |
||
acidosis (!A). In acidosis, Na+ influx therefore |
reabsorption, i.e., the more Na+ reabsorbed by |
|||
|
decreases, Na+-K+-ATPase slows down, and the |
the principle cell, the more K+ secreted. |
! |
|
|
|
|
||
Despopoulos, Color Atlas of Physiology © 2003 Thieme
All rights reserved. Usage subject to terms and conditions of license.









 Mg
Mg 3–8%
3–8%  0.5–3%
0.5–3%