
Gale Encyclopedia of Genetic Disorder / Gale Encyclopedia of Genetic Disorders, Two Volume Set - Volume 2 - M-Z - I
.pdf
Wolman disease
information has shown that many of these individuals may achieve more development than was previously believed possible.
The following management recommendations have been made by Drs. Battaglia and Carey:
•Feeding problems should be addressed and may require intervention such as placement of a gastrostomy tube.
•Characterization of seizures is important and treatment with antiepileptic medications such as valproic acid should be investigated and may help control the seizure activity in many individuals.
•Skeletal abnormalities such as clubfoot should be addressed and treatment should be considered. It should not be assumed that a clubfoot does not need to be addressed because the child will never walk. Children with WHS have learned to walk unassisted.
•As approximately 30% of individuals may have congenital heart defects, the heart should be examined. Usually, the heart lesions are not severe and may be repaired easily or may not even require surgery.
•Hearing loss may occur and because some children are able to learn to talk in short sentences, they should be screened for hearing problems.
•Eye abnormalities may be present and thus an ophthalmology exam should be performed to rule out any eye problems, even if no obvious signs are present.
•In regards to the development of patients with WHS, it is suggested that individuals participate in personal development programs to assist with social skills and occupational therapy for motor skills.
Prognosis
Infants with WHS may be stillborn or die in the newborn period and prognosis during the newborn period depends upon what birth defects are present. It has been estimated that approximately 35% of individuals who have WHS die within the first two years of life. Many individuals with WHS survive to adulthood. Universally, children with WHS have severe or profound developmental retardation, however, there are many affected individuals who are able to walk and some that are able to talk in short sentences. It is evident that many patients seem to proceed farther than was previously thought possible. The actual lifespan for individuals who have WHS is unknown, although there are several individuals who have WHS who are in their 20s–40s.
Resources
BOOKS
Schaefer, G. Bradley, et al. Wolf-Hirschhorn Syndrome (4p-): A Handbook for Families. Munroe-Meyer Media Center, 1996. (1-800-656-3937).
PERIODICALS
Battaglia, Agatino, and John C. Carey. “Health Supervision and Anticipatory Guidance of Individuals With WolfHirschhorn Syndrome.” American Journal of Medical Genetics (Semin. Med. Genet.) 89 (1999): 111–115.
ORGANIZATIONS
National Organization for Rare Disorders (NORD). PO Box 8923, New Fairfield, CT 06812-8923. (203) 746-6518 or (800) 999-6673. Fax: (203) 746-6481. http://www
.rarediseases.org .
WEBSITES
4pSupport Group. http://www.4p-supportgroup.org .
Renee A. Laux, MS
I Wolman disease
Definition
Wolman disease is a rare inherited defect in the body’s metabolism of fats (lipids).
Description
Wolman disease, also known as lysosomal acid lipase disease, is a lethal genetic disorder caused by the lack of the enzyme lysosomal acid lipase. Lysosomal acid lipase is a cellular enzyme widespread throughout the body. It is important in the breakdown of certain body lipids called triglycerides and cholesteryl esters. Individuals without active enzyme accumulate abnormally large amounts of these lipids in their cells. This build-up interferes with the normal metabolic functions of the cells and leads to severe neurological and physical symptoms and early death. A milder disease, cholesteryl ester storage disease (CESD), is caused by mutations in the same gene, but affected individuals may not show symptoms until adulthood.
Genetic profile
Inheritance pattern
Wolman disease is an autosomal recessive disorder affecting both males and females. In individuals with this disorder, both copies of the gene that codes for lysosomal
1208 |
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |

KEY TERMS
Adrenal gland—A triangle-shaped endocrine gland, located above each kidney, that synthesizes aldosterone, cortisol, and testosterone from cholesterol. The adrenal glands are responsible for salt and water levels in the body, as well as for protein, fat, and carbohydrate metabolism.
Amniocentesis—A procedure performed at 16–18 weeks of pregnancy in which a needle is inserted through a woman’s abdomen into her uterus to draw out a small sample of the amniotic fluid from around the baby. Either the fluid itself or cells from the fluid can be used for a variety of tests to obtain information about genetic disorders and other medical conditions in the fetus.
Autosomal recessive—A pattern of genetic inheritance where two abnormal genes are needed to display the trait or disease.
Carrier—A person who possesses a gene for an abnormal trait without showing signs of the disorder. The person may pass the abnormal gene on to offspring.
Chorionic villus sampling (CVS)—A procedure used for prenatal diagnosis at 10–12 weeks gestation.
Under ultrasound guidance a needle is inserted either through the mother’s vagina or abdominal wall and a sample of cells is collected from around the early embryo. These cells are then tested for chromosome abnormalities or other genetic diseases.
Enzyme—A protein that catalyzes a biochemical reaction or change without changing its own structure or function.
Gene—A building block of inheritance, which contains the instructions for the production of a particular protein, and is made up of a molecular sequence found on a section of DNA. Each gene is found on a precise location on a chromosome.
Heterozygote—Having two different versions of the same gene.
Lysosomal—Pertaining to the lysosomes, special parts (organelles) of cells that contain a number of enzymes important in the breakdown of large molecules such as proteins and fats.
Mutation—A permanent change in the genetic material that may alter a trait or characteristic of an individual, or manifest as disease, and can be transmitted to offspring.
disease Wolman
acid lipase are abnormal. Both parents of an affected child have one abnormal copy of the gene, but usually do not show symptoms because they also have one normal copy. The normal copy provides approximately 50% of the usual enzyme activity, a level adequate for the body’s needs. Individuals with one abnormal copy of the gene and 50% enzyme activity are said to be carriers or heterozygotes. Because both parents of a child with Wolman disease are carriers, they have a 25% risk in each subsequent pregnancy of having another child who is affected with the same disorder.
Gene location
The gene for acid lipase is located on the long arm of chromosome 10 at 10q23.2-q23.3. A number of different types of mutations in this gene, all resulting in a lack of enzyme function, have been identified in patients diagnosed with Wolman disease. These include deletions of small portions of the gene, as well as changes in specific nucleotides, the building blocks of the gene. The different mutations may explain why symptoms vary from one individual to another. However, the presence of variabil-
ity in symptoms even among siblings who have inherited the same mutations from their parents, suggests there may be other, as yet unknown, genetic or environmental factors that affect the severity of the disease. Milder forms, such as the related disorder CESD, appear to be associated with gene mutations that result in only partial loss of enzyme function.
Demographics
In the general population, Wolman disease is exceedingly rare, with approximately 50 or fewer welldescribed cases to date (2000). CESD is thought to be more common. Individuals with Wolman disease have been reported in various parts of the world including Western Europe, North America, Iraq, Iran, Israel, China, and Japan.
Signs and symptoms
Symptoms of Wolman disease appear in the first few weeks of life. Forceful vomiting and distention of the abdomen usually alert parents to a problem. Other
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
1209 |
Wolman disease
general symptoms in the early stages of this disease are watery diarrhea or fat in the stools, fever, and a yellow tint to the skin (jaundice). Medical examination reveals massive enlargement of the liver and spleen (hepatosplenomegaly) due to a build-up of fats that cannot be broken down. Other common findings are severe anemia, calcium deposits in the adrenal glands, and a general decline in mental development.
Diagnosis
Diagnosis can be difficult because there are no general laboratory tests that point specifically to Wolman disease. Infants with hepatosplenomegaly and evidence of malnutrition should have a careful neurological examination and x rays of the abdomen to check for calcium deposits in the adrenal glands. If Wolman disease is suspected on the basis of these tests, acid lipase activity can be measured in the laboratory using white blood cells or skin cells. An absence of acid lipase activity confirms the diagnosis.
Carrier testing
Individuals suspected of being a carrier of Wolman disease can be confirmed by measuring acid lipase activity in their white blood cells. Carriers will typically demonstrate 50% of normal enzyme activity.
Mutation detection
Specific DNA tests that check for changes in the normal sequence of nucleotides in the acid lipase gene can usually detect the particular gene mutation in an affected individual or carrier. This type of test is only available in a few, very specialized DNA laboratories.
Prenatal diagnosis
Couples who have had one child with Wolman disease may be offered prenatal testing in future pregnancies. Prenatal testing is accomplished by measuring acid lipase activity either in cells from a chorionic villus sampling (CVS) at about 10–12 weeks of pregnancy or in amniotic fluid cells obtained by amniocentesis between the sixteenth and eighteenth weeks of pregnancy. Alternatively, if specific gene mutations have been identified in parents because they have already had a affected child, fetal DNA from chorionic villus cells
or amniotic fluid cells can be studied to look for these same mutations in the fetus. Carrier couples who are considering prenatal diagnosis should discuss the risks and benefits of this type of testing with a geneticist or genetic counselor.
Treatment and management
There is no specific treatment for Wolman disease. There have been attempts to treat the milder CESD with low-fat diets and cholesterol-lowering drugs, and there has been at least one report of a liver transplant in a patient with CESD. Replacement of the missing enzyme has not been reported.
Prognosis
Infants diagnosed with Wolman disease usually die by six months of age.
Resources
BOOKS
Assmann, G., and U. Seedorf. In The Metabolic & Molecular Bases of Inherited Disease. Edited by C.R. Scriver, et al. New York: McGraw-Hill, 2001.
PERIODICALS
Anderson, R. A., et al. “Lysosomal Acid Lipase Mutations That Determine Phenotype in Wolman and Cholesterol Ester Storage Disease.” Molecular and Genetic Metabolism 68, no. 3 (Nov 1999): 333–45.
Krivit, W., et al. “Wolman Disease Successfully Treated by Bone Marrow Transplantation.” Bone Marrow Transplant 26, no. 5 (Sept 2000): 567–70.
Lohse, P., et al. “Molecular Defects Underlying Wolman Disease Appear To Be More Heterogeneous Than Those Resulting in Cholesteryl Ester Storage Disease.” Journal of Lipid Research 40, no. 2 (Feb 1999): 221–8.
ORGANIZATIONS
National Organization for Rare Disorders (NORD). PO Box 8923, New Fairfield, CT 06812-8923. (203) 746-6518 or (800) 999-6673. Fax: (203) 746-6481. http://www
.rarediseases.org .
WEBSITES
“Wolman Disease.” Online Mendelian Inheritance in Man.
http://www.ncbi.nlm.nih.gov/entrez/dispomim
.cgi?id 278000 .
Sallie Boineau Freeman, PhD
1210 |
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
X
I Xeroderma pigmentosum
Definition
Xeroderma pigmentosum is a rare inherited genetic disease. People with this condition develop skin and eye cancers at young ages because their DNA is extremely susceptible to damage caused by ultraviolet radiation. Xeroderma (dry, scaly skin) and pigmentosum (freckling and abnormal skin coloring) refer to changes that occur after exposure to sunlight or other ultraviolet radiation.
Description
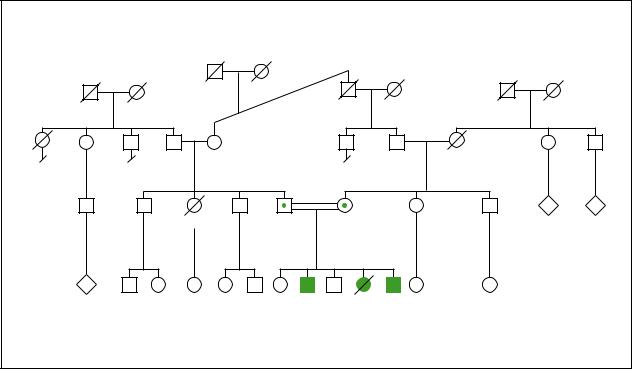
Xeroderma pigmentosum refers to a group of similar conditions. Each subgroup is designated by a letter or a roman numeral. Xeroderma pigmentosum is also often abbreviated XP. XP A and XP I are the same, as are XP B and XP II, XP C and XP III, etc. There are seven types of xeroderma pigmentosum designated A–G or I–VII. An eighth type of XP is called the “variant” type. XP VIII/XP H was once a separate subgroup; now it known to be part of XP D/XP IV.
Each of the eight types of xeroderma pigmentosum has its own DNA defect. However, each section of DNA affected is involved in the same process. These defects affect the body’s ability to repair DNA damage, especially DNA damage to the skin caused by exposure to ultraviolet radiation. Sunlight is the most common source of ultraviolet radiation. Everyone’s DNA is damaged when it is exposed to sunlight. However, the body has complex and very effective methods to repair the DNA damage. This repair mechanism does not work properly in people with xeroderma pigmentosum. They quickly accumulate damage to their DNA if they are exposed to ultraviolet radiation. Cumulative DNA damage leads to cancer, especially of the skin and the eyes.
DeSanctis-Cacchione syndrome refers to the combination of xeroderma pigmentosum along with mental retardation, short stature, and other symptoms.
KEY TERMS
Autosomal—Relating to any chromosome besides the X and Y sex chromosomes. Human cells contain 22 pairs of autosomes and one pair of sex chromosomes.
Cancer cells—Have characteristics that distinguish them from normal cells and non-cancerous cells; they are threatening, harmful, and resistant to treatment
Carcinoma—Any cancer that arises in the epithelium, the tissue that lines the external and internal organs of the body.
Chromosome—A microscopic thread-like structure found within each cell of the body and consists of a complex of proteins and DNA. Humans have 46 chromosomes arranged into 23 pairs. Changes in either the total number of chromosomes or their shape and size (structure) may lead to physical or mental abnormalities.
Deoxyribonucleic acid (DNA)—The genetic material in cells that holds the inherited instructions for growth, development, and cellular functioning.
Malignant—A tumor growth that spreads to another part of the body, usually cancerous.
Trichothiodystrophy (TTD) is sometimes caused by the same DNA change that causes XP D, and rarely XP B. People with TTD also have brittle hair and nails, and physical and mental retardation.
Genetic profile
Xeroderma pigmentosum is inherited as an autosomal recessive condition. Everyone inherits one set of
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
1211 |

Xeroderma pigmentosum
This woman has a severe case of xeroderma pigmentosum. Her right eye is affected, as well as her left cheek. (Custom Medical Stock Photo, Inc.)
genetic material from each parent. People with xeroderma pigmentosum inherited one non-functional XP gene from each parent. Their parents have one normal gene and one abnormal gene (of that particular pair); they are called “carriers.” Carriers do not have the autosomal recessive conditions because the normal gene in the pair protects them. Two carrier parents have a one in four chance with each pregnancy to have an affected child. A person with xeroderma pigmentosum will have an affected child only if the child’s other parent is a carrier or is affected with XP.
The genetics of xeroderma pigmentosum are a bit complicated. The genetic defect in seven of the subgroups has been identified. Each subgroup (A–G) has its own abnormal gene. Each person with xeroderma pigmentosum has a particular subtype, which is associated with one specific abnormal gene. For example, a person with XP type A has no normal XP A gene but does not have XP type B and does not have the abnormal genes associated with XP type B. The genes for types A, B, C, D, E, F, and G are on chromosomes 9, 2, 3, 19, 11, 16, and 13. If two people with different forms of xeroderma pigmentosum had a child, the child would not have xeroderma pigmentosum. But if two people with the same type of xeroderma pigmentosum mated, all of their children would also have xeroderma pigmentosum.
This discussion involves two different types of DNA changes. The first DNA change is the change that the person with xeroderma pigmentosum inherits from both parents. This change (mutation) affects the repair enzymes and is present in every cell in his or her body. The second type of change discussed is additional DNA mutations that result from exposure to ultraviolet radiation. Since the skin and eyes are commonly exposed to ultraviolet radiation that damages DNA and since the body’s repair
system is not working, people with this condition have a high rate of mutation in the exposed organs. These mutations often manifest themselves as cancers—abnormal, uncontrolled growths. Thus, it is the combination of genetic defect and environmental exposure that causes the manifestations of this disease. The first DNA change would not be nearly as problematic if it did not predispose the person with it to accumulate many additional DNA mutations.
Demographics
Xeroderma pigmentosum occurs in every ethnic group. It occurs equally in men and women. Approximately 1 in 250,000 people in the United States have xeroderma pigmentosum. The most common types are A, C, D, and the variant type.
Signs and symptoms
People with xeroderma pigmentosum have photosensitive skin. This means that their skin is hypersensitive to the effects of sunlight. Development of cancer at a young age is the most serious consequence. The eyes are also affected. Some people with xeroderma pigmentosum are affected intellectually, but not all. The symptoms a person will have are somewhat predictable based on which mutation he or she has.
Cutaneous symptoms
Skin manifestations usually begin in infancy. Early effects of skin exposure to minimal ultraviolet radiation include acute sunburn, blistering, freckles, increased or decreased pigment, birthmark-like spots, inflammation, dryness, and rough spots. The face, hands, neck, and arms are more severely affected because of increased sun exposure. Multiple scars may develop. The skin is normal at birth.
The average age at which people with xeroderma pigmentosum develop the first skin cancer is eight years. The risk to develop skin cancer is increased 1,000 times over the risk of the general population. A cell accumulates multiple abnormalities in its transition from a normal cell to a cancer cell. Cancers that occur frequently in people with xeroderma pigmentosum include squamous cell carcinoma, basal cell carcinoma, and malignant melanoma. Basal cell cancers are malignant and, if untreated, are characterized by relentless local invasion, but not metastasis elsewhere in the body. Squamous cell cancers are also malignant and, like basal cell carcinomas, tend to be local, although they are occasionally capable of metastasis. Malignant melanoma, as the name implies, is also malignant, but is much more agressive than either basal
1212 |
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
|
|
Xeroderma Pigmentosum |
|
Xeroderma |
||||
|
|
Autosomal Recessive |
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
pigmentosum |
|
|
|
|
|
|
|
d.62y |
|
|
|
|
|
|
|
|
|
|
d.54y |
|
First cousins |
|
|
Emphysema |
|
||
Uterine |
|
once removed |
|
|
|
|
||
cancer |
|
|
|
|
|
|
N |
N |
2 |
|
|
|
|
|
|
||
HIV |
|
41y |
39y |
|
|
|
||
N |
2 |
4 |
|
|
|
|
3 |
|
|
|
15y |
12y 10y |
d.8y |
6y |
|
||
|
|
|
Mental |
Malignant |
|
Developmental delay |
|
|
|
|
retardation |
melanoma |
Many freckles |
|
|||
|
|
Squamous |
|
|
|
|
|
|
|
|
cell carcinoma |
|
|
|
|
||
(Gale Group)
cell or squamous cell cancers of the skin. It is especially threatening because if not diagosed and treated early, it commonly will spread to internal organs and can be fatal. Cancer may occur on the eyes, lips, and tongue.
Ocular symptoms
Most people with xeroderma pigmentosum also have extremely light sensitive eyes. Their eyes easily become irritated, red, and swollen. Abnormal growths may appear. Cataracts may occur at an unusually young age.
Other symptoms
The other symptoms associated with xeroderma pigmentosum are variable. Many people who are affected have only eye and skin manifestations. Mental deterioration may occur; when it does, it usually worsens over time. Neurological symptoms are not believed to be associated with sun exposure. Some people have one or a combination of: deafness, poor reflexes, lower intelligence, or spasticity (in addition to ocular and cutaneous symptoms).
Diagnosis
Xeroderma pigmentosum may be suspected based on a person’s history of skin changes that occurred after
minimal exposure to sunlight. The diagnosis is confirmed by a blood test or a skin test. The skin or blood cells are sent to a specialty laboratory. Studies are performed to determine whether the cells are hypersensitive to ultraviolet radiation. Scientists may examine whether abnormal changes can be seen in the chromosomes. The type of xeroderma pigmentosum may be determined by genetic studies or other specialized studies.
Genetic testing for xeroderma pigmentosum is complicated because there are eight different genes involved. Genetic tests are usually very specific, looking for a change in one gene. To confirm a diagnosis of xeroderma pigmentosum by DNA testing, scientists must look for multiple changes in eight different genes.
Prenatal diagnosis may be possible, especially if genetic studies have already been performed on an affected sibling and the parents.
Treatment and management
The only treatment for xeroderma pigmentosum is avoiding harmful exposure to ultraviolet radiation and treating/removing growths as they occur. The DNA damage caused by exposure to ultraviolet light accumulates over time and the resulting DNA damage is irreversible.
Life is changed dramatically when a family member has xeroderma pigmentosum. Extreme measures must be
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
1213 |

X-linked hydrocephaly
taken to completely avoid exposure to the sun. Preventative measures include: sunglasses, tightly woven long-sleeved clothing, wide brim hats, sunblock, and protective window coverings (at home, in the car, and at school). Children do not play outside during the day. All sources of ultraviolet radiation are avoided, even exposure to certain light bulbs. These precautions are critical to survival. Levels of ultraviolet radiation at home and at school can be measured with special instruments. Abnormal skin growths and other symptoms are treated/removed as they arise. Regular visits are made to the eye doctor, dermatologist, and neurologist. Often psychosocial support is also helpful.
Treatments that would deliver DNA repair proteins into the skin of affected patients are under investigation. Some people with xeroderma pigmentosum may be offered other types of medication, like isotretinoin. The dermatologist weighs the benefit of prescribing a medication against the side effects associated with that medication.
Because our bodies have different mechanisms for fixing different forms of DNA damage, people with xeroderma pigmentosum are most susceptible to DNA damage by ultraviolet radiation. Some other exposures have been associated with the type of DNA damage caused by ultraviolet radiation. Therefore, people with xeroderma pigmentosum should also avoid exposure to tobacco and certain other drugs.
Prognosis
Life expectancy is significantly reduced due to morbidity associated with the cancers. Researchers have not determined how effective preventative measures are, e.g. avoidance of ultraviolet radiation.
In the 1990s scientists discovered a great deal about DNA repair mechanisms, and about the genes associated with xeroderma pigmentosum. The range of symptoms associated with each type have been better defined. The media has also been interested in XP, giving it more attention than is typical for such a rare condition. Advocates in the XP community have developed helpful resources for other affected families, such as Camp Sundown. Hopefully the first decade of 2000 will also lead to new insights, services, and possibly new treatments.
Resources
PERIODICALS
Cleaver, James. “Stopping DNA Replication in its Tracks.” Science (9 July 1999): 212.
McPhee, A.T. “Trapped in Darkness.” Current Science (19 September 1997): 10.
Miller, Samantha, and Joseph Tirella. “Into the Night.” People Magazine (24 August 1998): 90.
Williams, Monte. “Little Prisoner of the Light.” New York Times (14 May 1997): B1.
ORGANIZATIONS
National Arthritis and Musculoskeletal and Skin Diseases Information Clearinghouse. One AMS Circle, Bethesda, MD 20892-3675. (301) 495-4484.
National Cancer Institute. Office of Communications, 31 Center Dr. MSC 2580, Bldg. 1 Room 10A16, Bethesda, MD 20892-2580. (800) 422-6237. http://www.nci.nih
.gov .
Skin Cancer Foundation. 245 Fifth Ave., Suite 1403, New York, NY 10016. (800) 754-6490. info@skincancer.org.
Task Force on Xeroderma Pigmentosum, American Academy of Dermatology. Box 4014, Schaumburg, IL 60168-4014. (708) 330-0230.
Xeroderma Pigmentosum Registry. New Jersey Medical School, Dept. of Pathology, 185 South Orange Ave., Room C-520, Newark, NJ 07103-2714. (201) 982-4405.
Xeroderma Pigmentosum Society, Inc. PO Box 4759, Poughkeepsie, NY 12602. (518) 851-2612. xps@xps.org.http://www.xps.org .
WEBSITES
Horenstein, Marcelo G., and A. Hafeez Diwan. “Xeroderma Pigmentosum.” eMedicine Journal 2 (February 5, 2001).www.emedicine.com/derm/topic462.htm .
“Understanding Xeroderma Pigmentosum.” National Institutes of Health Patient Information Publications. http://www
.cc.nih.gov/ccc/patient_education/pepubs/xeroderma
.pdf .
OTHER
Xeroderma Pigmentosum and Cockayne’s Syndrome. Video-
tape. American Registry of Pathology and Armed Forces
Institute of Pathology, 1996. 888-838-1297.
Michelle Queneau Bosworth, MS, CGC
I X-linked hydrocephaly
Definition
Hydrocephaly refers to the accumulation of cerebrospinal fluid (CSF) in the fluid-filled cavities, called ventricles, that are located deep in the core of the brain. The designation X-linked indicates that this form of hydrocephaly results from a mutation in a gene that is located on the X chromosome, in this case the L1 cell adhesion molecule (L1CAM) gene.
Description
Cell adhesion molecules (CAMs) provide the traffic signals that guide the cells of developing organs to
1214 |
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |

KEY TERMS
Adducted thumbs—Thumbs clasped across the palm.
Aphasia—Loss of previously acquired ability to speak, or to understand written or spoken language.
Brain ventricles—A set of four connected cavities that are located deep in the core of the brain. Cerebrospinal fluid is made by cells lining the walls of the first two ventricles, then flows through the third, then fourth ventricle before flowing out of the brain. The fluid-filled cavities provide mechanical cushion for the brain, and the CSF provides nutrients to, and carries metabolic wastes away from, the cells of the brain.
Cell adhesion molecule—Any one of several thousand proteins that together control the cell-to-cell communication that must take place in order for cells to migrate to their proper places, develop into the proper types of cells, and make the appropriate connections with other cells.
Corpus callosum—A thick bundle of nerve fibers deep in the center of the forebrain that provides communications between the right and left cerebral hemispheres.
Corticospinal tract—A bundle of long nerve fibers that runs from the motor control region of the cere-
bral cortex to the spinal cord, where it connects to nerves that control movement in the legs.
Cydrocephaly—Excessive accumulation of cerebrospinal fluid in the brain ventricles.
Macrocephaly—A head that is larger than normal.
Penetrance—The degree to which individuals possessing a particular genetic mutation express the trait that this mutation causes. One hundred percent penetrance is expected to be observed in truly dominant traits.
Spastic paraplegia—Inability to walk, due to lack of proper neural control over the leg muscles.
Stenosis—The constricting or narrowing of an opening or passageway.
Ventriculoperitoneal shunt—A tube equipped with a low pressure valve, one end of which is inserted into the lateral ventricles, the other end of which is routed into the peritoneum, or abdominal cavity.
X-linked—Located on the X chromosome, one of the sex chromosomes. X-linked genes follow a characteristic pattern of inheritance from one generation to the next.
migrate to their proper places and make the appropriate connections with the cells with which they interact. The L1CAM protein is embedded in the membrane of nerve cell axons. Axons are projections from the nerve cell body that carry impulses to sometimes distant targets. As a developing axon grows toward its target, its leading end is capped by a growth cone similar in function to that of a plant root. The growth cone of a developing axon is rich in L1CAM. The L1CAM protein has a large, complex extracellular domain (portion of the protein outside the nerve cell), a transmembrane domain within the nerve cell membrane, and a small intracellular domain (portion inside the nerve cell). The extracellular domain acts as a feeler, and binds to CAMs that are either on the surface of other cells or floating in the extracellular fluid. The binding of the L1CAM protein to various CAMs in its environment sends signals into the nerve cell that direct the projecting axon to grow to the appropriate length, follow the course required for it to reach its target, and stop when appropriate.
The L1CAM protein is critical for proper development of several long fiber tracts in the forebrain. These include the corpus callosum, which is a thick fiber bridge that connects the left and right cerebral hemispheres, and the corticospinal tract, which extends from the motor control region of the cerebral cortex down to the spinal cord.
The developing axon often extends many cell diameters away from the cell body, and must interact with CAMs from many different sources to insure that it follows the correct route to its target. The complex extracellular domain of the L1CAM protein interacts with a variety of CAMs in the environment to serve several important functions. Because of the many functions L1CAM serves, the specific brain changes and functional handicaps that are seen in any individual patient with an L1CAM mutation depend on which of L1CAM’s functions are lost and which are spared by the specific mutation that occurs in the L1CAM gene. Most families have their own unique L1CAM mutation. Some mutations abolish all of L1CAM’s functions, while others change only a small
hydrocephaly linked-X
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
1215 |
X-linkedhydrocephaly |
X-linked Hydrocephaly |
|
(Gale Group)
piece of the L1CAM protein. Therefore, there is marked variability between patients in terms of which brain structures are most affected and what the primary physical and behavioral symptoms will be. Because of this variability, patients with different L1CAM mutations have been given diagnoses such as X-linked hydrocephaly (XLH), X- linked spastic paraplegia type 1 (SPG1), hydrocephaly with stenosis of the aqueduct of Sylvius (HSAS), X-linked agenesis of the corpus callosum (XLACC), and MASA syndrome (mental retardation, aphasia, shuffling gait, adducted thumbs). Because these patients all presented with such different combinations of brain changes and functional handicaps, it was originally thought that these were all distinct disorders, with different biological causes. As of 2001, an effort is being made to unite these disorders under the general heading L1CAM spectrum, to reflect the fact that these are not distinct disorders, but merely alternative possible consequences of L1CAM mutations.
Genetic profile
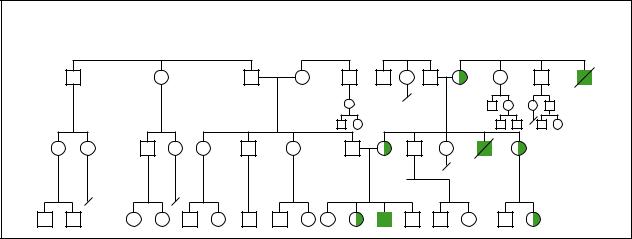
The L1CAM gene is located close to the end of the long arm of the X chromosome, in the band referred to as Xq28. Since the L1CAM gene is on the X chromosome, usually only males are affected. This is because females have two X chromosomes, while males have only one. In a female, if one X chromosome has an L1CAM mutation on it, the non-mutated L1CAM gene on the other X chromosome can usually provide enough good L1CAM protein to support normal brain development. Males, on the other hand, having only one X chromosome, cannot compensate for an X-linked gene mutation.
The inheritance pattern for L1CAM spectrum disorders follows the typical X-linked inheritance pattern. Males are usually the only ones affected, and most
females who carry L1CAM mutations are unaffected. There may be several affected brothers in a single family. In addition, in a family where the L1CAM mutation has been passed through several generations, the normally developed mothers of affected males may have affected brothers, or the normally developed sisters of affected males may have affected sons. If a female carries a mutation in L1CAM, she has a 50% chance of passing the mutation to each of her children. Therefore, approximately half her sons will be affected, and approximately half her daughters will be carriers. There is no known case in which an affected male has reproduced.
L1CAM mutations exhibit 100% penetrance, meaning that any male who has the L1CAM mutation will be affected, albeit with varying degrees of severity. This contrasts with some other disorders, in which some family members are unaffected, despite having the same gene mutation that has been seen in other affected family members.
Demographics
The incidence of hydrocephaly from all causes is approximately 1 in 2,000 live births in the general population. The X-linked form is thought to account for approximately 5% of the total cases of hydrocephaly, or approximately 1 in 25,000 to 50,000 males. In very rare cases a female may be affected, usually mildly. There are no systematic data comparing the incidence of L1CAM spectrum disorders in different races.
Signs and symptoms
Most patients with mutations in L1CAM exhibit mental retardation (MR), the degree of which can vary
1216 |
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
from mild to severe. The vast majority also exhibit hydrocephaly, which can be mild and not require any medical intervention, or severe enough to be life-threatening. The most severe cases of hydrocephaly are associated with stenosis (narrowing or pinching closed) of the aqueduct of Sylvius. The aqueduct of Sylvius (also called the cerebral aqueduct) is a narrow channel connecting the third ventricle, located deep in the midbrain, to the fourth ventricle, located underneath the cerebellum in the posterior part of the brain. The brain’s cerebrospinal fluid (CSF) is made by cells lining the first two ventricles, called the lateral ventricles, which are located in the forebrain. The CSF normally flows from the first two ventricles, through the third ventricle, then into the fourth ventricle, before flowing out of the brain. Stenosis of the aqueduct of Sylvius stops the outflow of CSF, and causes an accumulation of fluid, and pressure, primarily in the first two ventricles. Since there is no mechanism to stop CSF production in the lateral ventricles, this form of hydrocephaly is progressive. The pressure can become so great that it stretches the developing skull bones, which are still not fully hardened, resulting in the child having a head that is visibly enlarged (macrocephaly). In the process, the brain tissue is pressed against the skull, with predictably devastating effects on brain function. Many of the more severely hydrocephalic patients are either stillborn or die within one year of birth.
Approximately 80% of patients with L1CAM mutations exhibit adducted thumbs (clasped across the palm). A smaller percentage exhibit aphasia (lack of speech), or problems with leg control that range from walking with a shuffling gait to spastic paraplegia that leaves them unable to walk at all.
The most common finding in brain imaging studies is the absence (agenesis) or underdevelopment (hypoplasia) of the corpus callosum. The corpus callosum is a large fiber tract that projects between the left and right hemispheres of the brain and enables information to be transferred from one hemisphere to the other. It is uncertain whether the abnormalities in the corpus callosum are an important cause of these patients’ MR. It is most likely that the pressure exerted on the developing brain tissue by the hydrocephaly is a more consistent and important cause of the MR seen in these patients. Another brain structure seen to be underdeveloped in some patients with L1CAM mutations is the corticospinal tract. The corticospinal tract begins in the motor control region of the cerebral cortex and runs downward to connect with the spinal cord neurons that control the legs. Abnormal development of the corticospinal tract is probably the cause of the shuffling gait/spastic paraplegia seen in some patients with L1CAM mutations.
Diagnosis
In the more severely hydrocephalic patients, hydrocephaly can be seen by ultrasound at 20 weeks gestation, or approximately half-way through the fetal period. For less severely affected patients, some degree of hydrocephaly is usually noted within a year after birth, along with a general developmental delay. These babies do not roll over, sit up, or reach for objects as early as babies typically do. In rarer cases, some mildly affected patients are not diagnosed until an age at which speech problems or problems with their walking gait can be observed. Adducted thumbs, when present, are noticeable from birth or are sometimes even visible upon ultrasound analysis.
Genetic testing involves a search for mutations in the L1CAM gene in patients with L1CAM spectrum disorders. The sequence of the L1CAM gene from the affected patient is compared to the normal L1CAM sequence. DNA is usually obtained from a blood sample for postnatal diagnosis. For prenatal diagnosis, DNA can be extracted from amniotic fluid cells obtained by amniocentesis, or from chorionic villus sampling.
Treatment and management
In the most severely hydrocephalic cases, the baby must be delivered by Cesarian section, because the head has grown too large by the end of the pregnancy for the baby to be delivered through the vagina. For the more severely affected patients, a ventriculoperitoneal shunt can be used to reduce the pressure inside the brain. The shunt is a tube inserted into the lateral ventricles that allows the CSF to drain into the peritoneum, or abdominal cavity. This provides a means for the CSF to flow out of the brain in cases of HSAS, in which the aqueduct of Sylvius has been closed and the CSF can not flow out of the brain by the usual channel. Shunting markedly reduces the pressure on the brain, and has saved many patients’ lives. However, shunting will not prevent these patients from having MR or other L1CAM spectrum features.
Other methods for managing cases of L1CAM spectrum disorders are focused on the specific features the individual patient exhibits. Special education is almost always necessary, with the specific program designed to accommodate the degree of cognitive disability seen in the individual patient. Physical therapy and mechanical aids such as walkers can be used to help patients with milder degrees of spastic paraplegia. Speech therapy has also benefited some of the less severely aphasic patients. There is generally little improvement when these therapies are applied to more severely affected patients.
hydrocephaly linked-X
GALE ENCYCLOPEDIA OF GENETIC DISORDERS |
1217 |
