
- •Foreword
- •1. General Introduction
- •2. Processes and Techniques for Droplet Generation
- •2.1.0 Atomization of Normal Liquids
- •2.1.1 Pressure Jet Atomization
- •2.1.3 Fan Spray Atomization
- •2.1.4 Two-Fluid Atomization
- •2.1.5 Rotary Atomization
- •2.1.6 Effervescent Atomization
- •2.1.7 Electrostatic Atomization
- •2.1.8 Vibration Atomization
- •2.1.9 Whistle Atomization
- •2.1.10 Vaporization-Condensation Technique
- •2.1.11 Other Atomization Methods
- •2.2.0 Atomization of Melts
- •2.2.1 Gas Atomization
- •2.2.2 Water Atomization
- •2.2.3 Oil Atomization
- •2.2.4 Vacuum Atomization
- •2.2.5 Rotating Electrode Atomization
- •2.2.7 Electron Beam Rotating Disk Atomization
- •2.2.9 Centrifugal Shot Casting Atomization
- •2.2.10 Centrifugal Impact Atomization
- •2.2.11 Spinning Cup Atomization
- •2.2.12 Laser Spin Atomization
- •2.2.14 Vibrating Electrode Atomization
- •2.2.15 Ultrasonic Atomization
- •2.2.16 Steam Atomization
- •2.2.17 Other Atomization Methods
- •3.1.0 Droplet Formation
- •3.1.1 Droplet Formation in Atomization of Normal Liquids
- •3.1.2 Secondary Atomization
- •3.1.3 Droplet Formation in Atomization of Melts
- •3.2.0 Droplet Deformation on a Surface
- •3.2.3 Droplet Deformation and Solidification on a Cold Surface
- •3.2.4 Droplet Deformation and Evaporation on a Hot Surface
- •3.2.5 Interaction, Spreading and Splashing of Multiple Droplets on a Surface
- •3.2.6 Sessile Droplet Deformation on a Surface
- •3.2.7 Spreading and Splashing of Droplets into Shallow and Deep Pools
- •4.1.0 Concept and Definitions of Droplet Size Distribution
- •4.2.0 Correlations for Droplet Sizes of Normal Liquids
- •4.2.1 Pressure Jet Atomization
- •4.2.5 Rotary Atomization
- •4.2.6 Effervescent Atomization
- •4.2.7 Electrostatic Atomization
- •4.2.8 Ultrasonic Atomization
- •4.3.0 Correlations for Droplet Sizes of Melts
- •4.3.1 Gas Atomization
- •4.3.2 Water Atomization
- •4.3.3 Centrifugal Atomization
- •4.3.4 Solidification and Spheroidization
- •4.4.0 Correlations for Droplet Deformation Characteristics on a Surface
- •4.4.1 Viscous Dissipation Domain
- •4.4.2 Surface Tension Domain
- •4.4.3 Solidification Domain
- •4.4.4 Partial Solidification Prior to Impact
- •5.1.0 Energy Requirements and Efficiency
- •5.2.0 Modeling of Droplet Processes of Normal Liquids
- •5.2.1 Theoretical Analyses and Modeling of Liquid Jet and Sheet Breakup
- •5.2.2 Modeling of Droplet Formation, Breakup, Collision and Coalescence in Sprays
- •5.2.3 Theories and Analyses of Spray Structures and Flow Regimes
- •5.2.5 Modeling of Multiphase Flows and Heat and Mass Transfer in Sprays
- •5.3.0 Modeling of Droplet Processes of Melts
- •5.3.4 Modeling of Multiphase Flows and Heat Transfer in Sprays
- •5.4.0 Modeling of Droplet Deformation on a Surface
- •5.4.1 Modeling of Deformation of a Single Droplet on a Flat Surface
- •5.4.2 Modeling of Droplet Deformation and Solidification on a Cold Surface
- •6. Measurement Techniques for Droplet Properties and Intelligent Control of Droplet Processes
- •6.1.0 Measurement Techniques for Droplet Size
- •6.1.1 Mechanical Methods
- •6.1.2 Electrical Methods
- •6.1.3 Optical Methods
- •6.1.4 Other Methods
- •6.2.0 Measurement Techniques for Droplet Velocity
- •6.3.0 Measurement Techniques for Droplet Number Density
- •6.4.0 Measurement Techniques for Droplet Temperature
- •6.5.0 Measurement Techniques for Droplet Deformation on a Surface
- •6.6.0 Intelligent Control of Droplet Processes
- •Index
Droplet Generation 65
2.2.0ATOMIZATION OF MELTS
Atomization of melts has, in principle, some similarity to the atomization of normal liquids. The atomization processes originally developed for normal liquids, such as swirl jet atomization, two-fluid atomization, centrifugal atomization, effervescent atomization, ultrasonic piezoelectric vibratory atomization, and Hartmann-whistle acoustic atomization, have been deployed, modified, and/or further developed for the atomization of melts. However, water atomization used for melts is not a viable technique for normal liquids. Nevertheless, useful information and insights derived from the atomization of normal liquids, such as the fundamental knowledge of design and performance of atomizers, can be applied to the atomization of melts.
Atomization of melts has many applications and advantages for the production of powders and components compared to conventional techniques. Research and applications of atomization of melts have a fairly short history. Very little work had been done on this subject before the twentieth century. The main obstacle in the development of atomization of melts was a lack of suitable materials and techniques for handling molten metals. Over the past decades, atomization of melts has become the dominant mode for powder production due to its attractive features. First, high powder production rates make the technology viable and competent economically. Second, for certain atomization methods, it is possible to control powder size, size distribution, shape and surface morphology. Third, atomization offers a high degree of flexibility in alloying and control over impurity levels. Presently, it is the only viable fabrication technology for highly alloyed specialty alloy powders. The atomized powders are prealloyed and exhibit composition homogeneity. In addition, the production capacity may range from hundreds of grams to tonnage quantities. By the early 1980’s, over 60% by weight of all the powders produced in North America were manufactured by atomization.[4] It was estimated that the worldwide atomization capacity is approaching 106 metric tons per year.
66 Science and Engineering of Droplets
Atomization of melts has also been used in advanced materials processing and synthesis of net or near-net shape components without the formation of powders. Thermal spray, plasma spray deposition,[43] spray atomization deposition,[3] centrifugal spray deposition, and spray co-injection deposition are some examples of this category of applications. In thermal spray processes, a solid material in the form of powder, fiber or wire is directed into a plasma or combustion flame where it is rapidly molten into a spray of droplets and accelerated to a high velocity towards a target substrate to form a surface coating or a free-standing component. The powder feed is usually obtained from atomization of melts. Spray atomization deposition generally refers to a material synthesis approach that involves the energetic disintegration of a molten material by high-speed gas jets into micro-sized droplets, followed by the immediate deposition of solidified, semi-solid, and liquid droplets on a substrate to form a coherent, near fully dense material of desired shape. This process has also been termed spray casting, spray rolling, or spray forming, depending on whether or what post-processing steps follow the deposition. Various shapes, such as disk, tube, strip, and sheet, can be deposited by manipulating the substrate and/or changing the atomizer parameters. Refined microstructure and improved physical/mechanical properties can be achieved as a result of rapid solidification. Therefore, spray atomization deposition has attracted considerable attention as a viable processing alternative for structural materials over the past two decades.
Numerous atomization techniques have evolved for the production of metal/alloy powders or as a step in spray forming processes. Atomization of melts may be achieved by a variety of means such as aerodynamic, hydrodynamic, mechanical, ultrasonic, electrostatic, electromagnetic, or pressure effect, or a combination of some of these effects. Some of the atomization techniques have been extensively developed and applied to commercial productions, including (a) two-fluid atomization using gas, water, or oil (i.e., gas atomization, water atomization, oil atomization), (b) vacuum atomization, and (c) rotating electrode atomization. Two-fluid atomization
Droplet Generation 67
has been the predominant technology and accounts for over 95% of the atomization capacity worldwide.[4] Some other atomization techniques have been evaluated in laboratory and pilot scale operations and may be considered as near-commercial methods, such as (a) ultrasonic gas atomization, (b) rotating disk atomization, (c) electron beam rotating disk atomization, and (d) roller atomization. A number of other atomization techniques do not appear to have established significant commercial applications but are of high scientific interest. These include, for example, (a) centrifugal shot casting atomization, (b) centrifugal impact atomization, (c) spinning cup atomization, (d) laser spin atomization, (e) Durarc® process, (f) vibrating electrode atomization, (g) ultrasonic atomization, (h) steam atomization, (i) electromagnetic and electrostatic atomization, and (j) impaction and pressure atomization. These methods demonstrate engineering innovation and may be used to produce high quality powders of a variety of advanced specialty materials. Some of them may be commercialized in the near future. The development of these techniques has not only advanced atomization technology in general but also improved our fundamental understanding of atomization mechanisms. As the need for advanced powder and particulate materials increases, more elaborate atomization techniques will be required.
Similar to the atomization of normal liquids, the basic requirements for atomization quality are a small droplet size and a narrow size distribution. In the atomization of melts, relatively fine droplets can be obtained using some techniques and, as a result, high cooling rates, ranging from 102 to 106 °C/s, can be achieved which allow the control of powder microstructure to a fine degree. Cooling rates of 105 to 106 °C/s are readily attainable in substrate quenching of fine liquid droplets using rotating discs, high speed rolls, or other metallic substrate systems. However, many atomization techniques are incapable of producing a narrow size distribution, i.e., the yields of useful powders are low, thus the production costs are high. Therefore, the requirement of a narrow size distribution is very important to reducing the costs, particularly of high alloy materials, and to reproducing microstructures of powders.
68 Science and Engineering of Droplets
In addition to the difference in the size distributions of droplets/particles produced, the overall production costs are apparently the point of differentiation among all atomization techniques under consideration. As the criteria for the evaluation of a specific atomization technique/system, the following factors are of importance: high yields, minimum use of expensive gases, clean droplets/ particles, and high throughput.
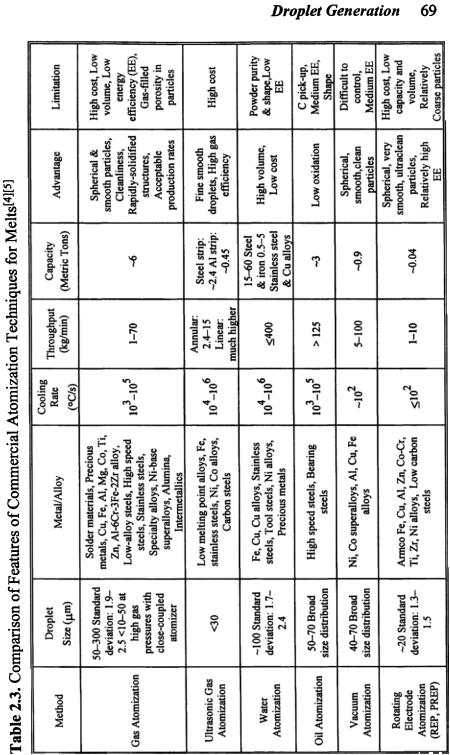
In spite of the differences among the various atomization techniques for metallic melts, there are some features in common. First, the metal to be atomized is generally molten by high-frequency induction or resistance heating, fuel firing, or arc melting in a crucible (tundish). Melting equipment and procedures are similar. For reactive metals, induction melting is conducted in vacuum. With rare exceptions, the liquid metal is delivered through a refractory or ceramic nozzle. Refractory-free, clean melting systems used in casting processes (for example, electroslag refining system) may be employed for metal melting in atomization to reduce ceramic inclusions and defects. In the electroslag refining system (ERS), the liquid metal is transferred directly from the ERS pool to the atomization system via a ceramic-free copper funnel, i.e., a cold-walled induction guide. Second, in contrast to the atomization of normal liquids, the cooling of liquid metals occurs simultaneously during the atomization of melts, leading to an increase in liquid viscosity and surface tension and eventual solidification of atomized droplets. One of the issues to be considered in the atomizer design is therefore the possible blockage of the pouring system due to solidification and/or buildup of the liquid metal at the metal delivery nozzle tip caused by inappropriate flow pattern (typically recirculation flow) and rapid cooling. Cooling and solidification also affect critically the resultant droplet size distribution and determine, to a large extent, the droplet shape. Third, some post-processing and treatment systems for some atomization techniques may be similar. A comparison of the features of various atomization techniques is given in Tables 2.3, 2.4 and 2.5.
Droplet Generotion |
69 |
~
~
~
"Q)
~
I..
tS
~
G)
g.
.fj
G)
~
5
~
s
~
'-ca
~
~
u c,0
~
G)
~
G)
~
c,0
~
0
~
.~
p.
§
u
.
~
N
~
-
~
~
§
.~
.8
~
..
f
>
~
<
";;'
~§
.-E- o
tU |
O |
~ |
.- |
tU |
~ |
C)6
l--a:
.-= .- bOE ='Cb
e~
.~-=
01)
=---
|
"' |
o=u |
o |
o ~ |
|
u |
'- |
;.., 0
~
~
~e
-:1. 0,,-,
o
O.~
"'
~
0
~
~
~~,..::
0 |
|
..t1J |
W"OC |
|
|
|
|
|
~~ |
|
.~ |
|
.9 |
|
~ |
~~ |
|
|
|
.2 |
|||
..JO |
|
;>,C |
.-"' |
|
|
~ |
|
.-0 |
|
|
|
|
|
~ |
|
a |
..2:'.~ |
||||||
...J |
|
|
u |
|
|
|
|
~..J |
|
|
|
|
|
|
|
||||||||
-bJ) |
|
|
|
|
-ou |
|
|
|
|
O |
|
|
g.~ |
.. --"~ |
|
|
J |
|
|
|
~ |
||
|
|
|
|
|
|
|
|
u |
|
o-.."w |
|
.e |
~ |
-oe |
|
|
",OE>ta |
|
.+: Q |
||||
~ |
|
';e |
h |
G |
iE ' |
|
.-u"' |
.- |
|
|
|
|
|
|
|
|
|
||||||
..-c |
|
|
|
|
|
|
|
|
|
~== |
|
B~~ |
|
|
|
||||||||
..: |
|
= |
E |
.~ |
[Q |
e |
~ |
|
|
|
...o-w |
|
.~ .-.r: |
|
I: |
c |
.- |
|
|
~0~.. |
|
||
|
|
~ |
|
-8 = |
|
Q."Q~ |
|
,..0'0 |
|
|
|
|
|||||||||||
bJ)i5 |
|
.g~~c. |
|
|
|
~ |
|
~~ |
|
u .. |
|
.-" |
|
.. |
~~>~~:I: |
|
o |
||||||
|
|
|
|
|
~~ |
|
~ |
|
0 |
|
~ |
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
||||||||||||||
.:I:-> |
|
|
|
Q |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
U |
|
|
."0 |
|
Q) |
|
|
|
.- |
|
~ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
.n |
|
|
|
|
~, |
|
|
.. |
|
c |
|
|
|
|
Q~ |
|
'EiI |
||||
-y- |
|
Q) |
--~ |
.~ |
|
|
|
Q) |
-OS |
DO |
|
|
|
.a |
|
|
|||||||
"Q |
|
"' |
.n |
.~ |
.n |
-Q) |
~ |
..; |
;.., |
e |
t; |
o |
|
|
QJ~ |
|
.- |
||||||
|
.~ |
.n |
"0 |
|
-.. |
|
|
DO(.) |
|
"~ |
|
-.."' |
|
|
;>(6."..C |
|
|
||||||
|
|
|
|
|
|
|
|
|
O |
|
= |
o |
|
~-.. |
|
|
|
|
|||||
~~~;:=~.0= |
|
|
|
|
|
.-C |
|
"' |
|
00- |
|
|
.~.2 |
|
>. |
||||||||
|
|
|
|
|
e |
:I: |
.!! |
"0 |
u |
|
|
|
|
||||||||||
"'=.~0=~o |
.n |
~ |
|
Q,.~ |
|
|
.-.0 |
|
|
-a-,,-~ |
|
|
|
||||||||||
.~ |
|
Q,- |
|
|
|
|
|
|
|
|
|
..-5 |
|
.- |
~ .-QJ |
,., |
|||||||
|
|
|
|
|
|
|
|
|
"' |
" |
(.) |
> |
~ |
|
|
|
" |
|
|||||
|
|
|
|
|
|
|
|
|
"g |
|
|
|
|
.-" |
|
|
i |
;> - |
|||||
t-s~~~8'Q |
|
|
|
|
|
|
f]trj |
|
.cO |
|
|
~o~ |
|
|
..-"' |
|
|
.- |
|||||
|
|
|
|
|
|
|
|
|
|
|
|
QJ... |
|
|
- |
||||||||
.=oQ):aJ:3"'= |
.n |
|
< |
-g |
~U~ |
U |
bO~ |
~ |
|
Q.°c |
E |
|
.c |
|
0 |
Q..! |
|
||||||
~ |
|
o |
O |
.~ |
|
.-"Q, |
|
{/) |
|
|
|
||||||||||||
|
|
e |
|
~ |
|
|
|
.. |
"' |
O |
|
":E |
|
o |
|
|
", |
|
Q.o |
|
|
|
QJ |
|
|
|
|
|
|
|
|
--'1 |
|
|
|
|
|
|
|||||||||
|
|
.n |
|
~ |
|
|
|
Q, |
|
.. |
|
|
|
|
|
|
|
V)e |
|
"' |
|
~ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
"" |
Q. |
|
-.,.,..."' |
> |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Q,"- |
|
..I |
~ |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
.-D |
|
|
~"!"'.9. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
]"",01") |
|
|
|
|
~ |
|
|
|
~ |
|
||||
|
|
|
|
~ |
|
|
|
|
1;;-"1" |
|
"10~'a |
~ |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
...<0 |
|
0.:..= |
|
|
|
Q |
|
|
|
9 |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
~ |
"1". |
1 |
'OeCUI---a |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
{I) |
N |
|
~~~~ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
0
~
"'0
i'
~
." |
E:: |
.-g |
|
|
|
|
|
5°.~.. |
|
... |
.; |
|
|||
.u |
u |
-~ |
,!!) |
la |
|
||
..."a |
|
~ |
..-9 |
.- |
.= |
|
|
..bO |
..bO |
1!. |
e |
~ |
|||
~~N.-"'Z |
= |
"' |
|
||||
.N |
|
|
~ |
.- |
|||
|
, |
---O |
|
|
|
., |
|
-a-::i,~.".~~<~ |
|
|
|
|
|||
...M..~-."... |
..§ "a |
~ |
e |
||||
~ |
~ |
.!. ~ |
|||||
e |
|
.U |
,..,~ |
~=a |
1!. |
||
..;'('90."."a..~ |
|
|
|
||||
..""-' |
|
~,"a..UC. |
|
|
|
||
- |
]"' |
.,B..~ |
|
|
|
||
° |
|
|
~~."c."' |
~ |
|
|
|
~..NO |
|
|
|
|
|
||
|
S |
..J |
|
|
|
|
|
"EI~="O...0\;0~~"Q.~ |
-5"0 |
|
|
||||
~..on |
J |
-.-"'=N0 |
|
.- |
|
||
~ |
|
= |
|
|
|||
rno |
|
|
~~C.)e |
|
|
|
|
° |
|
.--tII) |
|
= |
|
' |
o |
O ~V.-~.-"'~ |
|
'B |
o |
"! |
|||
'i't~ |
|
|
..u |
|
|
||
O"0N |
|
Q. |
|
|
|||
',",
c o
.~ N
.a
~
[3
"
|
~ |
laV\..-; |
.- |
--la |
|
=.J.UO.: |
..50.: |
~ |
<N..Jt)
~
\00
i
"ot"~
.;.,j |
|
|
~;... |
|
|
-0 |
|
|
"'- |
|
|
;..."a |
|
|
o |
|
"' |
-0 |
|
|
"aU- |
|
-.. |
- |
|
|
C |
z |
- |
.-"' |
|
.c |
0 |
|
Q.:1!. O b!)...C
.= :!. la
~"'u
.."' e ~
~]
O \U
---'1~
0
M
V
~c
O |
.g |
u |
~ |
.-cN .- |
|
~ |
6 |
.E~
::J
|
|
o |
|
|
|
|
o |
|
|
|
|
'\'ii |
|
|
'Qo |
|
|
||
|
|
7 |
|
|
..,.~ |
|
|
||
"' |
"'. |
|
|
|
"' ... |
|
|
||
..o |
|
|
|
|
c= |
|
.u |
"' |
|
.~ |
|
|||
~:Zs |
|
|
||
|
" ... |
|
|
|
"' |
"' |
|
|
|
0 |
|
.."' e |
|
|
-.9 |
"' |
= |
0 |
|
'a |
|
|||
= |
|
-.- |
u |
|
|
0 |
|
||
u |
|
o |
~ |
|
= |
"E--~ |
|
||
|
" |
|
|
|
u.!!! |
|
|
|
|
|
" .. |
|
|
|
~...9 |
"' |
|
|
|
"0 I
~~
"0-
~=~
~ O .
~.-N
=
O .- 0 >
-U
1"0
=
0
~ .~
1U .~
~e o
~
on
~
/\
""0 I
~
co
=
.5
"
a1
"'"
-"' ,,-
""
~,g
-0 "'
"
0.
"'
~
~
|
= |
'g |
.g |
0 |
= |
...~ CD .-
o~
1- .-
0:!...' II") ~
.~
=
0
.~
~
N
.e
~
n
o o
j'
...,
NO
7
..
~
"
u
<
.".",
>.>. o o
~:a
..
~
~
o u
z
~
~.g
O =
...00 ~.-
o~
.,.. .-
6~
..r N
.;;
~
e .2
=1U = N
(.) .- = e
;>B
~
|
|
o |
|
|
|
|
|
i |
|
|
|
|
~ |
|
|
|
|
|
|
VI |
|
||
..-= |
o |
|
|
||
u |
|
|
|
|
|
o .0 |
|
|
|||
8 |
|
|
~ |
|
|
c~ |
o |
|
|
||
N |
|
|
|
|
|
|
-..J |
|
|
||
-"' |
|
-- |
|
||
< |
|
|
|
|
|
|
-~" |
|
|
||
8.9~ |
|
|
|
||
" |
-= |
|
|
||
|
.- |
|
|
||
~z |
|
|
|
||
8 |
|
..- |
|
||
~~ |
|
|
|
||
-oM |
I |
|
|
||
|
|
|
|||
to-.; |
|
|
|
||
-0 .. |
|
|
|||
! |
|
|
=." |
|
|
|
|
|
O |
|
. |
|
|
.-- |
|
|
|
ri1~ |
|
|
|
||
0.- |
|
|
|
|
|
't.g |
|
|
|
||
|
|
|
|
=--- |
|
|
|
uo~ |
.~ |
~ |
|
bO"0 |
|||||
.= |
|
0 |
|
~ |
~ |
i |
|
~ |
|
.~ |
~ |
'0 |
|
|
U |
e |
~. |
~!iJg~ < |
~~ |
||||

70 Science and ~--Bineering of Droplets
l:!-n5
~
~
'-UJ
~
Q)
~
~
tS
{/}
Q)
~
0"'
]
(.)
Q) t-;
~
0
~
e o
<
-
cU
~
Q)
~
u I
~
Q)
Z
~
0
{/}
Q)
.a
(\S
Q)
~
~
0
§
{/}
.~
S- o
U
~
N -~
"Q
=
~
§ |
|
.~ |
|
.e |
|
:.:j |
|
~ |
|
'i |
|
;> |
|
-0 |
|
< |
|
cQ |
,- |
Q ~ |
"' |
tUQ)~ |
|
§-~~ |
|
u'-' |
|
~
Q.'C
..c: .-
~~
e~
~'-'
~
bOU
~ 0
.~ '-' '0 U
8~
>-
.9
~
I
~
~---
'6 §.
"Q.,-, 0
...
C)
"8
~
~
~-"'- |
|
,..., |
|
~ |
|
|
|
||
|
O |
.0 |
~ |
. |
|
|
|||
..9;"",6. |
|
-a",-""- |
|
a |
|
"' |
|||
-t' |
.f;' ~ |
~~~o |
|
-~~ |
00 |
(U .- |
|||
'-' Q) C,).-;. Q) .-='C,) |
=' |
||||||||
> |
~ |
= |
"' |
~ |
.+; (U 0 |
Q) |
8 |
~ |
|
.~ |
g. |
9 |
|||||||
~ |
o.~ |
§".s |
.E |
|
p |
||||
-(J~ |
|
|
|
||||||
|
|
|
|
|
|
|
|
||
~ |
|
|
u |
|
~ |
|
|
|
|
~ |
|
-.~ |
u |
a |
~ |
|
-a |
|
|
|
|
|
u |
|
|
0 |
|
:2 |
|
|
~¥J |
|
|||
u-s |
~ |
|
"' |
.~ |
~ |
|
|
|
5 |
|||
|
|
|
|
|
~ |
|||||||
-a-ou~ |
.-o |
|
|
:S=.d 01 |
|
.?;-~ |
|
c |
5 |
|||
|
|
|
|
Q) ... |
||||||||
.~ ~ |
~ |
|
!a |
~ |
:E |
|
~~ |
|
|
~ |
0- |
|
|
|
.~ |
|
|
0 |
=' |
||||||
U |
0. |
Z |
-.-0 |
|
|
|
|
|||||
.d |
|
|
|
|
|
|
|
~[ |
|
|||
0. |
|
|
|
|
|
|
|
~ |
|
|
|
|
rn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0- |
|
|
|
|
I/"\ |
|
|
|
||
|
|
~I |
|
|
|
|
9 |
|
|
|
|
|
|
|
00 |
|
|
|
|
~ |
|
|
'1 |
||
|
|
'j |
|
|
|
|
|
|
||||
|
|
|
|
|
|
"i' |
|
|
||||
|
|
"' |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
>- |
|
|
t--o |
|
|
|
|
|
|
|
|
u |
|
|
|
|
- |
|
|
|
|
|
...'0 |
|
.~ |
~ |
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
I |
|
|
|
|
- |
|
|
~ |
0 |
|
~ |
|
|
|
|
|
|
" |
|
|
-- |
|
|
|
|
|
|
|
|
|
|
|
|
~ |
|
"'..u |
|
O |
|
|
"0 |
|
.-a |
"'~ |
Q |
|
|
|
>. |
~ |
|
|
..- |
|
|
|
|
< |
|
||
|
|
|
|
> |
u |
=' |
|
|
||||
O~"'U |
|
|
|
|
|
I... |
"' |
|
|
|
||
-~.- |
|
.""'- |
|
£ |
!3 "' |
.- |
;:3'", |
|||||
."" |
|
.- |
||||||||||
"' |
.-0 |
|
|
|||||||||
...~-C/) |
.~ |
|
|
. |
|
~u~E-- |
|
Uu |
|
|||
u |
|
|
|
|
~ |
|
|
|||||
P.u-", |
|
|
|
|
'lJ .~ :a |
..g |
|
|||||
"'= - |
|
|
|
|
|
|
|
|
|
|
=00 |
|
|
|
|
u |
|
u |
~g~ |
|
|
00 |
|
||
I |
|
.U |
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Z< |
|
|
|
|
C/) |
|
|
~ a |
|
-5!. |
|
|
|
|
|
|
|
|
|
~ |
|
c |
|
|
|
|
] |
|
.~'::! |
|
o |
|
o |
|
O |
|
||
|
|
|
~ |
u |
.§ |
|
00 |
|
||||
O |
-0 |
|
~ |
|
I |
|
|
|
|
|||
'-0 |
|
|
|
|
|
|
z |
.~ |
.c |
|
|
|
! |
B |
|
> |
to . |
|
|
"' |
.- |
|
~ |
|
|
|
|
|
u- |
|
|
0 |
|
~ |
|
|
||
|
|
|
|
|
|
|
~ |
|
||||
|
|
|
|
|
0 |
|
.- |
|
|
|||
|
00-0 |
|
|
|
|
|
|
|||||
|
|
|
|
r-. |
|
-0 |
|
|
||||
|
|
|
|
|
|
|
v |
|
|
|
|
|
~ |
|
Q |
|
|
|
|
~ |
|
|
|
= |
|
.~ |
|
|
|
|
|
|
|
|
|
|||
O |
|
.5! |
|
|
= |
|
.~ |
§ |
|
|
0 |
|
|
|
|
|
|
|
t3ab/)~O0O |
|
|
~.~ |
|
||
oo1S2' |
|
~ |
G) = |
.-~ |
|
|
||||||
.5 |
|
.-00 |
|
|
|
|||||||
S§e, |
|
|
|
&1 |
~ |
"E |
§ |
ffi |
~ |
.s |
||
|
|
|
|
|
|
|
|
|
~ |
|||
~< |
|
|
|
|
|
|
~<-- |
|
|
|||
|
|
|
|
|
|
|
|
|
||||
""'~
Droplet Generation 71
Table 2.5. Comparison of Features of Other Atomization Methods for Melts[4][5]
|
Droplet |
|
Cooling |
Through- |
Capacity |
|
|
||
Method |
Size |
Metal/Alloy |
Rate |
put |
(Metric |
Advantage |
Limitation |
||
|
(mm) |
|
(°C/s) |
(kg/min) |
Tons) |
|
|
||
Centrifugal |
150–1000 |
Fe, Co, Ni, |
|
|
|
|
Narrow |
Coarse |
|
Ti alloys, |
|
|
|
|
|||||
Shot Casting |
Standard |
2 |
5 |
£1 |
|
size |
|||
Alumina, |
— |
particles, |
|||||||
Atomization |
deviation: |
10 |
-10 |
|
distribution |
||||
Uranium |
|
|
|
|
Low EE |
||||
(CSC) |
1.3–1.4 |
|
|
|
|
|
|||
monocarbide |
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
||
|
|
Si,Stainless |
|
|
|
|
|
|
|
Centrifugal |
|
steels,Ni, |
|
|
|
|
Fine |
|
|
|
Cu, |
|
|
|
0.5–0.9 |
|
|||
|
|
|
|
particles, |
|
||||
Impact |
10–1000 |
Fe81.5B14.5S |
|
|
0.5–0.9 |
Low volume |
|||
|
|
´10-3 |
Rapid |
||||||
Atomization |
|
i3C1, |
|
|
|
|
quenching |
|
|
|
|
Raney® type |
|
|
|
|
|
||
|
|
|
|
|
|
|
|
||
|
|
alloys |
|
|
|
|
|
|
|
|
|
Sn, Pb, Al, |
|
|
|
|
Fine, clean, |
|
|
|
|
|
|
|
|
spherical |
|
||
Spinning |
<10–300 |
Cu, Zn |
|
|
|
|
Relatively |
||
|
|
|
|
particles, |
|||||
alloys, |
|
|
|
|
|||||
Cup |
Standard |
5 |
6 |
|
0.5–1.4´ |
Narrow |
low volume |
||
|
|
||||||||
Atomization |
deviation: |
Stainless and |
10 |
-10 |
— |
10-3 |
size |
& |
|
(RSC) |
1.5–1.7 |
High speed |
|
|
|
|
distribution |
productivity |
|
tool steels, |
|
|
|
|
|||||
|
|
|
|
|
|
Small |
|
||
|
|
Superalloys |
|
|
|
|
|
||
|
|
|
|
|
|
facility |
|
||
|
|
|
|
|
|
|
|
||
Laser |
|
|
|
|
|
|
Spherical, |
Low volume |
|
|
Ni-Al-Mo, |
~105 |
|
|
homogeneo |
& |
|||
Spin |
~100 |
— |
— |
us, |
productivity, |
||||
Ti-6Al-4V |
|||||||||
Atomization |
|
|
|
|
|
clean |
Poor EE, |
||
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
particles |
Cost |
|
|
|
|
|
|
|
|
Spherical, |
|
|
|
|
|
|
|
|
|
clean |
|
|
Durarc® |
³250 |
Ti and Zr |
— |
— |
— |
particles, |
Low volume |
||
Process |
alloys |
Less |
|||||||
|
|
|
|
|
limitation |
|
|||
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
on starting |
|
|
|
|
|
|
|
|
|
materials |
|
|
Vibrating |
|
Mild steel, |
|
|
|
|
Spherical, |
Low volume |
|
Electrode |
300–500 |
Cr-Ni steel, |
|
|
|
|
high-purity |
||
— |
~0.2 |
— |
& |
||||||
Atomization |
|
Cu-Ni alloy, |
particles, |
||||||
|
|
|
|
|
productivity |
||||
(VEP) |
|
W |
|
|
|
|
Simple |
||
|
|
|
|
|
|
||||
72 |
Science and Engineering of Droplets |
|
|
|||||||||
Table 2.5. (Cont’d.) |
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Droplet |
|
Cooling |
Through- |
Capacity |
|
|
|||
|
|
Method |
Metal/Alloy |
Rate |
put |
(Metric |
Advantage |
Limitation |
||||
|
|
Size (μm) |
||||||||||
|
|
|
|
(°C/s) |
(kg/min) |
Tons) |
|
|
||||
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
Solder |
|
|
|
|
|
|
|
|
|
30–50 |
materials, |
|
|
|
Spherical & |
|
||
|
|
|
|
Welding |
|
|
|
|
||||
|
|
|
USWA: |
|
|
|
high quality |
|
||||
|
|
|
electrodes, |
|
|
|
Low volume |
|||||
|
Ultrasonic |
|
>100 |
USWA: |
|
|
particles, |
|||||
|
|
Ag-, Cu-base |
0.02–0.8 |
Low |
& |
|||||||
|
Atomization |
Medium |
~107 |
Low cost, |
||||||||
|
|
|
|
size |
alloys, |
|
|
|
Small unit, |
productivity |
||
|
|
|
|
Dispersion- |
|
|
|
|
||||
|
|
|
distribution |
|
|
|
High EE |
|
||||
|
|
|
hardened |
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
alloys |
|
|
|
|
|
|
|
|
|
|
|
|
Carbon-, low- |
|
|
|
|
|
|
|
|
|
|
|
|
alloy-, |
|
|
|
Between |
|
|
|
Steam |
|
Coarse |
stainless |
~103 |
— |
— |
gas and |
Irregular |
||
|
Atomization |
|
steels, Co-, |
water |
particle shape |
|||||||
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
Ni-base |
|
|
|
atomization |
|
|
|
|
|
|
|
|
superalloys |
|
|
|
|
|
|
|
|
50–500 |
Sn, Pb, Sb, |
|
|
|
Narrow size |
Limited to |
|||
|
|
Pressure |
Standard |
|
|
|
||||||
|
|
Bi, In, Mg, |
— |
~17 |
— |
distribution |
low melting |
|||||
|
Atomization |
deviation: |
||||||||||
|
Al, Zn |
|
|
|
High EE |
point metals |
||||||
|
|
|
|
1.4 |
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
||
|
Vibrating- |
103−104 |
|
|
|
|
Very |
Coarse |
||||
|
Standard |
|
|
|
|
narrow size |
particles,Very |
|||||
|
|
Orifice |
Ca, Zn, Al |
Low |
Very low |
Low |
low volume |
|||||
|
|
deviation: |
distribution, |
|||||||||
|
Atomization |
|
|
|
|
& |
||||||
|
|
<1.05 |
|
|
|
|
High EE |
|||||
|
|
|
|
|
|
|
|
productivity |
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Electro- |
600−850 |
|
|
0.05 |
|
Narrow size |
Low volume |
|||
|
|
Magnetic |
Narrow size |
Sn, Pb |
— |
− |
Low |
|
& |
|||
|
|
0.12 |
distribution |
|||||||||
|
Atomization |
distribution |
|
|
|
productivity |
||||||
|
|
|
|
|
|
|||||||
|
|
|
|
103 |
− |
|
Fe, Steels, |
|
|
|
Simple, |
|
|
Impaction |
3 |
15 |
Cast iron, |
|
|
|
High EE, |
Very coarse |
|||
|
|
|
× |
|
× |
|
Low |
Large |
Large |
|
|
|
|
Atomization |
|
103 |
|
Ferroalloys, |
Large |
particles |
|||||
|
|
|
|
|
|
|
Mattes |
|
|
|
throughput |
|
|
Electrostatic |
|
|
|
|
|
|
|
|
Potentially |
Difficulties in |
|
|
|
— |
|
— |
— |
Very low |
Low |
narrow size |
practical |
|||
|
Atomization |
|
|
|||||||||
|
|
|
|
|
|
|
|
|
distribution |
operation |
||
|
|
|
|
|
|
|
|
|
|
|
||
This section describes the atomization processes and techniques for metal droplet generation. Advantages and drawbacks of the atomization systems are discussed along with typical ranges of operation conditions, design characteristics, and actual and potential applications. Commonly used atomization media and their thermophysical properties are also included.
