
English for Geographers
.pdfEnglish For Geographers_____________________________________________
2. Read and translate the following text using a dictionary:
Matter
Suppose there were no limit to the power of our microscopes, so that we could examine a drop of water under stronger and stronger lenses indefinitely. What sort of microscopic world would we discover when the drop was enlarged, say, a million times? Would we still see structureless, transparent, liquid water? Or would we perhaps see distinct particles, the building blocks, as it were, of the water that to our unaided senses is a completely uniform substance? Long ago people began to suspect that matter, despite its appearance of being continuous, actually possesses a definite structure on a microscopic level. This suspicion did not take on a more concrete form until early in the last century. Since then the existence of atoms and molecules, the ultimate particles of matter in its common forms, has been amply demonstrated, and their own ultimate particles have been identified and studied as well.
Elements and Compounds
Elements are substances that cannot be decomposed or transformed into one another by ordinary chemical or physical means. The earth contains only a limited number of elements, and all other materials consist of two or more of them combined in various ways. Of the 105 known elements (not all found on the earth), 11 are gases, 2 are liquids, and the rest are solids at room temperature and atmospheric pressure. Hydrogen, oxygen, chlorine, and neon are familiar gaseous elements; bromine and mercury are the two liquids; iron, zink, tin, aluminium, copper, lead, silver, gold, carbon, and sulphur are among the solid elements.
Some materials consist of two or more elements united in a compound; water is a compound of the elements hydrogen and oxygen. The elements in a compound are combined in definite, invariable proportions to form a new substance with characteristic properties of its own. In water, every gram of oxygen is combined with precisely 0.126 g of hydrogen, and it is a liquid at room temperature whereas hydrogen and oxygen are gases. Other materials consist of mixtures of elements or of compounds in which the separate substances do not lose their identities as they do in the case of a compound, and can be present in variable proportions.
The ultimate particles of an element are called atoms. A molecule is a group of atoms that stick together tightly enough to act as a unit. Many elemental gases consist of molecules instead of individual atoms. Thus gaseous oxygen contains molecules each of which is a pair of oxygen atoms
82
_____________________________________________English For Geographers
bound together by forces whose nature we shall explore shortly. Other elemental gases, for instance helium and neon, consist of individual atoms. Most elemental solids and liquids are assemblies of individual atoms.
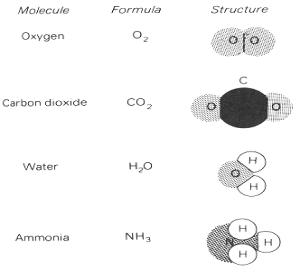
Many compounds consist of molecules. The molecules of a compound have specific compositions and structures. Each water molecule contains two hydrogen atoms and one oxygen atom with the hydrogen atoms 105˚ apart, for example, while each ammonia molecule contains three hydrogen atoms 107.5˚ apart.
Fig. 9. Structures of several common molecules
Chemical Symbols
By convention an atom of an element is represented by an abbreviation of the element’s name. For many elements the first letter is used; an atom of oxygen is O, an atom of hydrogen H, an atom of carbon C. When the names of two elements begin with the same letter, two letters are used in the abbreviation for one or both: Cl stands for an atom of chlorine, He for helium, Zn for zinc. For some elements abbreviations of Latin names are used: a copper atom is Cu (cuprum), an iron atom Fe (ferrum), a mercury atom Hg (hydrargyrum). These abbreviations are called the symbols of the elements.
83
English For Geographers_____________________________________________
Two or more atoms joined to form a molecule are represented by writing their symbols side by side: a carbon monoxide molecule is CO, a zinc sulphide molecule ZnS, a mercuric oxide molecule HgO. When a molecule contains two or more atoms of the same kind, a subscript indicates the number present: The familiar expression H2O means that a molecule of water contains two H atoms and one O atom; a molecule of oxygen, containing two O atoms, is written O2; a molecule of carbon tetrachloride (CCl4) contains one C atom and four Cl atoms; a molecule of nitrogen pentoxide (N2O5) contains two N (nitrogen) atoms and five O atoms. Each subscript applies only to the symbol immediately before it. These expressions for molecules are called formulas.
As a shorthand method of expressing the results of a chemical change, the formulas of the substances involved can be combined into a chemical equation. An equation includes the formulas of all the substances entering the reaction on the left-hand side with the formulas of all the products on the right-hand side. The formulas may be written in any order and are connected by + signs; between the two sides of the equation is placed an arrow. Thus, when carbon burns, the two substances that react are carbon (C) and oxygen (O2), and the only product is carbon dioxide (CO2):
C + O2 → CO2
This equation means, in words: “carbon reacts with oxygen to form carbon dioxide”.
3.Answer the following questions:
1.What world is the writer talking about in the first paragraph?
2.What has been proved to exist over the last century?
3.How many known elements are there?
4.What kinds of elements are there?
5.What kinds of element are copper, mercury, and chlorine?
6.What is a compound?
7.Do the elements of a compound change?
8.What is the difference between an atom and a molecule?
9.What is a symbol of an element?
10.Give an example of a chemical equation.
4.Put questions to the following statements:
1.Long ago people began to suspect that matter actually possesses a definite structure on a microscopic level.
2.Many compounds consist of molecules.
3.Some materials consist of two or more elements united in
84
_____________________________________________English For Geographers
a compound.
4.Each water molecule contains two hydrogen atoms and one oxygen atom.
5.For some elements abbreviations of Latin names are used.
5.Look at paragraph 1 and say which words are used to mean:
●a piece of glass through which you look in cameras and other instruments
●to make bigger
●which can be seen through
●fully
6.Look at paragraphs 2 and 3 and say which words have the opposite meaning to:
●unusual
●separate
●changing
●inexactly
7.Look at paragraph 4 and say which words in the text you could replace with:
●closely
●so
●look into
●for example
●groupings
8.Look at paragraphs 6 – 8. Can you explain the following words:
●abbreviation
●subscript
●formula
9.Express the following in words:
●()
●+
●→
85
English For Geographers_____________________________________________
10.Can you explain what the following are? Try not to look back at the text when you explain!
1.CO2
2.Ammonia
3.O2
4.H2O
11.Unscramble the following words and translate them:
pmiscroscoe, clhiorne, cmoeleul, rfoamuls, cporpe, sustbaencs, yogxen, absrevbiaotin.
12. Match the word with its explanation:
reaction |
- a statement of equality; |
symbol |
- a short form of a word or expression or the act |
|
of abbreviating; |
formula |
- the smallest unit into which any substance can |
|
be divided without losing its own chemical nature, |
|
usually consisting of two or more atoms; |
abbreviation |
- a letter, number, or sign that represents a |
|
sound, an amount, or chemical substance; |
molecule |
- a series of numbers or letters that represent |
|
a scientific rule; |
equation |
- a chemical change that happens when two or |
|
more substances are mixed together; |
expression |
- something that is produced through a natural |
|
or chemical process; |
product |
- a sign or group of signs that represent idea or |
|
quantity. |
Section 2
1.Think of as many words as possible related to the theme ”Recycling”.
How can each of us recycle objects?
2.Read the following text with the help of a dictionary and answer the questions:
Recycling
Julie Lewis is wearing an expensive-looking pair of boots. They are durable yet fashionable. To look at them you would never know that they were made entirely of recycled materials. Recycling has become extremely
86
_____________________________________________English For Geographers
popular in the US in recent years when the number of recycling schemes has increased by 500 percent. 65 percent of aluminium cans are recycled plus a quarter of paper and 20 percent of glass.
America even exports its waste. Taiwan buys used paper to make more paper and Japan uses American scrap metal and makes it into new cars, which it then sells back to the US.
Disposal of wastes poses a major problem. Landfill sites can cause pollution of water supplies.
A hugely successful scheme has been operating in Palm Beach County since 1988. Rubbish is sorted out into different categories. Paper, glass and plastic are sold to recycling firms. Other materials are used to make soil, which is then used for growing fruit and vegetables. Of course, recycling can be expensive.
Manufacturers in Germany are trying to solve the problem at the production stage, by looking at how their goods are packaged. Soap powders are concentrated and toothpaste tubes are sold without boxes.
1.Americans now recycle far more than they used to because a) they are competing with Germany
b) they want to make a profit from recycling c) they need more aluminium
d) they produce more rubbish than other countries
2.What does the Palm Beach County scheme involve?
a)making energy out of waste
b)selling fruit and vegetables for profit
c)producing plastic bottles
d)opening new recycling businesses
3. One of the arguments against recycling is that
a)it makes no sense using landfill sites
b)it damages the environment
c)no one is interested in running the scheme
d)it can be uneconomical to ship waste products cross-country German manufacturers
a)are concentrating on improving soap powders
b)sell toothpaste in boxes instead of tubes
c)have cut down on packaging
d)are trying to make goods cheaper
87
English For Geographers_____________________________________________
3.Fill in the gaps with the appropriate synonymous words from the list below:
packaged, long-term, entirely, waste, wrapping, good investment,transport, site, sorted, desirable
We should try to dispose of our ……… in a way that won’t harm the environment (rubbish).
The hole in the ozone layer will have ……… effects on the world’s climate (long-lasting).
An empty piece of land outside town is the ………. for the new supermarket (location).
Reducing the size of classes in state schools is a(n) ……… aim
(attractive).
One of the most convenient means of ……… is the aeroplane (travel). Frozen vegetables are usually ……… in sealed plastic bags (put in). We should try to buy products with as little ……… as possible
(packaging).
Rubbish must be ……… before it is sent for recycling (separated into similar types).
Their house is built ……… of materials taken from demolished buildings (totally).
That unique table was a ……… ; it has tripled in value since I bought it
(profitable purchase).
4.Fill in the correct word derived from the words in brackets:
If we want to find a 1) ……… (solve) to the world’s waste disposal problems we should look at Palm Beach county in America. The county has an 2) ……… (ambition) recycling scheme which has been 3) ………
(extreme) successful. Waste is almost entirely recycled. Even the kitchen waste is used to provide 4) ……... (electric).
5.Choose the correct item:
1.Honey is a sweet ……… that is produced by bees.
a. material |
b. substance |
c. matter |
d. fabric |
Factories must stop pumping toxic ……… into rivers.
a. garbage |
b. rubbish |
c. litter |
d. waste |
Each sweet comes ……… in silver paper.
a. enclosed |
b. folded |
c. packaged |
d. wrapped |
Her mother works in a bank as a financial ……… .
a. councillor |
b. counsellor |
c. adviser |
d. informer |
88
_____________________________________________English For Geographers
She ……… out some old clothes and gave them to a charity shop.
a. Sorted |
b. selected |
c. separated |
d. segregated |
The new ……… to help young people find jobs has been very |
|||
successful. |
|
|
|
a. Pattern |
b. scheme |
c. plot |
d. system |
6. Fill in the gaps with the appropriate word(s) from the list below: plastic, environmentally aware, recycling, environment, products, aluminium, decompose, disposing of, bottle banks
It is important these days to try to be as 1) ……… as possible, which means 2) ……… our waste instead of 3) ……… it in the usual way. For example, instead of throwing 4) ……… cans away, they can be crushed and taken for recycling, and bottles can be taken to 5) ……… instead of being put in the rubbish bin. We should try to use as little 6) ……… as possible because it takes a long time to 7) ……… and to buy recycled paper to write on. Recycled 8) ……… are often the same price as normal ones, but are a lot kinder to the 9) ……… .
7. Put the verbs in brackets into the correct tense:
If I were a scientists, I 1) ……… (find) a way to repair the hole in the ozone layer. Scientists believe that if world temperatures 2) ……… (rise), sea levels 3) ……… (rise) too and many towns 4) ……… (be flooded). It is also possible that many fertile places 5) ……… (become) desert. If this happened, there 6) ……… (be) chaos throughout the world. If people had disposed of their rubbish more carefully, the environment 7) ……… (not/be polluted) in the first place. Governments are also responsible for this situation, because if there had been tighter laws on pollution, factories 8) ……… (not/release) such harmful chemicals into the atmosphere.
8. Match the word with its explanation:
aluminium |
- the process of treating used objects or materials |
|
so that they can be used again; |
durable |
- food, paper, etc that is no longer needed and has |
|
been thrown away; |
scrap metal |
- things that are produced in order to be sold; |
scheme |
- a program, an official plan that is intended to |
|
help people in some way or to organize activities; |
recycling |
- a silver-white metal that is very light and is used |
|
to make cans, cooking pans, etc. It is a chemical |
|
element; |
|
89 |
English For Geographers_____________________________________________
goods |
- metal from old cars, machines that are no longer |
|
used for the purpose they were made for, but can |
|
be used again in another way; |
rubbish |
- staying in good condition for along time, even if |
|
used a lot. |
|
Unit 12 |
Section 1
1. Learn the following words and their translation. Check up yourself by
back translation: |
|
minerals |
- мінерали |
rocks |
- гірська порода |
aggregates |
- сукупність |
limestone |
- вапняк |
apparent |
- очевидний |
discern |
- розпізнавати |
soluble |
- розчинний |
silicates |
- силікати |
abundant |
- багатий, численний |
mica |
- слюда |
feldspar |
- польовий шпат |
conspicuous |
- помітний, значний |
ore |
- руда |
iron |
- залізо, залізна руда |
galena |
- галеніт |
native |
- самородний, чистий, щирий |
redundancy |
- дублювання |
lattice |
- просторова решітка кристала |
nomenclature |
- термінологія, найменування |
constituents |
- компоненти |
cleavage |
- кліваж, шарування |
minute |
- ретельний |
cracks |
- тріщина, розколина, шпара |
|
90 |
_____________________________________________English For Geographers
2. Read and translate the following text using a dictionary:
Minerals
Rocks are aggregates of substances called minerals, which as a rule are crystalline solids with fairly definite compositions and structures. Some rocks, for instance limestone, consist of a single mineral only, but the majority consists of several minerals in varying proportions. The different minerals in a coarse-grained rock like granite are apparent to the eye; in fine-grained rock, the separate minerals can be discerned with the help of a microscope.
What Minerals Are
It is not difficult to understand why certain substances occur as minerals and why others do not. We expect to find the more chemically inactive elements, such as gold, platinum, and sulphur, in the free state, whereas chemically active elements, such as sodium, calcium, and chlorine, are always found in combination as compounds. Compounds readily soluble in water, such as sodium chloride, sodium carbonate, and potassium nitrate, form deposits in desert regions but are rare elsewhere. Substances that tend to react with oxygen occur only well below the surface away from the oxygen of the atmosphere. Unstable compounds like phosphorus pentoxide are necessarily absent from the earth’s crust.
Silicates are by far the most abundant minerals; mica, feldspar, and topaz are familiar examples. Carbonates are another important class, its most conspicuous representative being the carbonate of calcium called calcite. Oxides and hydrated oxides include such common materials as hematite (ferric oxide), the chief ore of iron, and bauxite (hydrated aluminium oxide), the chief ore of aluminium. Various metals are obtained from deposits of sulphide minerals, such as galena (lead sulphide) and sphalerite (zinc sulphide). Elements that occur free, or native, were mentioned above. Less frequent as minerals are sulphates, phosphates, and chlorides.
Unfortunately the study of minerals requires the learning of a special list of names, some of them apparently duplicates of other names. As an example, the mineral whose formula is CaCO3 is given the name calcite instead of the chemical name calcium carbonate. For this seeming redundancy there two reasons:
91
