
ECHO 2013 / Acute Coronary Syndromes Echo in the Assessment of Complications
.pdf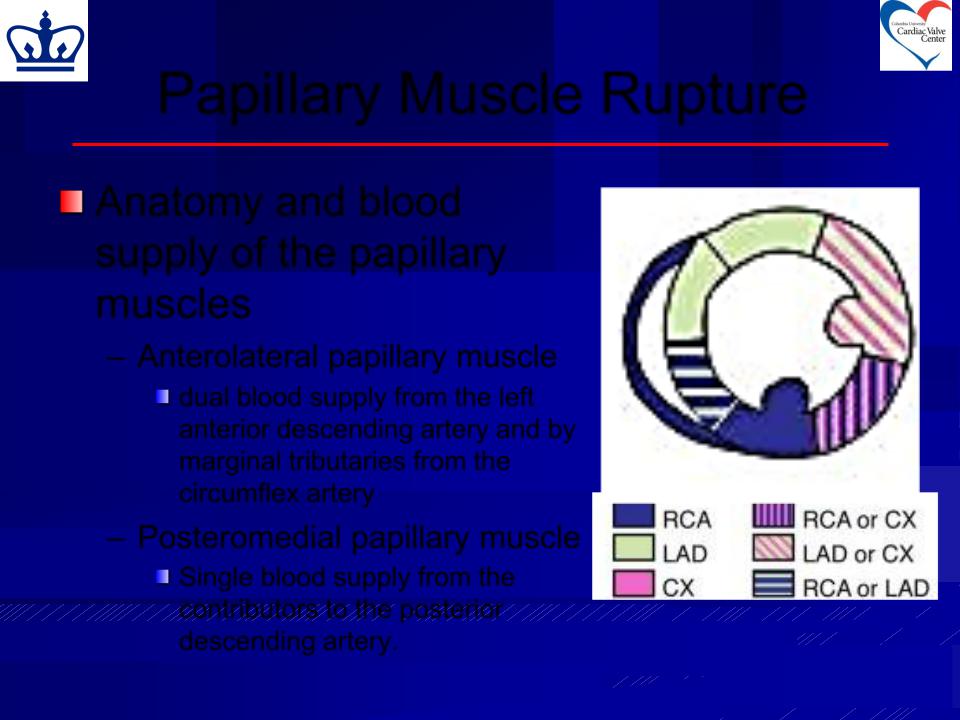
Papillary Muscle Rupture
Anatomy and blood supply of the papillary muscles
– Anterolateral papillary muscle
dual blood supply from the left anterior descending artery and by marginal tributaries from the circumflex artery
– Posteromedial papillary muscle
Single blood supply from the contributors to the posterior descending artery.
Papillary Muscle Rupture
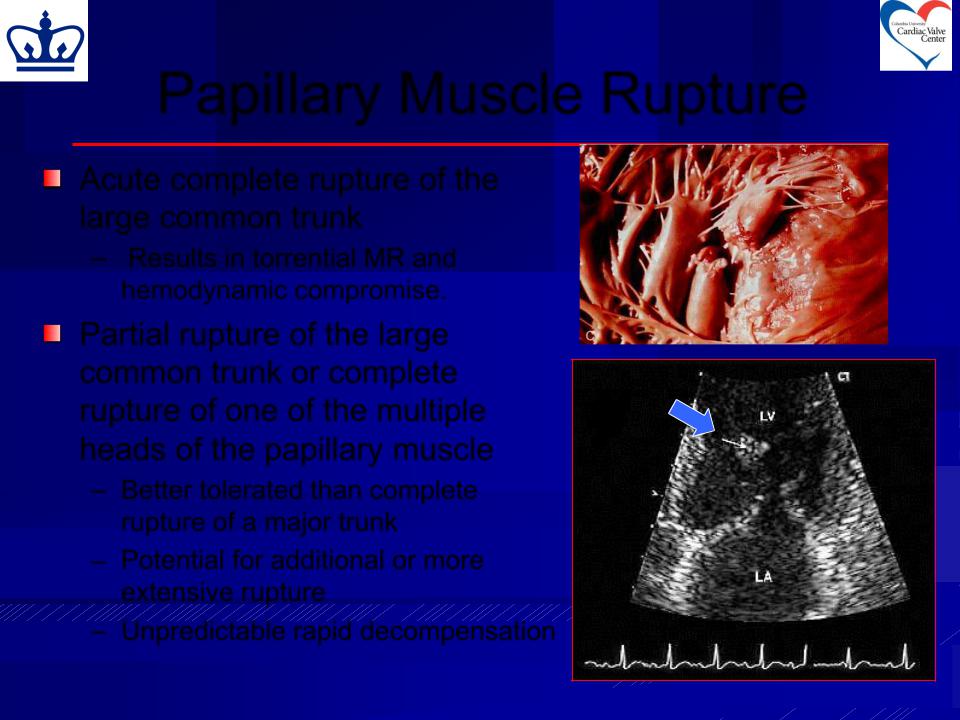
Acute complete rupture of the large common trunk
– Results in torrential MR and hemodynamic compromise.
Partial rupture of the large common trunk or complete rupture of one of the multiple heads of the papillary muscle
–Better tolerated than complete rupture of a major trunk
–Potential for additional or more extensive rupture
– Unpredictable rapid decompensation

Papillary Muscle Rupture
Clinical presentation and diagnosis
–PMR entails <5% of ischemic MR
–Incidence ~ 1% of all AMIs and accounts for 5% of the mortality following AMI
More common with inferior wall MI
Nearly twice the mortality seen in those with a ventricular septal defect after an acute infarct
–Usually occurring 2-7 days post AMI
–Clinical presentation
Acute rapid hemodynamic deterioration
Pulmonary edema/cardiogenic shock
Severe dyspnea with raised venous pressure and rales throughout the lung fields
Murmur of mitral regurgitation may be present but may be inaudible because of the rapid equalization of left atrial and left ventricular pressures
Acute rupture of the ventricular septum may present in a similar manner but patients may not have the severe dyspnea and orthopnea seen in patients with severe mitral regurgitation: a loud systolic murmur and thrill are almost always present.

Papillary Muscle Rupture
Diagnostic test of choice: Echocardiography
– TTE
Flail mitral valve leaflet with systolic prolapse into the left atrium
Mobile mass (papillary muscle) attached to the chordae tendinae and to the mitral valve
Abnormal cutoff of one PM
–Diagnosis suspected by the presence of hemodynamic compromise concomitant with preserved systolic function
Papillary Muscle Rupture
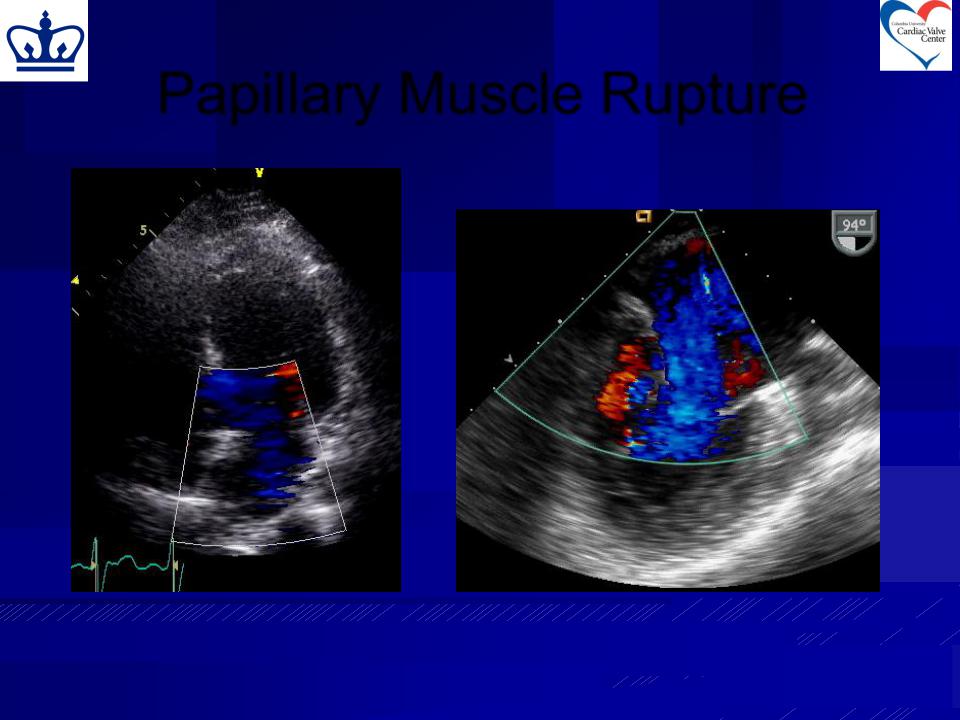
TTE Color: eccentric jet, area  poorly-imaged (equilibration of TEE Color Doppler
poorly-imaged (equilibration of TEE Color Doppler
LA and LV pressures)
Papillary Muscle Rupture
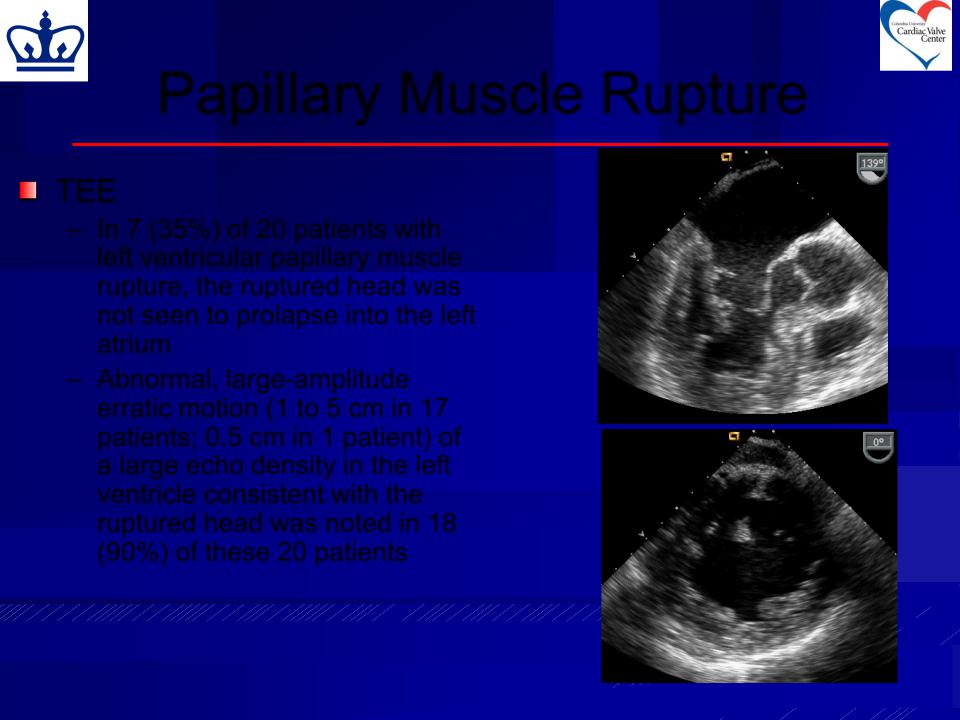
TEE
–In 7 (35%) of 20 patients with left ventricular papillary muscle rupture, the ruptured head was not seen to prolapse into the left atrium
–Abnormal, large-amplitude erratic motion (1 to 5 cm in 17 patients; 0.5 cm in 1 patient) of a large echo density in the left ventricle consistent with the ruptured head was noted in 18 (90%) of these 20 patients
Moursi MH et al. Circulation. 1996;94:1003-1009
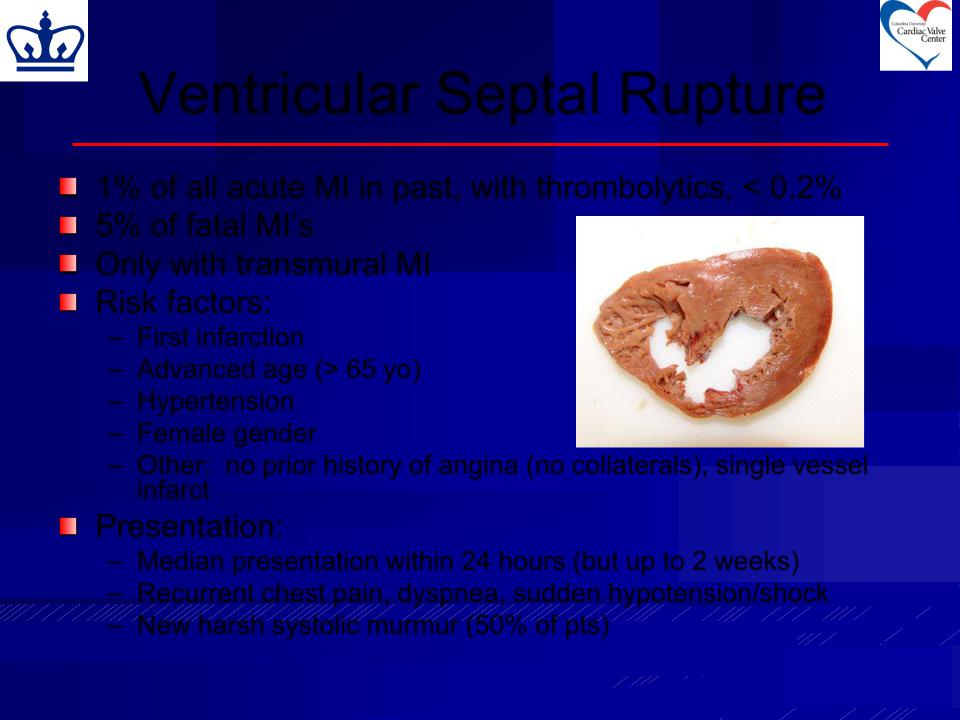
Ventricular Septal Rupture
1% of all acute MI in past, with thrombolytics, < 0.2%
5% of fatal MI’s
Only with transmural MI Risk factors:
–First infarction
–Advanced age (> 65 yo)
–Hypertension
– Female gender
–Other: no prior history of angina (no collaterals), single vessel infarct
Presentation:
–Median presentation within 24 hours (but up to 2 weeks)
–Recurrent chest pain, dyspnea, sudden hypotension/shock
– New harsh systolic murmur (50% of pts)
TTE
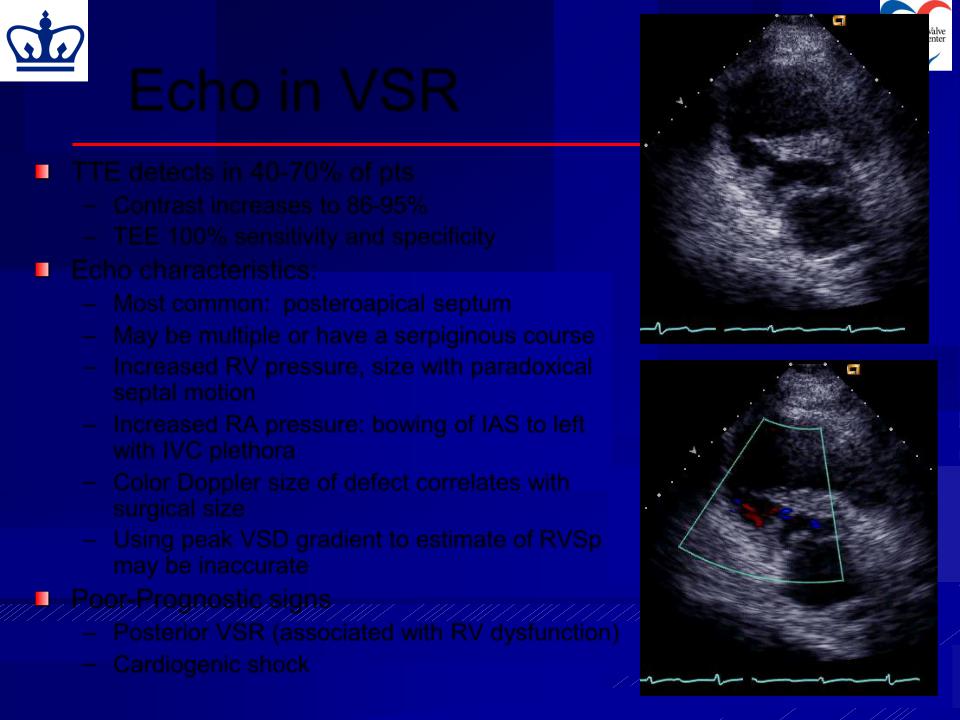
Echo in VSR
TTE detects in 40-70% of pts
–Contrast increases to 86-95%
–TEE 100% sensitivity and specificity
Echo characteristics:
– Most common: posteroapical septum |
|
|
– May be multiple or have a serpiginous course |
|
|
– Increased RV pressure, size with paradoxical |
TTE |
|
septal motion |
||
|
– Increased RA pressure: bowing of IAS to left with IVC plethora
–Color Doppler size of defect correlates with surgical size
–Using peak VSD gradient to estimate of RVSp may be inaccurate
Poor-Prognostic signs
– Posterior VSR (associated with RV dysfunction)
– Cardiogenic shock
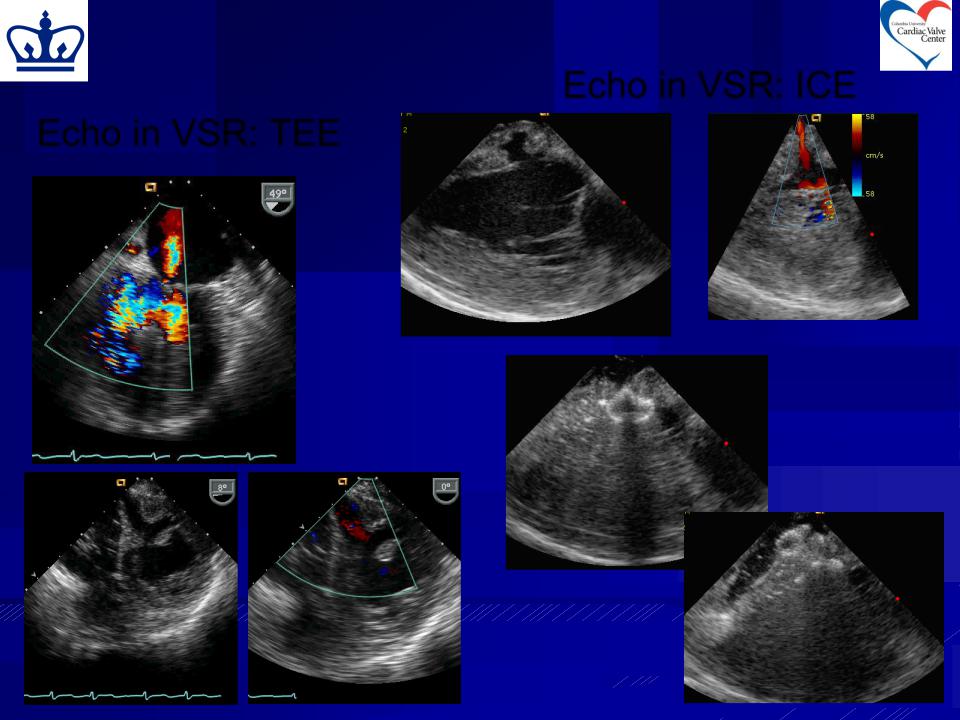
Echo in VSR: ICE
Echo in VSR: TEE
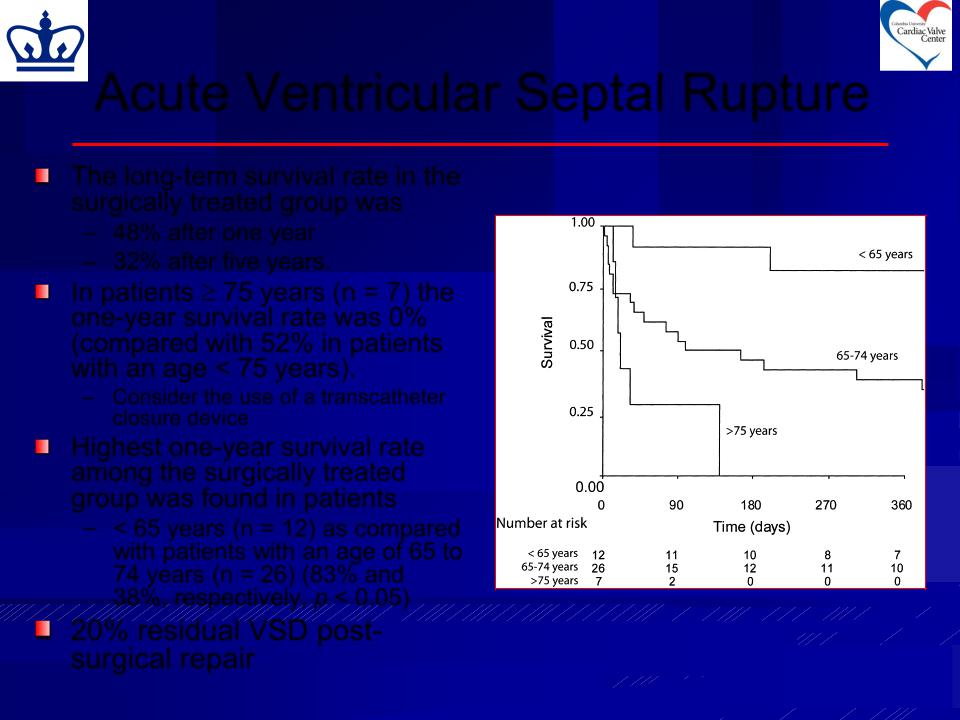
Acute Ventricular Septal Rupture
The long-term survival rate in the surgically treated group was
–48% after one year
–32% after five years.
In patients 75 years (n = 7) the one-year survival rate was 0% (compared with 52% in patients with an age < 75 years).
– Consider the use of a transcatheter closure device
Highest one-year survival rate among the surgically treated group was found in patients
–< 65 years (n = 12) as compared with patients with an age of 65 to 74 years (n = 26) (83% and
38%, respectively, p < 0.05)
20% residual VSD postsurgical repair
Poulsen SH et al. Ann Thor Surg 2008;85:591-1596
