
- •Биологическое окисление 1
- •Содержание
- •Биоэнергетика
- •История учения о БО
- •Antoine Lavoisier
- •Теория активации кислорода
- •Критическая оценка теории Баха-Энглера
- •Хромогены и гистогематины
- •Редокс-реакции, редокс потенциал
- •Maкроэргичность ATФ
- •ATФ-AДФ цикл
- •Образование субстратов БО
- •Stages 1 and 2
- •Stage 3
- •Ферменты и коферменты БО
- •Структура FAD и FMN
- •Мх: локализация
- •Общий план строения Мх
- •Internal structure of a mitochondrion
- •The Comparative Characteristics
- •Membrane Composition:
- •Tricarboxylic Acid Cycle
- •Krebs’
- •Role of TCA
- •Plastic Role of
- •Regulation of
- •Regulation of the TCA Cycle (cont’d)
- •Inhibitors of Krebs Cycle
- •Content
- •Introduction
- •The Ways of Oxygen Consumption
- •Biologic Oxidation (BO)
- •Biomedical Importance of BO
- •Energy Conversion: Mitochondria
- •Chemiosmotic Coupling
- •Electron Transporting Chain, ETC
- •Electron Transporting Chain (ETC)
- •ETC functions
- •ETC Complexes: Overview
- •Complex I (NADH-CoQ reductase)
- •Coenzyme Q (CoQ) or Ubiquinone
- •Complex II (Succinate-CoQ reductase)
- •Complex II and III
- •Complex IV: Cytochrome c Oxidase
- •ATP/ADP translocase
- •Respiratory Chain Functioning
- •Functional scheme of ETC
- •Inhibitors of Oxidative Phosphorylation
- •The Structures of Several Inhibitors of
- •The Sites of Action of Several Inhibitors of ETC and/or OP
- •Several Uncouplers of OP
- •Uncoupler Action
- •Endogenous Uncouplers Enable
- •P/O Ratio
- •Disorders of Mitochondrial
- •Clinical Manifestation and
- •Some Mitochondrial Diseases
- •LHON
- •MERRF, MELAS et al.
- •Can Mitochondrial Diseases be Treated?
- •Cytochromes P450 are monooxygenases important for the detoxification of many drugs
- •Cytochrome b5
- •Monooxygenase System (Microsomal
- •Functioning of Microsomal
- •Microsomal Oxidation and Cytochrome P450

Chemiosmotic Coupling
sunlight
 foodstuff
foodstuff
High Energy
Electrons
Transmembrane electrochemical proton gradient
Active |
|
ATP |
|
Bacterial |
membrane |
|
synthesis |
|
flagellar |
transport |
|
|
|
rotation |
|
|
The common pathway used by mitochondria, chloroplasts, and prokaryotes to harness energy for biological purposes operates by a process known as chemiosmotic coupling – reflecting a link between the chemical bond-forming reactions that generate ATP ("chemi") and membrane-transport processes ("osmotic").
The coupling process occurs in two linked stages, both of which are performed by protein complexes embedded in a membrane.

Electron Transporting Chain, ETC
(1/2)
The mitochondria contain the series of catalysts known as the respiratory chain (electron transporting chain, ETC) that collect and transport reducing equivalents and direct them to their final reaction with oxygen to form water.
ETC components are imbedded to the inner membrane of mitochondria.
07/06/19

Electron Transporting Chain (ETC)
(2/2)
07/06/19

ETC functions
1.It is the final common pathway in aerobic
 cells.
cells.
2.NAD+ and FAD are reduced to NADH + H+
and FADH2 respectively in most oxidation reactions. By ETC these coenzymes are reoxidized to NAD+ and FAD.
07/06/19

ETC Complexes: Overview
07/06/19

Complex I (NADH-CoQ reductase)
Contains:
.
.
FMN
FeS centres (22-24 iron- sulfur (Fe-S) proteins in 5-7 clusters).
Electron acceptor is CoQ
07/06/19

Coenzyme Q (CoQ) or Ubiquinone
O
H3C O

 CH3 H3C O
CH3 H3C O





O |
CH3 10 |
CoQ is a component of the inner mitochondrial membrane involved in the process of electron transport.
It draws electrons into the respiratory chain, not only from
NADH but also from succinate.
07/06/19

|
O |
CoQ10 |
|
|
H3C |
O |
CH3 |
|
|
|
|
|
|
|
H3C |
O |
|
|
|
|
|
|
|
|
|
O |
CH3 CH3 CH3 CH3 CH3 CH3 CH3 CH3 |
CH |
CH |
|
|
|
3 |
3 |
CoQ is oxidized by cytochromes, it is a collection point of electrons from several flavoprotein dehydrogenases.
07/06/19
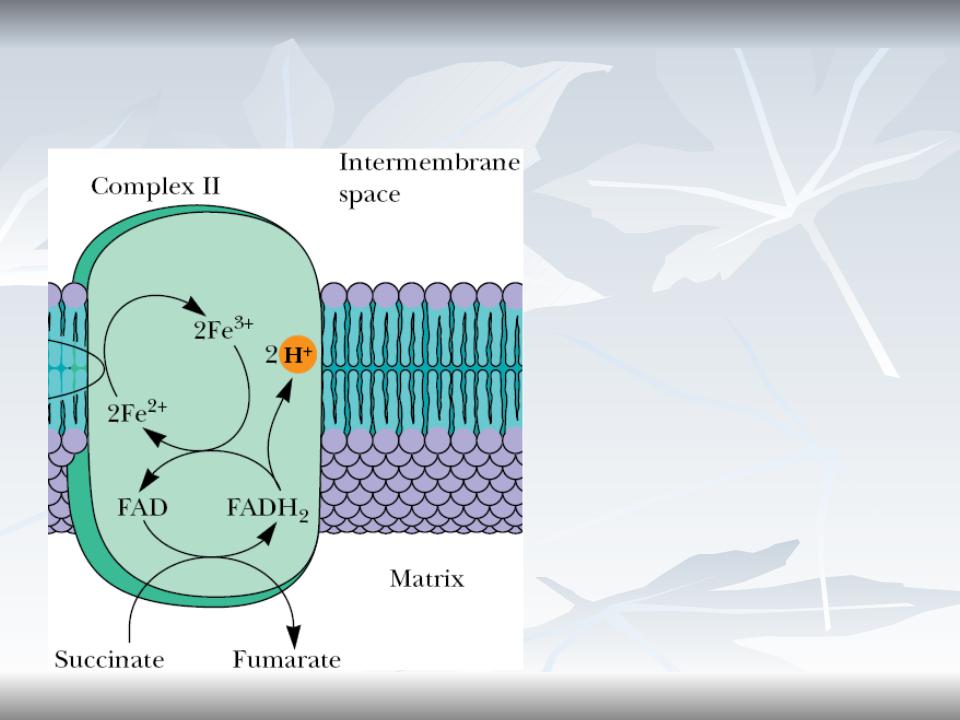
Complex II (Succinate-CoQ reductase)
Complex II contains FAD and 7-8 Fe-S proteins in 3 clusters and cytochrome
b560.
Complex II is perhaps better known by its other name— succinate dehydrogenase, the only TCA cycle enzyme that is an integral membrane protein in the inner mitochondrial membrane. This enzyme has a mass of approximately 100 to 140 kD
07/06/19
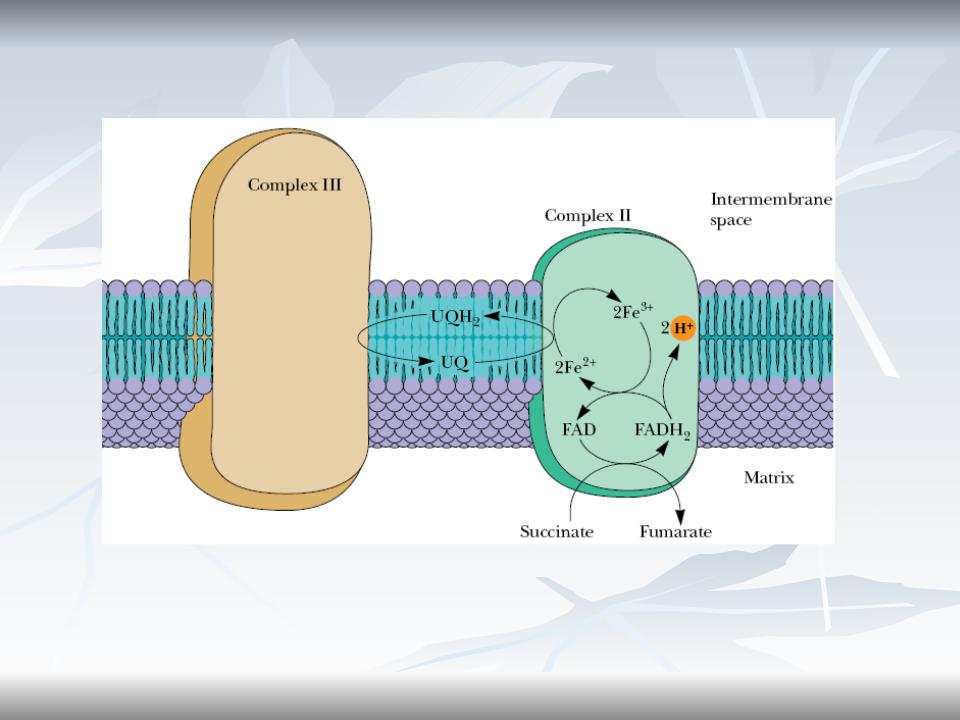
Complex II and III
CoQ accepts electrons from both complex I And complex II and donates electrons to complex III.
07/06/19
