
Карцев В.Г.Избранные методы с-за и модифик. гетероциклов т.1 , 2003
.pdf
Вместе с тем, анализируя выявленные (подчеркнем, локальные) тенденции влияния структуры на рассматриваемые "макрохарактеристики" СЕ, нельзя исключать такие важные факторы, как ионофорные и комплексообразующие свойства исследованных соединений, а также "реакционную способность" образующихся комплексов. Отметим, например, что замещение на серу атома кислорода в DPO фрагменте СЕ типа В, приводит к полной потере противомикробной активности DPSCE (D).
Нам представляется, что изложенное позволяет, с одной стороны, более обоснованно реализовать направленный синтез СЕ с заданными свойствами, а с другой – осуществить методами QSPR/QSAR молекулярный дизайн и прогноз СЕ с оптимизированной ("специализированной") структурой.
Литература
1.Золотов Ю.А., Кузьмин Н.М., Макроциклические соединения в аналитической химии, М.: Наука, 1993.
2.Яцимирский К.Б., Кольчинский А.Г., Павличук В.В., Таланова Г.Г.,
Синтез макроциклических соединений, Киев: Наукова думка, 1987.
3.Грагеров И.П., Краун-соединения в органическом синтезе, Киев: Наукова думка, 1994.
4.Grygorash R.Ya., Shishkin O.V., Shishkina S.V., et al., Abstr. 27th Int. Symp. on the Macrocyclic Chemistry, Utah (USA), 2002, p. A35.
5.Grygorash R.Ya., Shishkin O.V., Shishkina S.V., et al., Abstr. 2d Int. Symp. “Molecular Design and Synthesis of Supramolecular Architectures”, Kazan (Russia), 2002, p. 97.
6.Kamalov G.L., Kotlyar S.A., Abstr. 14th Conf. “Physical Methods in Coordination and Supramolecular Chemistry”, Chisinau (Rep. Moldova), 2002, p. L14.
7.Григораш Р.Я., Ткачук В.В., Котляр С.А., Камалов Г.Л., Тез. 15-ой междунар.
научн.-техн. конф. “Химические реактивы, реагенты и процессы малотоннажной химии” РЕАКТИВ-2002, Уфа (Россия), 2002, с. 62.
8.Котляр С.А., Городнюк В.П., Григораш Р.Я., Якшин В.В., Журн. прикл. хим.
1998 71 (9) 1569.
9.Городнюк В.П., Григораш Р.Я., Чуприн Г.Н., Котляр С.А., ЖОХ 1998 68
(6)1051.
10.Котляр С.А., Городнюк В.П., Григораш Р.Я., Чуприн Г.Н., ЖОХ 1998 68
(7)1189.
11.Котляр С.А., Чуприн Г.Н., Кирияк А.В. и др., в сб. Перспективные процессы и продукты малотоннажной химии, под ред. Рахманкулова Д.Л., Уфа:
РЕАКТИВ, 2002, с. 173.
12.Чуприн Г.Н., Котляр С.А., Кирияк А.В. и др., в сб. Перспективные процессы и продукты малотоннажной химии, под ред. Рахманкулова Д.Л., Уфа:
РЕАКТИВ, 2002, с. 76.
Генеральный спонсор и организатор – InterBioScreen Ltd. |
267 |
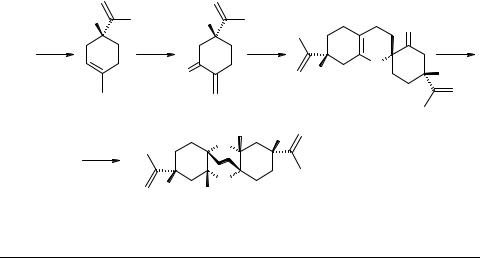
Biogenetic-type synthesis of (+)-cymbodiacetal, a constituent of Cymbopogon martini
Kamat S.P.*, D'Souza A.M.*, Paknikar S.K.**,
Dev V.***, Beauchamp P.S.***
*Department of Chemistry, Goa University, Goa, 403206 India
**Sidharth Chemicals, Kundai Industrial Estate, Kundai, Goa, 403115 India
***Department of Chemistry, California State Polytechnic University, Pomona CA, 91768 USA
tel.: + 91-832-245-4317, fax: + 91-832-245-2889 e-mail: srikamat@goatelecom.com
1. Introduction
Cymbodiacetal 1 was first isolated from the essential oil of the aerial parts of flowering Cymbopogon martinii, Gramineae [1]. The structure of 1 was established by spectroscopy (mass, IR, 1H and 13C NMR) and further confirmed by X-ray diffraction studies of its 1 : 1 solvate with CDCl3, which also established its absolute stereochemistry. The stereochemistry at C(2) and C(7) of 1 was shown to be same as that in R-(+)-limonene 2, the other component isolated from the essential oil [1].
The biogenesis of 1 involving the key intermediate 3 (Scheme 1) looked more attractive than the earlier proposal [1] especially in view of the fact that a large number of natural dimers of the type 3 are reported to be formed by hetero-atom Diels–Alder self dimerization [2–9]. We have now synthesized (+)-cymbodiacetal 1 starting with (+)-limo- nene oxide 4 via key intermediate 3. This is the first report on the synthesis of 1 [1].
Scheme 1
H |
H |
|
|
|
|
O |
|
|
|
|
|
|
|
GPP |
|
|
|
|
O |
|
O |
|
|
|
H |
H |
|
|
|
|
|
|||
2 |
5 |
|
|
|
3 |
|
9 |
10 |
|
OH 1 |
H |
|
|
8 |
O |
|
|
|
|
|
9a |
|
10a |
2 |
|
|
|
7 |
|
|
|
|||
5a |
|
4a |
3 |
|
|
|
|
O |
|
|
|
|
|
H 6 |
5 |
4 |
|
|
|
|
OH |
|
|
|
|||
|
1 |
|
|
|
|
|
Possible biogenetic pathway for cymbodiacetal 1
268 |
Устные доклады |
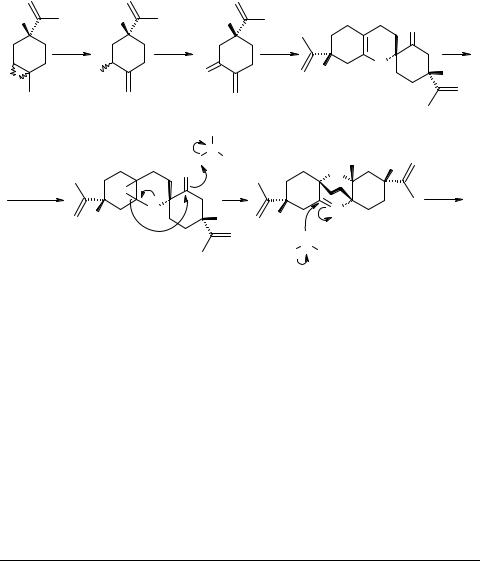
2. Results and discussion
Our synthetic strategy was based on the assumption that dimer 3 should be accessible through simple Diels–Alder dimerization of o-quinomethide 5 which in turn can be obtained from (+)-limonene oxide 4 (Scheme 2). Dimer 3 could then be transformed into monoepoxide 6 on treatment with one equivalent of moist peroxy acid. Under mild acidic conditions of the free acid formed from the peroxy acid, epoxide 6, assisted by the participation of ether and carbonyl oxygens could open up followed by attack of water to give cymbodiacetal 1 (Scheme 2).
|
|
|
|
|
|
|
|
|
|
Scheme 2 |
H |
|
H |
|
|
H |
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
|
LDA |
PCC |
|
|
|
|
|
|
|
|
O |
HO |
CH2Cl2 |
O |
|
|
|
H |
|
O |
H |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
4 |
|
7 |
|
|
|
5 |
|
|
3 |
|
|
|
|
|
H |
+ |
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
H |
|
H |
|
|
OH |
|
H |
|
moist |
|
O |
|
|
|
|
|
O |
|
|
m-CPBA |
|
O .. |
|
|
|
|
|
O |
|
1 |
|
H |
O |
|
|
H |
H |
|
|
|
|
|
.. |
|
|
|
+ |
|
|
|||
|
|
|
|
|
|
.. |
|
|
|
|
|
|
6 |
|
|
|
O |
|
|
|
|
|
|
|
|
|
H .. |
H |
|
|
|
|
Synthetic route to cymbodiacetal 1
Reaction of (–)-limonene oxide with lithium diisopropylamide (LDA) to give exocarveols followed by oxidation of these allylic alcohols to give the enantiomer of 5 has been reported [10]. We used this sequence with minor modifications.
Reaction of (+)-limonene oxide 4 with LDA gave the desired allylic alcohol 7 in 81% yield [10]. As expected, the product was a mixture of two diastereomers in the ratio of 1 : 1.82 as substantiated by GC. The allylic alcohol mixture 7 was oxidized with pyridiniumchlorochromate (PCC) in CH2Cl2 [11]. The usual work-up, followed by column chromatography on silica gel with hexane–diethyl ether (9 : 1) as the eluent, gave expected α,β-unsaturated ketone 5 in 68% yield. In view of the unstable nature of 5 [10], no other physical data (except for GC/MS) were recorded for further characterization.
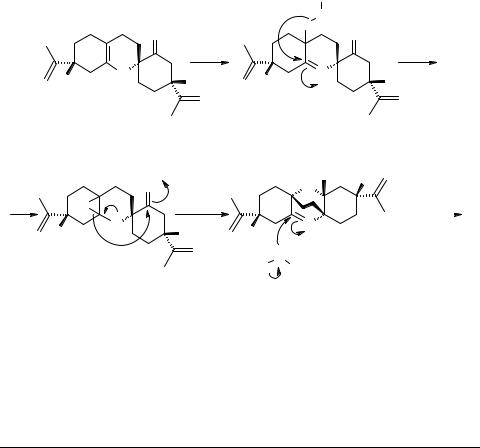
o-Quinomethides are reported [3, 6] to undergo heteroatom Diels–Alder dimerization on just standing at room temperature. Therefore, enone 5 was kept in a loosely stoppered flask at room temperature. A week later, GC of the contents of the flask showed appearance of a peak (≈10%) at retention time Rt = 66.5 min, and considerable reduction in the peak size of 5 at Rt = 29.9 min which disappeared almost completely at
Генеральный спонсор и организатор – InterBioScreen Ltd. |
269 |
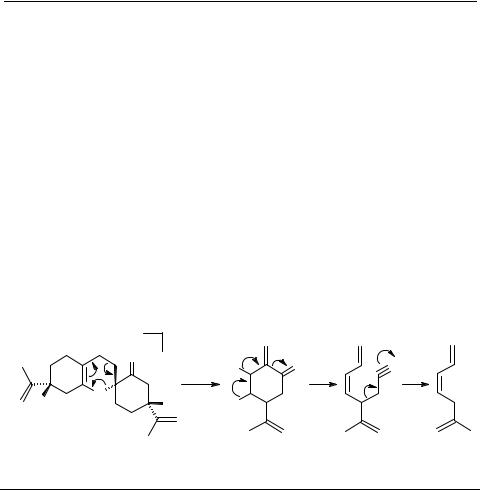
The transformation of 3 into (+)-cymbodiacetal 1 took place in an unusually simple manner, supporting the proposed biogenetic pathway (Scheme 1).
An interesting observation was made while handling 3. It was difficult to obtain 3 in pure form. The GC/MS of even 97% pure 3 showed a peak at m/z 316 in addition to the molecular ion (m/z 300) expected for 3. A reasonable explanation to this observation was the formation of epoxide 6 having molecular formula C20H28O3. Therefore, we thought of leaving the dimer exposed to diffused daylight expecting to obtain desired epoxy intermediate 6. Indeed, 6 must have been formed (not characterized), because chromatography of 3 over silica gel exposed to daylight for two days gave a crystalline compound (m.p. 213°C) and identified as cymbodiacetal 1. Direct comparison of the spectral data, (IR, 1H and 13C) unambiguously established the identity of the synthetic product with the natural cymbodiacetal 1 [1].
A probable mechanism for the formation of 6 by air oxidation [12, 13] of 3 and its conversion to 1 is presented in Scheme 4.
Scheme 4
|
|
|
|
H |
|
|
|
O |
|
|
O O |
O |
|
|
hν/O2 |
|
|
|
||
|
|
|
|
|
|
|
H |
O |
H |
H |
O |
H |
−OH |
|
.. |
|
||||
|
3 |
|
|
|
|
|
|
H+ |
Silica gel |
|
OH |
H |
|
|
O |
chromato- |
|
O |
Moisture from |
|
|
O .. |
graphy |
|
|
the solvent/air 1 |
|
|
|
|
O+ |
|||
H |
O |
H |
H |
|
|
|
|
|
|
.. |
|
|
|
|
6 |
|
O |
H |
|
|
|
|
H .. |
|
|
||
A probable mechanism for the formation of 6 and its conversion to 1 from 3
A pertinent question that arises from the observed direct formation of 1 from 3 by air oxidation followed by silica gel chromatography is, whether cymbodiacetal 1 a natural product, or an artifact? We believe 1 to be a natural product, which is formed by a [4+2]-cycloaddition reaction catalyzed by hetero-atom Diels–Alderase [14]. In conclusion, we have successfully carried out biomimetic synthesis of (+)-cymbodiacetal 1.
Генеральный спонсор и организатор – InterBioScreen Ltd. |
271 |


of 5. Column chromatography over silica gel and elution with hexane–diethyl ether (19 : 1) gave fractions rich in peak at Rt = 66.5 min (GC) and were combined. GC/MS of the major peak (77% intensity) showed m/z (rel. intensity) 300(M+, 55), 151(23), 135(28), 107(100), 95(50), 79(47), 67(32), 55(36), 41(30) indicating it to be the dimer 3, molecular formula C20H28O2. Further purification of a small sample (< 50 mg) by preparative HPLC using 5% diethyl ether in hexane as the eluent gave 97.4% pure (GC) dimer 3 (11 mg). The purified sample did not solidify. 1H NMR (CDCl3, 300 MHz) δ 1.65 (6H, s, 2CH3), 1.4-2.3 (16H s, m, 7CH2 and 2CH), 2.8 (2H, t1, J = 12.1, 11.6 Hz, –CH2–CO), 4.58 (2H, s, –C=CH2), 4.72 (2H, s, –C=CH2); 13C NMR (CDCl3, 300 MHz) δ 20.3 (C-9 or C-9'), 20.8 (C-9 or C-9'), 22.6 (C-10), 25.4 (C-6'), 27.3 (C-10'), 27.7 (C-5), 28.5 (C-5'), 33.0 (C-6), 38.9 (C-3'), 41.7 (C-4'), 43.4 (C-3), 48.5 (C-4), 79.3 (C-1), 105.5 (C-1'), 108.7 (C-8 or C-8'), 109.9 (C-8 or C-8'), 143.8 (C-2'), 147.4 (C-7 or C-7'), 149.2 (C-7 or C-7'), 212.4 (C-2); 13C NMR-DEPT, see Table 1. GC/MS of the minor (10%) peak showed m/z (rel. intensity) 316(M+, 43), 163(26), 149(58), 135(72), 120(100), 107(86), 95(48), 79(57), 67(40), 55(53), 41(38) indicating it to be the epoxide 6, molecular formula C20H28O3.
3.5. Cymbodiacetal (1)
The remaining portion of impure 3 (1.0 g) on chromatography over silica gel in diffused daylight and elution with hexane:diethyl ether (4:1) gave colorless needles (0.288 g), m.p. 213°C. (206-207°C [1]); [α]D = + 24.2, CHCl3 (+ 26o ± 5o, CHCl3 [1]); IR (KBr), νmax: 3379 (OH), 2941, 1649, 1450, 1180, 1128, 1080, 1006, 891 cm–1; 1H NMR
(CDCl3, 300 MHz) δ 1.68 (3H, s, CH3), 1.49-1.92 (6H, m, C8, C9, C11-H, s), 2.09 (1H, m, C1-H, s), 4.66 (2H, s, =CH2); 13C NMR (CD3OD, 300 MHz) δ 21.1 (CH3), 27.3 (C-1 or
C-3), 27.5 (C-1 or C-3), 34.1 (C-11), 41.9 (C-2 or C-4), 42.4 (C-2 or C-4), 72.8 (C-4a), 98.9 (C-5a), 109.1 (=CH2), 150.7 (–C=CH2).
References
1.Bottini A.T., Dev V., Garfagnoli D.J., et al., Phytochemistry 1987 26 (8) 2301.
2.Carreiras M.C., Rodriguez B., Lopez-Garcia R.E., Rabanal R.M., Phytochemistry 1987 26 (12) 3351.
3.Kakiuchi K., Ue M., Takeda M., et al., Chem. Pharm. Bull. 1987 35 (2) 617.
4.Richer J.C., Arlotto R., Can. J. Chem. 1975 53 3294.
5.Whittaker D., Banthrope D.V., Chem. Rev. 1972 305.
6.Hikino H., Aota K., Takemoto T., Chem. Pharm. Bull. 1967 15 (12) 1929.
7.Nakajima T., J. Pharm. Soc. Jpn. 1962 82 1278.
8.Klinck R.E., de Mayo P., Stothers J.C., Chemistry and Industry (London) 1961 471.
9.Takemoto T., Nakajima T., Yakugaku Zasshi 1957 77 1157.
10.Wang Q., Fan S.Y., Wong N.C., et al., Tetrahedron 1993 49 (3) 619 and ref. 16 within.
11.Ghisalberti E.L., Jefferies P.R., Stuart A.D., Aust. J. Chem. 1979 32 1627.
12.Lösing G., Degener M., Matheis G., Dragoco Report 1998 4 181.
13.Ngo K., Wong W., Brown G.D., J. Nat. Prod. 1999 62 549.
14.Rasser F., Anke T., Sterner O., Tetrahedron 2002 58 7785.
1Triplet observed due to overlap of the inner peaks of the two doublets expected from the two diastereotopic protons.
Генеральный спонсор и организатор – InterBioScreen Ltd. |
273 |


 NH
NH