
ЯМР спектороскопия
.pdf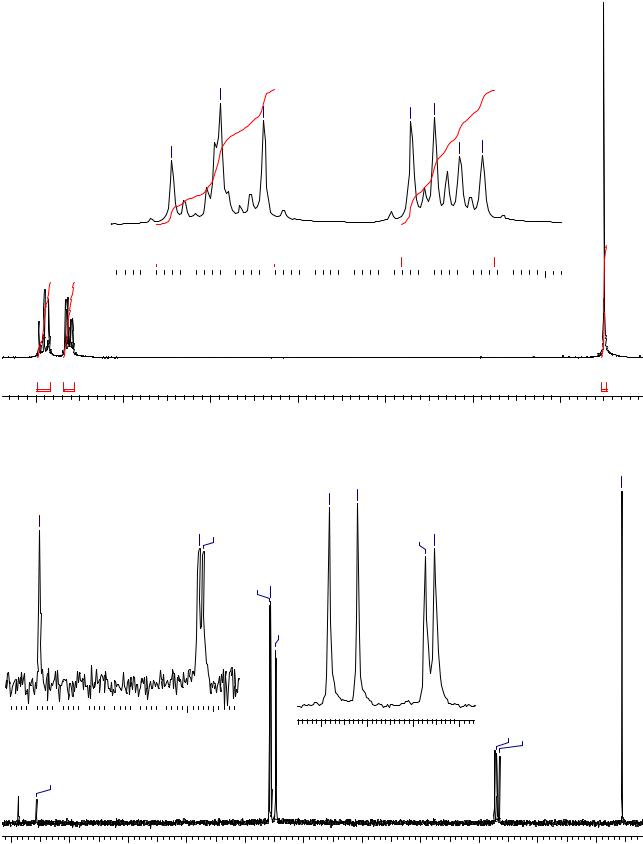
Задача 36.
Ниже приведены спектры соединения C7H7FO. Установите его структурную формулу и выполните полное отнесение сигналов в обоих спектрах.
2096.7 |
2087.5 |
2079.4 |
2051.4 |
2047.0 |
2042.2 |
2037.8 |
|
|
|
|
2.00 |
|
|
|
|
|
|
|
|
|
|
|
1.99 |
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7.00 |
6.95 |
6.90 |
6.85 |
|
6.80 |
|
6.75 |
|||||||||||||||||
2.001.99 |
|
|
|
|
|
|
|
2.99 |
7.0 |
6.5 |
|
6.0 |
5.5 |
5.0 |
|
4.5 |
4.0 |
158.86 |
|
155.78 |
155.70 |
115.91 |
115.60 |
114.88 |
114.78 |
55.72 |
|
|
|
115.91 |
115.60 |
|
|
|
|
|
|
|
|
114.78 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
156 |
||
|
|
|
159 |
158 |
157 |
|||||||||
158.86 |
155.78 |
155.70 |
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
116.0 |
115.5 |
115.0 |
114.5 |
77.48 |
77.06 |
76.62 |
|
|
|
||
|
|
|
|
160 |
150 |
140 |
130 |
120 |
110 |
100 |
90 |
80 |
70 |
60 |
190

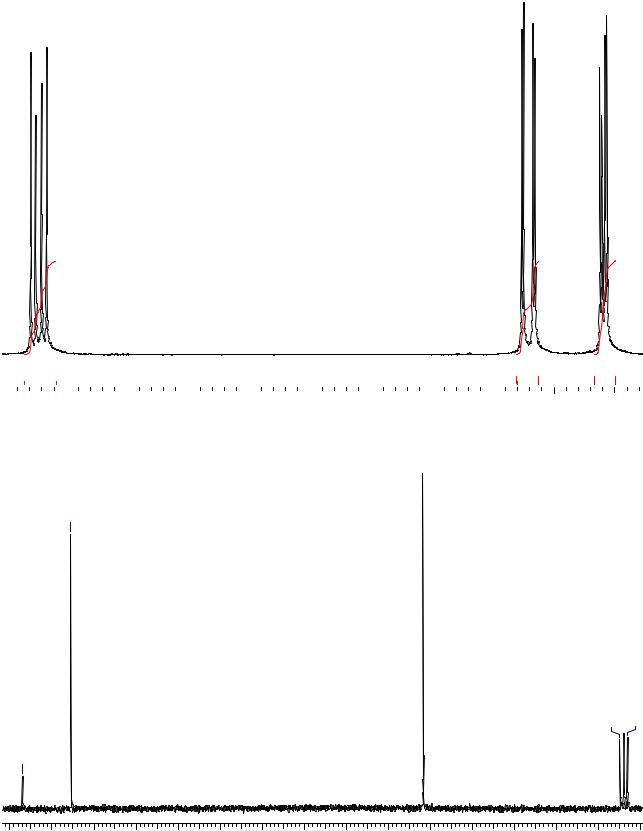
Задача 37.
Ниже представлены спектры 3-метилпентандиона-2,4. Объяснить картину, наблюдаемую в спектре 1Н ЯМР (для удобства анализа область сильного поля растянута).
3.70 |
3.68 |
3.72
|
|
|
|
|
|
1.00 |
|
|
|
|
|
|
|
|
3.70 |
3.65 |
|
5.96 |
4.82 |
|
|
2.41 |
|
|
|
|
2.2 |
2.1 |
2.0 |
1.9 |
1.8 |
1.7 |
1.6 |
1.5 |
1.4 |
0.74
16 |
15 |
14 |
13 |
12 |
11 |
10 |
9 |
8 |
7 |
6 |
5 |
1.34 1.32
 1.34 3.01
1.34 3.01
3.723.703.68
1.005.963.01
|
|
|
|
|
2 |
4 |
3 |
|
|||
|
|
28.73 |
23.42 |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
61.92 |
|
12.60 |
205.10 |
190.41 |
77.59 |
76.73 |
|
12.96 |
|
|
104.85 |
|
26.14 |
21.22 |
200 |
150 |
100 |
50 |
191

Задача 38.
Ниже приведены спектры соединений А и Б, имеющих формулу C9H9N. Установите их строение.
Соединение А:
1.00 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.13 |
1.10 |
|
|
|
|
0.99 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.01 |
||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7.6 |
|
7.5 |
7.4 |
7.3 |
|
7.2 |
7.1 |
|
7.0 |
6.9 |
6.8 |
6.7 |
6.6 |
6.5 |
|
|||||||||||||||||||||||||||||||||||||||
1.00 |
2.13 |
0.99 |
1.01 |
|
|
|
|
|
|
3.08 |
7.5 |
|
7.0 |
6.5 |
6.0 |
5.5 |
|
5.0 |
4.5 |
4.0 |
3.5 |
|
|
119.19 |
|
|
|
|
|
|
|
|
|
|
120.78 |
|
|
|
|
|
|
|
|
|
128.66 |
121.39 |
109.10 |
100.84 |
|
|
|
|
|
|
|
|
|
|
|
77.43 |
|
76.59 |
|
|
32.61 |
136.66 |
128.46 |
|
|
|
77.01 |
|
|
|
130 |
120 |
110 |
100 |
90 |
80 |
70 |
60 |
50 |
40 |
30 |
192

Соединение Б:
|
|
|
|
|
|
|
|
|
|
|
|
|
6.00 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.89 |
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3.8 |
3.7 |
3.6 |
3.5 |
|
3.4 |
3.3 |
3.2 |
3.1 |
3.0 |
2.9 |
2.8 |
2.7 |
2.6 |
2.5 |
2.4 |
2.3 |
|
2.2 |
2.1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
41.78 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
73.40 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
78.38
80 |
75 |
70 |
65 |
60 |
55 |
50 |
45 |
40 |
35 |
30 |
25 |
20 |
15 |
10 |
5 |
0 |
193

Задача 39.
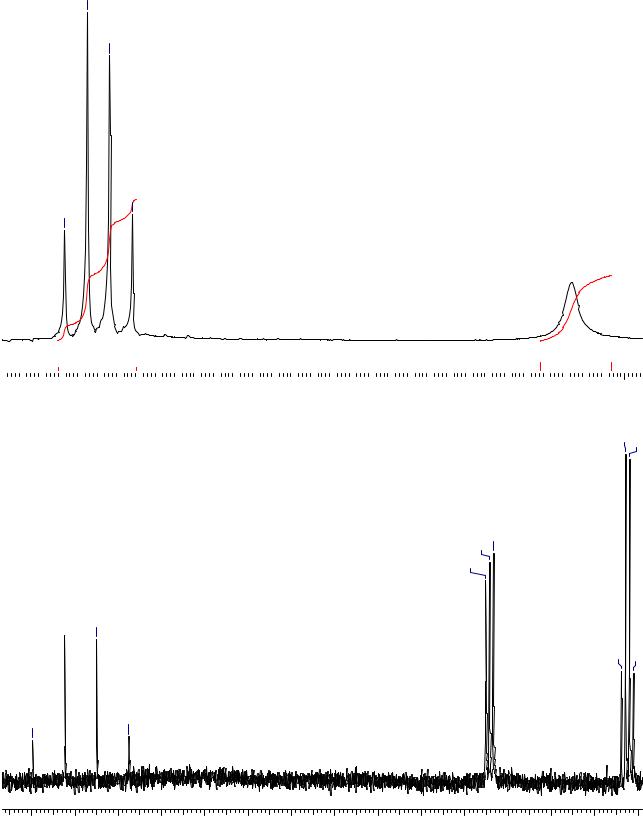
Ниже приведены спектры трех изомерных соединений А, Б и В состава C3H3ClO2. Установите их строение.
Соединение А:
7.48 |
6.28 |
7.53 |
6.24 |
|
1.00 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.07 |
|
|
|
|
|
|
|
|
1.09 |
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12.0 |
|
11.5 |
11.0 |
10.5 |
10.0 |
9.5 |
9.0 |
8.5 |
8.0 |
7.5 |
7.0 |
6.5 |
6.0 |
||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
124.25 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
140.64 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
169.87 |
77.50 |
170 |
160 |
150 |
140 |
130 |
120 |
110 |
100 |
90 |
80 |
194

Соединение Б:
6.85 6.24
6.88 |
6.27 |
0.94 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.00 |
|
|
|
|
1.01 |
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12.0 |
|
11.5 |
11.0 |
10.5 |
10.0 |
9.5 |
9.0 |
8.5 |
8.0 |
7.5 |
7.0 |
6.5 |
6.0 |
5.5 |
|||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
120.84 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
135.50 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
169.14 |
77.51 |
170 |
160 |
150 |
140 |
130 |
120 |
110 |
100 |
90 |
80 |
195
Соединение В:
1.00 |
|
|
|
|
|
|
1.00 |
1.00 |
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5.0 |
|
|
|
7.0 |
6.5 |
6.0 |
5.5 |
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
100.88 |
|
|
|
|
|
|
|
142.76 |
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|||||
77.48 |
77.06 |
76.64 |
|
|
|
||
|
|
|
|
148.54
150 |
145 |
140 |
135 |
130 |
125 |
120 |
115 |
110 |
105 |
100 |
95 |
90 |
85 |
80 |
75 |
196
Задача 40.
Ниже приведены спектры соединения C2H3F3O. Какова его структура? Объясните форму сигналов в спектрах 1Н и 13С ЯМР, определите величины КССВ (спектр регистрировали на приборе с рабочей частотой 300 МГц)
3.94
3.91
3.97 |
3.88 |
|
|
|
|
|
|
|
|
2.00 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0.93 |
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.05 |
|
4.00 |
|
3.95 |
|
|
3.90 |
|
3.85 |
3.80 |
3.75 |
3.70 |
3.65 |
3.60 |
3.55 |
3.50 |
3.45 |
3.40 |
3.35 |
3.30 |
3.25 |
|||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
61.50 |
61.03 |
||
77.59 |
77.15 |
76.73 |
126.20 |
122.51 |
|
|
|
|
61.97 |
60.56 |
129.89 |
118.82 |
130 |
125 |
120 |
115 |
110 |
105 |
100 |
95 |
90 |
85 |
80 |
75 |
70 |
65 |
60 |
197

|
|
|
Таблица 2. Характерные величины химических сдвигов протонов различных групп. |
|
|
|
||||||
Тип протона |
11 |
10 |
9 |
8 |
7 |
6 |
5 |
4 |
3 |
2 |
1 |
0 |
C CH3 |
|
|
|
|
|
|
|
|
|
|
|
|
C CH(2) |
|
|
|
|
|
|
|
|
|
|
|
|
C CH |
|
|
|
|
|
|
|
|
|
|
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
C CH |
|
|
|
|
|
|
|
|
|
|
|
|
C CH3 |
|
|
|
|
|
|
|
|
|
|
|
|
C CH(2) |
|
|
|
|
|
|
|
|
|
|
|
|
C CH(2,3) |
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
|
O CH(2,3) |
|
|
|
|
|
|
|
|
|
|
|
|
N CH(2,3) |
|
|
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
|
|
|
C CH(2,3) |
|
|
|
|
|
|
|
|
|
|
|
|
C O |
|
|
|
|
|
|
|
|
|
|
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
C O |
|
|
|
|
|
|
|
|
|
|
|
|
OH |
|
|
|
|
|
|
|
|
|
|
|
|
C OH |
|
|
|
|
Ф Е Н О Л Ы |
|
|
|
C П И Р Т Ы |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NH(2) |
|
|
|
|
А М И Д Ы |
|
|
|
А М И Н Ы |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
Таблица 3. Диапазоны химических сдвигов в спектрах 13С ЯМР некоторых группировок. (если заместитель не указан, подразумевается алкильная группа)
Тип атома С |
|
180 |
160 |
140 |
120 |
100 |
80 |
60 |
40 |
20 |
|
|||||||||||||||||||||
-CH3, -CH2-, - |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH< |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-CH= (алкены) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-CH= |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(ароматика) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-СH≡ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
=C-C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(аллильный) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
=C-C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(бензильный) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
=С= |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Hal-С |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
>N-C (амины) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-O-C (спирты) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-O-C (эфиры) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-S-C (тиолы) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-SO-C (оксиды) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-SO2-C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(сульфоны) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-C(=O)-C |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-CHO |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(альдегиды) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
>C=O (кетоны) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-COOH |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-COOR |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-COONR2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-C≡N |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Фураны |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Пирролы |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Тиофены |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Пиридины |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Б
