
- •Course of lectures «Contemporary Physics: Part2»
- •Interactions Involving Neutrons
- •Interactions Involving Neutrons
- •Nuclear Fission
- •Nuclear Fission
- •Nuclear Fission
- •Nuclear Reactors
- •Nuclear Reactors
- •Nuclear Reactors
- •Nuclear Reactors
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Nuclear Fusion
- •Radiation Detectors
- •Radiation Detectors
- •Radiation Detectors
- •Radiation Detectors
- •Uses of Radiation
- •Uses of Radiation
- •Uses of Radiation
- •Uses of Radiation

Course of lectures «Contemporary Physics: Part2»
Lecture №14
Applications of Nuclear Physics.
Interactions Involving Neutrons.
Nuclear Fission. Nuclear Reactors.
Nuclear Fusion. Radiation
Damage. Radiation Detectors.
Uses of Radiation.

Interactions Involving Neutrons
To understand nuclear fission and the physics of nuclear reactors, we must first understand how neutrons interact with nuclei. Because of their charge neutrality, neutrons are not subject to Coulomb forces and as a result do not interact electrically with electrons or the nucleus. Therefore, neutrons can easily penetrate deep into an atom and collide with the nucleus. A fast neutron (energy greater than approximately 1 MeV) traveling through matter undergoes many collisions with nuclei, giving up some of its kinetic energy in each collision. For fast neutrons in some materials, elastic collisions dominate. Materials for which that occurs are called moderators because they slow down (or moderate) the originally energetic neutrons very effectively. Moderator nuclei should be of low mass so that a large amount of kinetic energy is transferred to them in elastic collisions. For this reason, materials that are abundant in hydrogen, such as paraffin and water, are good moderators for neutrons.
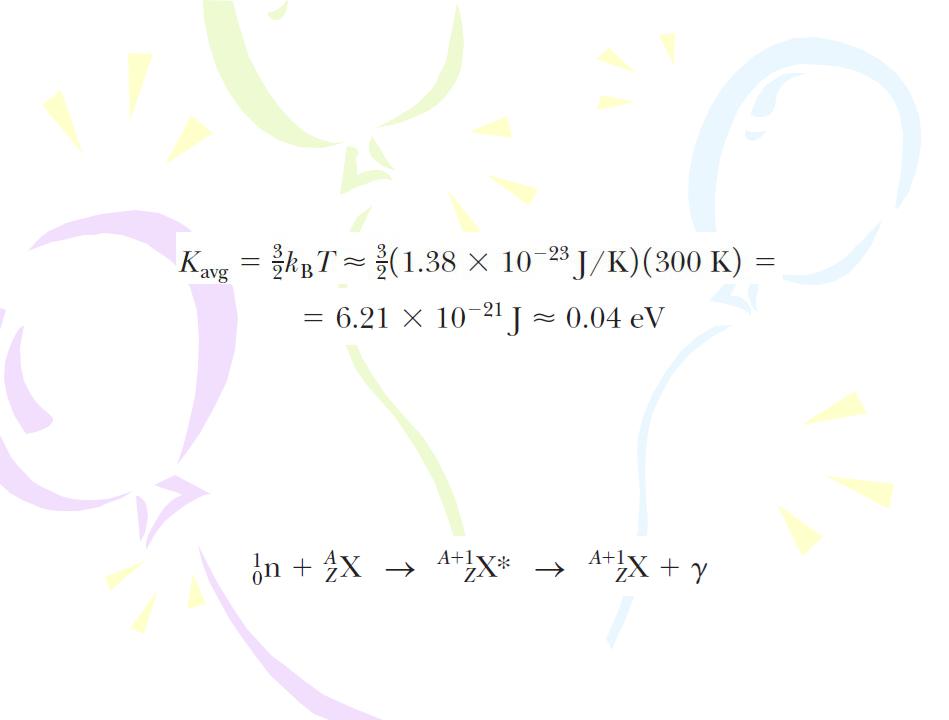
Interactions Involving Neutrons
Eventually, most neutrons bombarding a moderator become thermal neutrons, which means they have given up so much of their energy that they are in thermal equilibrium with the moderator material. Their average kinetic energy at room temperature is
Once the neutrons have thermalized and the energy of a particular neutron is sufficiently low, there is a high probability the neutron will be captured by a nucleus, an event that is accompanied by the emission of a gamma ray. This neutron capture reaction can be written
The neutron-capture rate for neutrons passing through any sample depends on the type of atoms in the sample and on the energy of the incident neutrons.

Nuclear Fission
A nuclear fission occurs when a heavy nucleus, such as 235U, splits into two smaller nuclei. 
The absorption of the neutron creates a nucleus that is unstable and can change to a lower-energy configuration by splitting into two smaller nuclei. In such a reaction, the combined mass of the daughter nuclei is less than the mass of the parent nucleus, and the difference in mass is called the mass defect.
where 236U* is an intermediate excited state that lasts for approximately 10-12 s before splitting into medium-mass nuclei X and Y, which are called fission fragments.
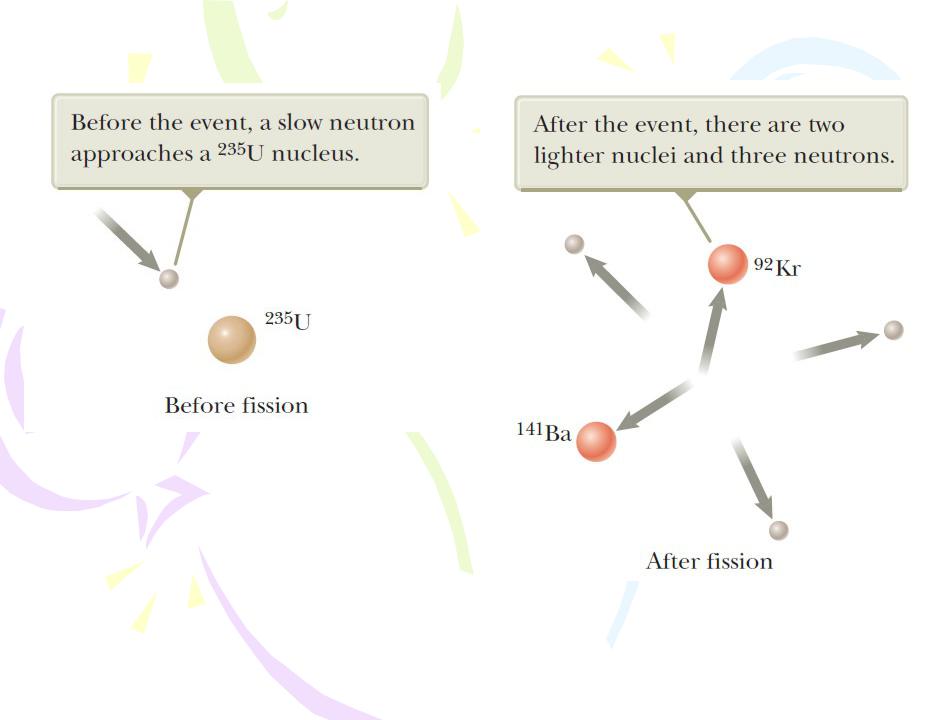
Nuclear Fission
A nuclear fission event
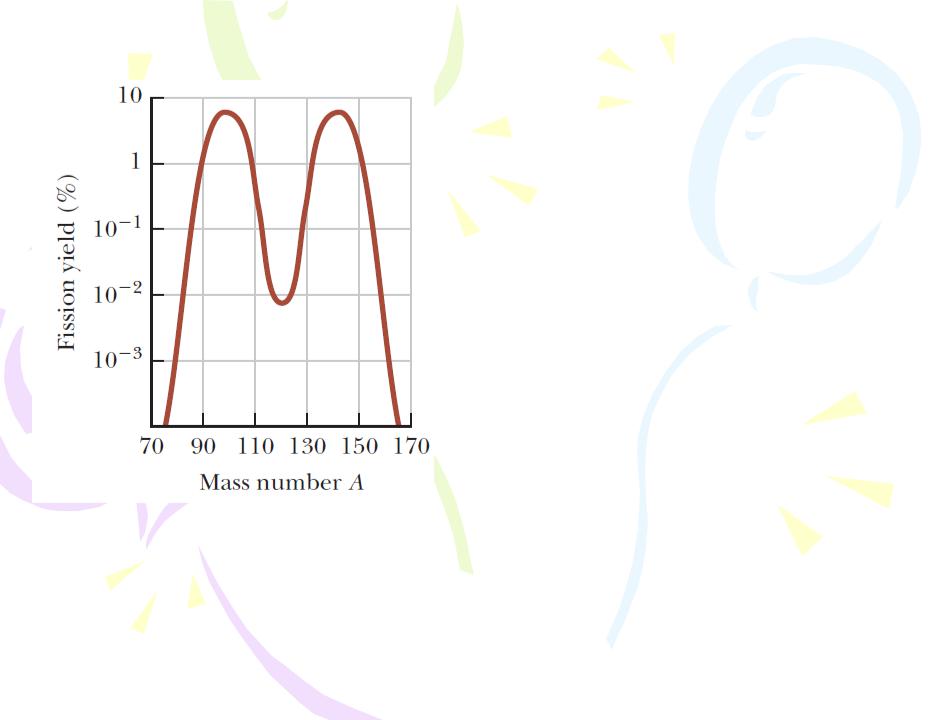
Nuclear Fission
Distribution of fission products versus mass number for the fission of 235U bombarded with thermal neutrons. Notice that the vertical axis is logarithmic. 
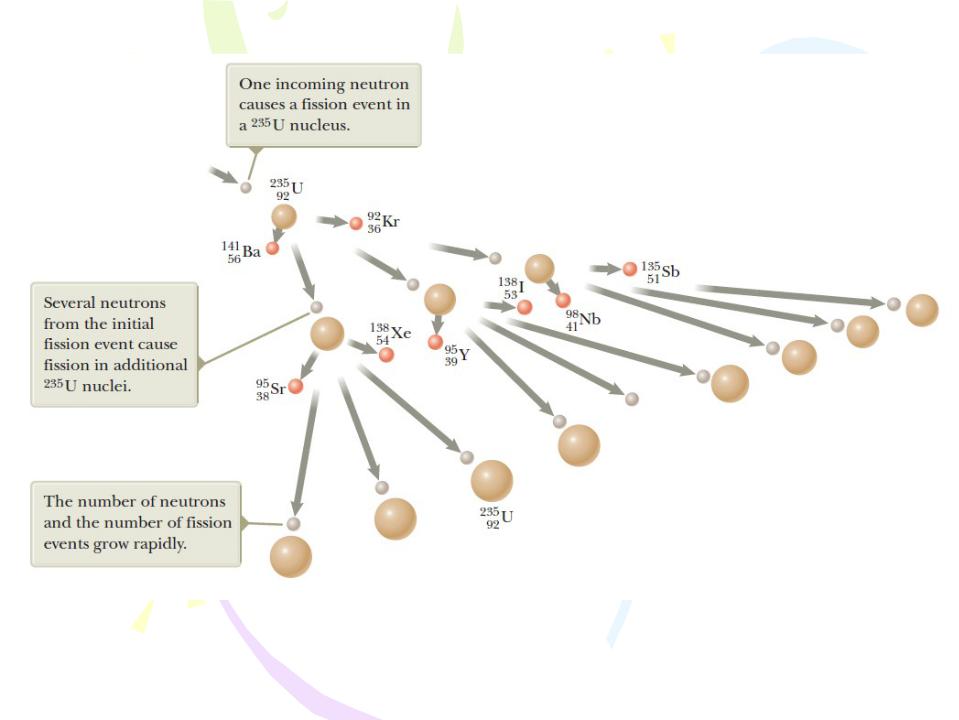
Nuclear Reactors
A nuclear chain reaction initiated by the capture of a neutron. Uranium nuclei are shown in tan, neutrons in gray, and daughter nuclei in orange.

Nuclear Reactors
Main components of a pressurized- water nuclear reactor. 
Nuclear Reactors
Control of Power Level
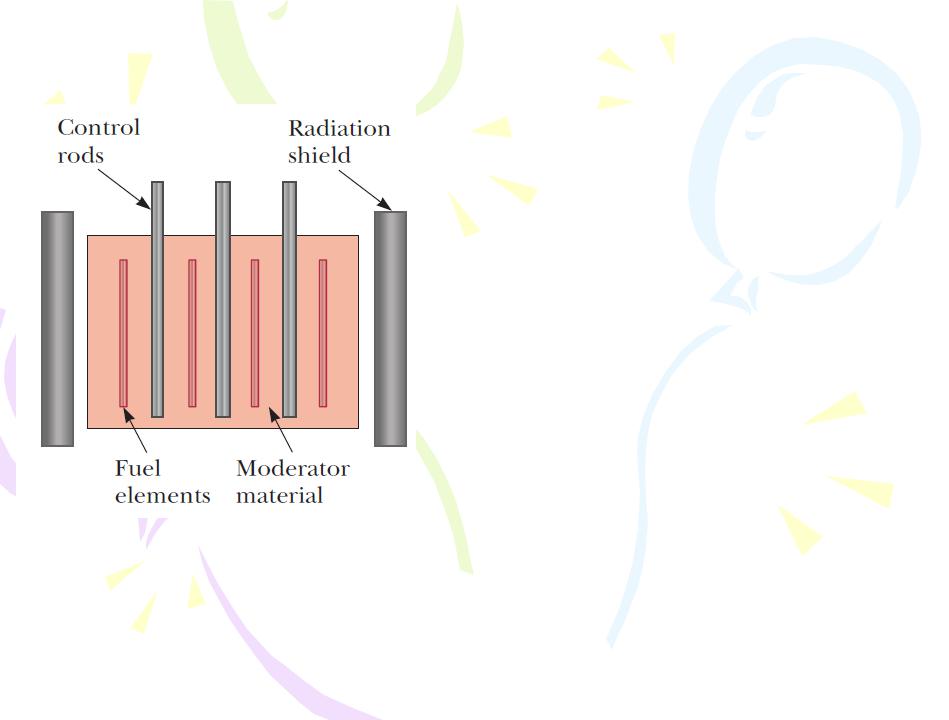
The basic design of a nuclear reactor core
Cross section of a reactor core showing the control rods, fuel elements containing enriched fuel, and moderating material, all surrounded by a radiation shield.
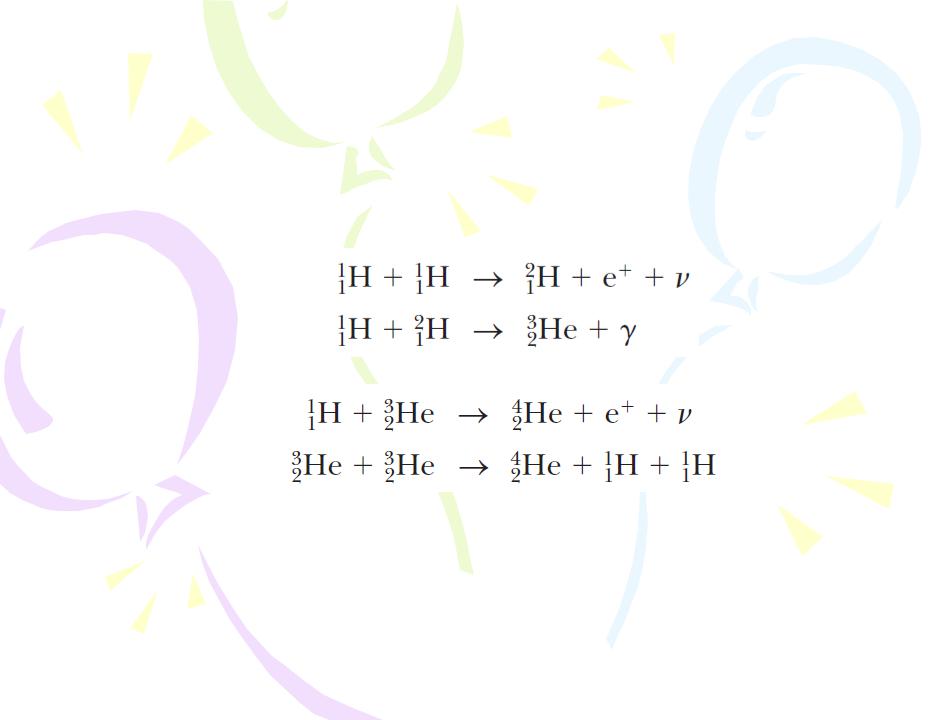
Nuclear Reactors
Safety and Waste Disposal
Two examples of energy-liberating fusion reactions are as follows:
These fusion reactions are the basic reactions in the proton–proton cycle, believed to be one of the basic cycles by which energy is generated in the Sun and other stars that contain an abundance of hydrogen.
