
clavien_atlas_of_upper_gastrointestinal_and_hepato-pancreato-biliary_surgery2007-10-01_3540200045_springer
.pdf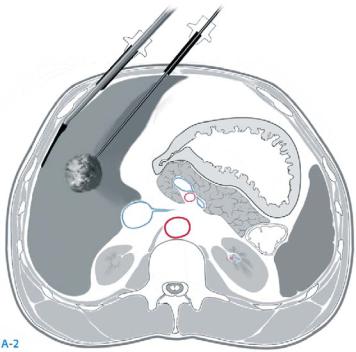
414 |
SECTION 3 |
Liver |
|
|
|
STEP 2 |
Assessment of tumors and ablation |
|
|
|
|
Laparoscopic intra-abdominal ultrasound (IOUS) is performed by either rigid or flexible IOUS probes to identify/confirm the location and the size of the lesions. The abdomen is explored for the presence of extrahepatic disease. As described for the open technique, the RFA probe is oriented parallel with the IOUS crystal to facilitate probe guidance (A-2). Limited mobility can make this more difficult than
with the open technique. The ablation process and monitoring is otherwise performed as described with the open approach.

Radiofrequency Ablation of Liver Tumors |
415 |
|
|
Postoperative Testing
Follow-up postablation imaging (CT or MRI) is performed 3–7days after procedure to assess completeness of ablation.
Postoperative Complications
■Short term:
–Pleural effusion
–Regional hemorrhage into needle track or into RFA lesion
–Fever
–Hepatic abscess (more common with enterobiliary anastomosis)
–Biliary stricture secondary to ablation near major bile duct
–Grounding pad burns
■Long term:
–Biloma
–Biliary fistula
–Ascites
–Hepatic insufficiency
–Arteriovenous fistula
Tricks of the Senior Surgeon
■When the lesion lies near a major blood vessel, thermal energy from the probe may be drawn away from the ablation zone, limiting ablation efficacy. This is known as the “heat-sink” effect. This can be limited using in-flow occlusion techniques (e.g., Pringle maneuver) or repositioning the array closer to the vascular structure.
■Stabilization of the radiofrequency probe at the skin or liver surface should be done during deployment to avoid “push back.”
■Depending on the device, monitoring of the impedance pattern and tine deployment shape by ultrasound can confirm success of the ablation in real time.
■With some devices, temperature profiles can also be helpful at confirming successful ablation, including consistent tine temperatures and adequately slow cool-down temperatures.

Selective Hepatic Intra-arterial Chemotherapy
Christopher D. Anderson, Ravi S. Chari
Hepatic intra-arterial infusion pump (HAIP) placement provides hepatic specific continuous infusion of chemotherapeutic agents. The chemotherapeutic agents selected for use with HAIP should exhibit a high degree of first pass kinetics in order to minimize systemic toxicity. Agents used include cisplatin, fluodeoxyuridine (FUDR), mitomycin C, and Adriamycin.
Indications and Contraindications
Indications |
■ |
Unresectable hepatic metastatic colorectal carcinoma |
|
■ |
Liver specific adjuvant chemotherapy following resection of colorectal metastases |
|
■ |
Use in hepatocellular carcinoma and other metastatic carcinomas can be considered |
|
|
under certain circumstances |
|
|
Portal hypertension (portal pressure >12 mm Hg) |
Contraindications |
■ |
|
|
■ |
Known extrahepatic malignancy |
|
■ |
Poor liver reserve (e.g., Child B or C cirrhosis) |
|
■ |
Severe coagulopathy (e.g. platelets <30000 ml) |
|
■ |
Active hepatitis |
Preoperative Studies
History: |
Specific for hepatic function (e.g., alcohol use, hepatitis) |
Clinical evaluation: |
To rule out obvious extrahepatic malignancy (lymph node exam) |
|
and underlying liver dysfunction (ascites, nutrition status, signs |
|
of portal hypertension) |
Triple phase CT, |
For definition of tumor and arterial anatomy |
CT angiogram (CTA) |
|
or MRI of abdomen: |
|
Chest CT: |
To rule out pulmonary metastases, except in the case of |
|
colorectal carcinoma |
FDG-PET: |
In cases of metastatic colorectal carcinoma |
Hepatic arteriogram: Not mandatory if HAIP is to be placed via laparotomy, but it may decrease operative time
Mandatory if HAIP to be placed laparoscopically, and CTA or MRA not performed
418 |
SECTION 3 |
Liver |
|
|
|
|
Procedure |
|
|
|
|
STEP 1 |
Incision and exposure |
|
|
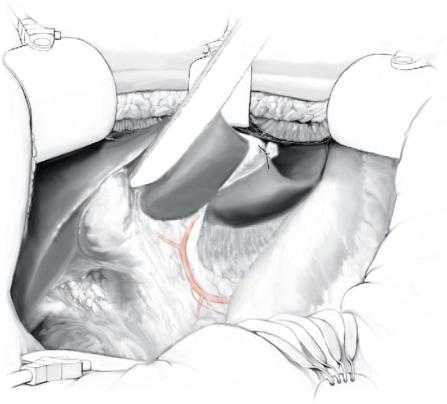
A right subcostal approach is used. The falciform ligament is divided and the porta |
|
|
|
|
|
hepatis is exposed further by gentle superior retraction of the liver. Cholecystectomy is |
|
|
performed to eliminate the postoperative complication of chemical cholecystitis induced |
|
|
by infusional chemotherapy. The gastrohepatic ligament is divided with care to avoid |
|
|
injury to a replaced left hepatic artery (if present). All duodenal and antral vessels must |
|
be ligated to eliminate the possibility of reflux of chemotherapy into these regions as this may lead to chemical duodenitis or gastritis. The common hepatic, gastroduodenal, and proper hepatic arteries are identified by dissection of the hepatoduodenal ligament and marked with vessel loops.
Selective Hepatic Intra-arterial Chemotherapy |
419 |
|
|
|
|
STEP 2 |
Determination of arterial anatomy |
|
|
|
|
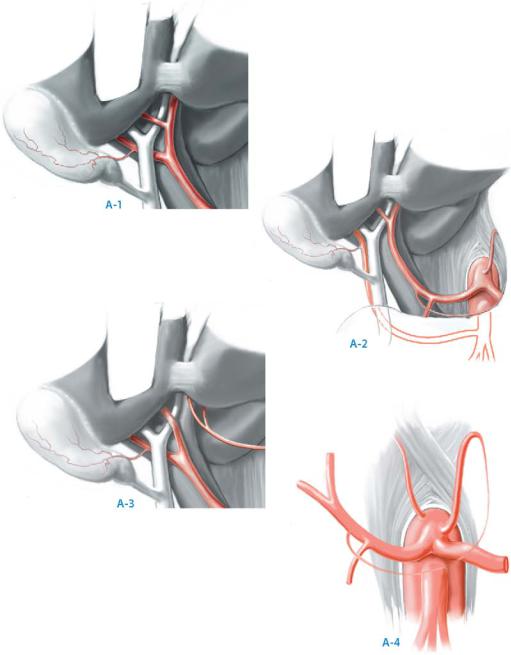
At this point it is imperative to accurately determine the hepatic arterial anatomy if no preoperative arteriogram was performed. The more common variants in hepatic arterial anatomy are shown in the figure: (A-1) the common hepatic artery may originate from the celiac trunk (typical). (A-2) A replaced right hepatic artery (RRHA) may arise directly from the superior mesenteric artery. (A-3) A replaced left hepatic artery (RLHA) may arise directly from the left gastric artery. (A-4) Common origin of the right hepatic artery, left hepatic artery and gastroduodenal artery. This is commonly referred to as the “trifurcation anatomy.”
420 |
SECTION 3 |
Liver |
|
|
|
STEP 3 |
Placement of the infusion pump in subcutaneous pocket |
|
|
|
|
The pump pocket is created in the right lower quadrant by making a transverse incision at the level of the umbilicus and dissecting to the anterior rectus sheath and the external oblique fascia. The pocket is extended laterally to near the iliac crest and inferiorly to just above the inguinal ligament. Some surgeons prefer that the pocket be developed through the initial subcostal incision, while others prefer it not to communicate with the laparotomy incision. The pump is primed with heparinized saline and proper pump function should be established before anchoring it to the fascia with 2-0 nonabsorbable braided suture. The arterial catheter is passed into the pocket by direct puncture.
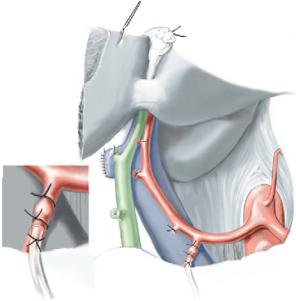
Selective Hepatic Intra-arterial Chemotherapy |
421 |
|
|
|
|
STEP 4 |
Placement of the arterial catheter |
|
|
|
|
Cannulation typically should be performed with a single catheter placed in the gastroduodenal artery (GDA). The origin of the GDA from the common hepatic artery is identified, and the proper hepatic artery from the GDA to the liver is skeletonized. Any branches leading to intestinal viscera are individually ligated and divided. This step prevents visceral misperfusion injuries. The GDA is further skeletonized for approximately 2cm distal to its origin and then ligated near the pancreas. A noncrushing vascular clamp is used to occlude the GDA at its origin. A transverse arteriotomy is made in the GDA approximately 1.5cm from its origin. The beaded infusion catheter is cut such that its port does not enter the common hepatic artery, but that at least one bead is in the GDA. The catheter tip should not enter the common hepatic artery to lessen the risk of catheter associated common hepatic artery thrombosis. The GDA is secured around the catheter using 4-0 nonabsorbable suture both proximally and distally to the catheter bead. This prevents advancement or retraction of the catheter.
To insure proper hepatic perfusion, 1–5cc of fluorescence is bolused into the catheter, and the liver is observed under a Woods lamp. Alternatively, 1–5cc of methylene blue will also visually confirm adequate perfusion; this is a useful method when placing HAIP laparoscopically.
422 |
SECTION 3 |
Liver |
|
|
|
STEP 5 |
Dealing with atypical arterial anatomy |
|
|
|
|
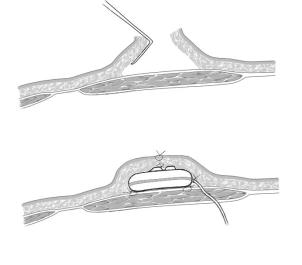
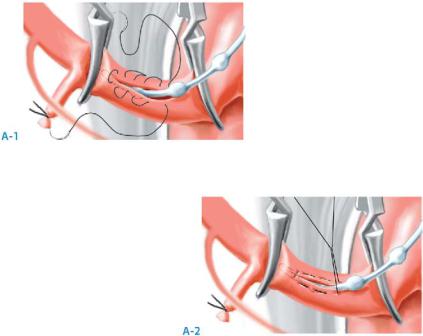
Following a hepatic resection, management of aberrant arterial anatomy is contingent upon the arterial supply of the remnant hepatic parenchyma. However, without a liver resection, the most common arterial variant encountered is the trifurcation anatomy. This may be approached by ligation of the GDA and directly cannulating the common hepatic artery well proximal to the bifurcation of the right and left hepatic arteries. The common hepatic artery is skeletonized and occluded proximally and distally with noncrushing vascular clamps. A longitudinal arteriotomy is performed and the catheter is inserted with the bead remaining outside the arteriotomy (A-1). A pursestring suture (6-0 nonabsorbable) is used to close the arteriotomy around the catheter and secure the catheter in place (A-2).
Replaced hepatic arteries may undergo isolation and clamping. If clamping is tolerated, the replaced vessel may be ligated and the liver can be perfused via the typical catheter placement. If clamping is not tolerated, a replaced right hepatic artery may be approached by placing one catheter in the GDA to perfuse the left hepatic and a second catheter directly into the replaced right hepatic artery as described for the trifurcation anatomy. A similar approach may be used for a replaced left hepatic artery. If two catheters are placed, it is important not to connect them to a single pump via a Y connector because of differential resistance in each vessel.

Selective Hepatic Intra-arterial Chemotherapy |
423 |
|
|
Postoperative Studies
Some centers routinely perform a nuclear medicine study of the HAIP on postoperative day3–5 to rule out extrahepatic perfusion. This consists of a sulfur colloid outline of the liver and then bolus injection of technetium micro-albumin aggregate (MAA)
via the pump bolus port. The two images are superimposed to insure adequate hepatic perfusion and to rule out extrahepatic visceral perfusion.
Complications
■Early:
–Visceral misperfusion
–Arterial injury and postoperative bleeding
–Hematoma or seroma of the pump pocket
■Late:
–Biliary stricture
–Hepatic artery thrombosis
–Occlusion or displacement of the catheter
–Pseudoaneurysm
–Pump pocket infection
Tricks of the Senior Surgeon
■Prime the pump and test its function early in the case. Be familiar with the specifics of the various pumps and the needles used to bolus/prime and fill the pump.
■When accessory vessels are known to exist or are discovered at the time of laparotomy, determination of their contribution to hepatic perfusion can be gauged by clamping and observing the parenchyma for color change: this should be performed early in the case so there is adequate time to determine the consequences of ligation versus the need for a second pump system.
■A postoperative MAA perfusion study is not essential and may be omitted if clear intraoperative studies indicate uniform hepatic perfusion with lack of reflux into the duodenum or stomach. In cases where accessory vessels are ligated, and a single catheter and pump are placed, an MAA study 1month after placement should be performed to demonstrate uniform perfusion
of the hepatic parenchyma.

Unroofing and Resection
for Benign Non-Parasitic Liver Cysts
Juan M. Sarmiento, David M. Nagorney
Hepatic cysts are classified according to the presence or absence of a parasitic etiology. They seldom lead to hepatic dysfunction and are mostly asymptomatic. The treatment is always individualized according to the origin and presence of symptoms. The choice between unroofing versus resection is dictated by site, number of cysts, malignant potential (cystadenoma/cystadenocarcinoma), and parasitic infection (see next chapter). Malignant potential is rare and is not a primary concern.
Indications and Contraindications
Indications |
■ |
Pain significantly affecting lifestyle |
|
■ |
Jaundice |
|
■ |
Infection |
|
■ |
Hemorrhage |
|
■ |
Portal hypertension |
|
■ |
Abdominal fullness or mass |
|
|
Asymptomatic patients |
Contraindications |
■ |
|
|
■ |
Patients amenable to percutaneous cyst aspiration and/or alcohol sclerosis under US |
|
|
or CT guidance (simple cysts lacking or with minimal capsular extension or cysts in |
|
|
segments 7 and 8) |
Preoperative Investigation and Preparation for Procedure
History: |
Polycystic disease of the kidney |
Clinical evaluation: |
Abdominal pain, jaundice, signs of portal hypertension |
Laboratory tests: |
AST, ALT, alkaline phosphatase, bilirubin, tumor markers |
|
(CEA, CA 19–9) and serologies (hydatid) |
CT scan: |
Location, accessibility to laparoscopic versus open approach, |
|
amount of remaining healthy parenchyma |
US: |
Location, compression of main vessels (especially hepatic veins), |
|
biliary dilatation |
MRI: |
Polycystic liver disease or complicated cases |
