
- •Preface
- •Contents
- •Procedures, Assays, and Normal Values
- •Normal Cells of the Blood and Hematopoietic Organs
- •The Individual Cells of Hematopoiesis
- •Bone Marrow: Cell Composition and Principles of Analysis
- •Abnormalities of the White Cell Series
- •Predominance of Mononuclear Round to Oval Cells
- •Prevalence of Polynuclear (Segmented) Cells
- •Erythrocyte and Thrombocyte Abnormalities
- •Hypochromic Anemias
- •Normochromic Anemias
- •Hyperchromic Anemias
- •Erythrocyte Inclusions
- •Thrombocyte Abnormalities
- •Cytology of Organ Biopsies and Exudates
- •Lymph Node Cytology
- •Branchial Cysts and Bronchoalveolar Lavage
- •Cytology of Pleural Effusions and Ascites
- •Cytology of Cerebrospinal Fluid
- •Introduction to the Physiology and Pathophysiology of the Hematopoietic System
- •Cell Systems
- •Principles of Regulation and Dysregulation in the Blood Cell Series and their Diagnostic Implications
- •Procedures, Assays, and Normal Values
- •Taking Blood Samples
- •Erythrocyte Count
- •Hemoglobin and Hematocrit Assay
- •Calculation of Erythrocyte Parameters
- •Red Cell Distribution Width (RDW)
- •Reticulocyte Count
- •Leukocyte Count
- •Thrombocyte Count
- •Significance of the Automated Blood Count
- •Bone Marrow Biopsy
- •Lymph Node Biopsy and Tumor Biopsy
- •Step-by-Step Diagnostic Sequence
- •The Individual Cells of Hematopoiesis
- •Eosinophilic Granulocytes (Eosinophils)
- •Basophilic Granulocytes (Basophils)
- •Monocytes
- •Lymphocytes (and Plasma Cells)
- •Megakaryocytes and Thrombocytes
- •Bone Marrow: Medullary Stroma Cells
- •Abnormalities of the White Cell Series
- •Predominance of Mononuclear Round to Oval Cells
- •Reactive Lymphocytosis
- •Relative Lymphocytosis Associated with Granulocytopenia (Neutropenia) and Agranulocytosis
- •Monocytosis
- •Acute Leukemias
- •Neutrophilia without Left Shift
- •Reactive Left Shift
- •Osteomyelosclerosis
- •Elevated Eosinophil and Basophil Counts
- •Clinically Relevant Classification Principle for Anemias: Mean Erythrocyte Hemoglobin Content (MCH)
- •Hypochromic Anemias
- •Iron Deficiency Anemia
- •Hypochromic Infectious or Toxic Anemia (Secondary Anemia)
- •Hypochromic Anemia with Hemolysis
- •Normochromic Anemias
- •Normochromic Hemolytic Anemias
- •Cytomorphological Anemias with Erythrocyte Anomalies
- •Bone Marrow Aplasia
- •Hyperchromic Anemias
- •Erythrocyte Inclusions
- •Hematological Diagnosis of Malaria
- •Thrombocyte Abnormalities
- •Thrombocytopenia
- •Lymph Node Cytology
- •Sarcoidosis and Tuberculosis
- •Non-Hodgkin Lymphoma
- •Metastases of Solid Tumors in Lymph Nodes or Subcutaneous Tissue
- •Branchial Cysts
- •Cytology of Pleural Effusions and Ascites
- •Cytology of Cerebrospinal Fluid
- •References
- •Index

44 Normal Cells of the Blood and Hematopoietic Organs
Eosinophilic Granulocytes (Eosinophils)
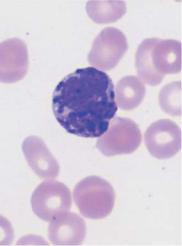
Eosinophils arise from the same stem cell population as neutrophils and mature in parallel with them. The earliest point at which eosinophils can be morphologically defined in the bone marrow is at the promyelocyte stage. Promyelocytes contain large granules that stain blue–red; not until they reach the metamyelocyte stage do these become a dense population of increasingly round, golden-red granules filling the cytoplasm. The Charcot–Leyden crystals found between groups of eosinophils in exudates and secretions have the same chemical composition as the eosinophil granules.
The nuclei of mature eosinophils usually have only two segments.
Diagnostic Implications. In line with their function (see p. 5) (reaction against parasites and regulation of the immune response, especially defense against foreign proteins), an increase of eosinophils above 400/µl should be seen as indicating the presence of parasitosis, allergies, and many other conditions (p. 124).
Basophilic Granulocytes (Basophils)
Like eosinophils, basophils (basophilic granulocytes) mature in parallel with cells of the neutrophil lineage. The earliest stage at which they can be identified is the promyelocyte stage, at which large, black–violet stained granules are visible. In mature basophils, which are relatively small, these granules often overlie the two compact nuclear segments like blackberries. However, they easily dissolve in water, leaving behind faintly pink stained vacuoles.
Close relations of basophilic granulocytes are tissue basophils or tissue mast cells—but these are never found in blood. Tissue basophils have a round nucleus underneath large basophilic granules.
Diagnostic Implications. In line with their role in anaphylactic reactions (p. 5), elevated basophil counts are seen above all in hypersensitivity reactions of various kinds. Basophils are also increased in chronic myeloproliferative bone marrow diseases, especially chronic myeloid leukemia (pp. 117, 120).
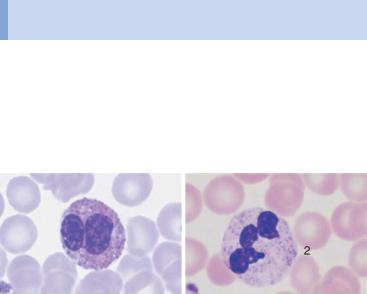
Round granules filling the cytoplasm: eosinophilic and basophilic granulocytes
a 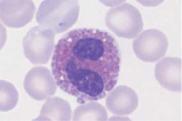
 b
b
c |
d |
e 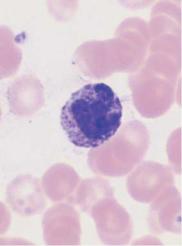
 f
f
Fig. 14 Eosinophilic and basophilic granulocytes. a–c Eosinophilic granulocytes with corpuscular, orange-stained granules. d In contrast, the granules of neutrophilic granulocytes are not round but more bud-shaped. e Basophilic granulocyte. The granules are corpuscular like those of the eosinophilic granulocyte but stain deep blue to violet. f Very prominent large granules in a basophilic granulocyte in chronic myeloproliferative disease.
45

46 Normal Cells of the Blood and Hematopoietic Organs
Monocytes
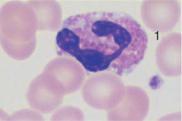
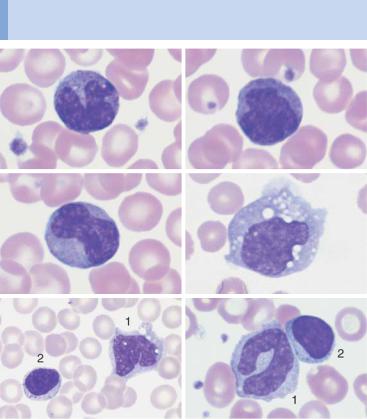
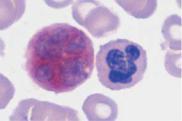
Monocytes are produced in the bone marrow; their line of development branches off at a very early stage from that of the granulocytic series (see flow chart p. 3), but does not contain any distinct, specific precursors that can be securely identified with diagnostic significance in everyday morphological studies. Owing to their great motility and adhesiveness, mature monocytes are morphologically probably the most diversified of all cells. Measuring anywhere between 20 and 40 µm in size, their constant characteristic is an ovoid nucleus, usually irregular in outline, with invaginations and often pseudopodia-like cytoplasmic processes. The fine, “busy” structure of their nuclear chromatin allows them to be distinguished from myelocytes, whose chromatin has a patchy, streaky structure, and also from lymphocytes, which have dense, homogeneous nuclei. The basophilic cytoplasmic layer varies in width, stains a grayish color, and contains a scattered population of very fine reddish granules that are at the very limit of the eye’s resolution. These characteristics vary greatly with the size of the monocyte, which in turn is dependent on the thickness of the smear. Where the smear is thin, especially at the feathered end, monocytes are abundant, relatively large and loosely structured, and their cytoplasm stains light gray–blue (“dove gray”). In thick, dense parts of the smear, some monocytes look more like lymphocytes: only a certain nuclear indentation and the “thundercloud” gray–blue staining of the cytoplasm may still mark them out.
Diagnostic Implications. In line with their function (see p. 5) as phagocytic defense cells, an elevation of the monocyte population above 7% and above 850/µl indicates an immune defense reaction; only when a sharp rise in monocyte counts is accompanied by a drop in absolute counts in the other cell series is monocytic leukemia suggested (p. 101). Phagocytosis of erythrocytes and white blood cells (hemophagocytosis) may occur in some virus infections and autoimmune diseases.
Monocytosis in cases of infection: always present at the end of acute infections; chronic especially in
–Endocarditis lenta, listeriosis, brucellosis, tuberculosis
Monocytosis in cases of a non-infectious response, e.g.,
–Collagenosis, Crohn disease, ulcerative colitis
Monocytoses in/as neoplasia, e.g.,
–Paraneoplastic in cases of disseminating tumors, bronchial carcinoma, breast carcinoma, Hodgkin disease, myelodysplasias (especially CMML, pp. 107f) and acute monocytic leukemia (p. 101)
Monocytes show the greatest morphological variation among blood cells
a |
b |
c |
d |
e |
f |
g 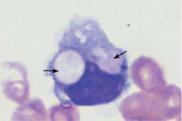
 h
h
Fig. 15 Monocytes. a–c Range of appearances of typical monocytes with lobed, nucleus, gray–blue stained cytoplasm and fine granulation. d Phagocytic monocyte with plasma vacuoles. e Monocyte (1) to the right of a lymphocyte with azurophilic granules (2). f Monocyte (1) with nucleus resembling that of a band neutrophil, but its cytoplasm stains typically gray–blue. Lymphocyte (2). g A monocyte that has phagocytosed two erythrocytes and harbors them in its wide cytoplasm (arrows) (sample taken after bone marrow transplantation). h Esterase staining, a typical marker enzyme for cells of the monocyte lineage.
47
