
- •Bioorganic chemistry
- •It is recommended to printing by
- •Contents
- •Introduction
- •1. Biologically important organic compounds classification and nomenclature
- •2. Organic compound structure
- •3. General laws of organic compound reaction ability
- •4. Amino acids, peptides and proteins
- •5. Carbohydrates: mono-, di- and poly-saccharides
- •Вопрос 74. What compounds are received at d-glucose and d-fructose reduction?
- •6. Nucleotides and nucleic acids
- •7. Lipids and low-molecular bioregulators
- •8. Organic compound identification
Вопрос 74. What compounds are received at d-glucose and d-fructose reduction?
A nswer.
At mono-saccharide reduction poly-alcohols are received (polyols)
which are called alditols.
Reduction is usually performed by hydrogen and there are metal
catalysts (palladium, nickel) or sodium bor-hydride. Hydrogen is
added on the place of the double bond between carbon-oxygen at
carbonyl group. At aldose reduction only one product is received
(polyol), for example, at D-glucose reduction six-atom alcohol is
received which is D-glucitol (L-sorbitol):
nswer.
At mono-saccharide reduction poly-alcohols are received (polyols)
which are called alditols.
Reduction is usually performed by hydrogen and there are metal
catalysts (palladium, nickel) or sodium bor-hydride. Hydrogen is
added on the place of the double bond between carbon-oxygen at
carbonyl group. At aldose reduction only one product is received
(polyol), for example, at D-glucose reduction six-atom alcohol is
received which is D-glucitol (L-sorbitol):
D-glucitol as well as D-glucose is optically active compound as in its molecule there are four asymmetric carbon atoms (2, 3, 4, 5) and there are no symmetry elements.
G lucose
reduction into sorbitol is the first stage of ascorbic acid chemical
synthesis. Sorbitol is oxidized using micro-organism Acetobacter
suboxydans; L-sorbose which is formed is transformed into ascorbic
acid at several stages.
lucose
reduction into sorbitol is the first stage of ascorbic acid chemical
synthesis. Sorbitol is oxidized using micro-organism Acetobacter
suboxydans; L-sorbose which is formed is transformed into ascorbic
acid at several stages.
At ketose reduction there is mixture containing two polyols, as carbon atom which is in the keto-group structure, after reduction turns into asymmetric and double localization of OH group and hydrogen atom is possible in the space (on the left or on the right in relation to carbon chain). For example, for D-fructose we have the following case:

D-glucitol and D-mannitol are optically active.
Poly-alcohols which are received at mono-saccharide reduction are crystal substances, well-soluble in water; they have sweet taste and could be used as sugar substitutes at diabetes mellitus (xylitol, sorbitol).
Question 75. At what aldose reduction optically inactive six-atom alcohols are formed?
A nswer.
At D-galactose and D-allose reduction we receive six-atom alcohols
dulcitol and allitol respectively. These alcohols
have symmetry plane which is between the third and fourth carbon
atoms, thus, these polyols do not have optical activity.
nswer.
At D-galactose and D-allose reduction we receive six-atom alcohols
dulcitol and allitol respectively. These alcohols
have symmetry plane which is between the third and fourth carbon
atoms, thus, these polyols do not have optical activity.
Question 76. What compounds are formed at L-galactose oxidation? Are they optically active?
Answer. Oxidation reaction is used at biochemical analysis (for example, blood count and urinalysis on sugar). Mono-saccharide oxidation product content and structure depends on monose nature and oxidation conditions (first of all it depends on oxidizer strength). Aldoses are easier oxidized than ketoses.
Under the action of weak oxidizers (silver oxide ammonia solution, copper hydroxide, bromine water) aldoses are transformed into aldonic acids (aldehyde group is oxidized into acid carbonyl group).
А )
silver mirror test (Tollence’s reaction):
)
silver mirror test (Tollence’s reaction):
External sign of this reaction is metal silver layer formation on test tube walls.
B )
reaction with copper hydroxide (II):
)
reaction with copper hydroxide (II):
External sign of this reaction is copper hydroxide (II) blue precipitate transformation into copper oxide (I) red precipitate.
C) oxidation by bromine water (external sign of reaction is bromine solution discoloration):

W ith
the help of strong oxidizer (dissolved nitric acid) aldose terminal
groups (aldehyde and primary alcohol group) are simultaneously
oxidized into carboxylic groups, forming aldaric (sugar) acids, for
example:
ith
the help of strong oxidizer (dissolved nitric acid) aldose terminal
groups (aldehyde and primary alcohol group) are simultaneously
oxidized into carboxylic groups, forming aldaric (sugar) acids, for
example:
This reaction could be used to find galactose as mucous acid is practically not soluble in water. It should be taken into consideration that though in the mucous acid molecule there are four asymmetric carbon atoms (2, 3, 4, 5), it is optically inactive as it has plane of symmetry.
I n
the organism with the help of enzymes primary alcohol group could be
oxidized and aldehyde group is preserved un-oxidized. Reaction
products in this situation are uronic acids:
n
the organism with the help of enzymes primary alcohol group could be
oxidized and aldehyde group is preserved un-oxidized. Reaction
products in this situation are uronic acids:
In the organism uronic acids play an important role: they form water soluble glycosides with medical substances and their transformation products (metabolites) and toxic substances and they discharge these glycosides with urine; this is the origin of these acids name (latin urina is urine). D-glucuronic and L-iduronic acids and their derivatives are poly-saccharide structural elements (pectin substances, heparin, hyaluronic acid, chondroitin etc.). At the process of uronic acid metabolism ascorbic acid is synthesized (it is not synthesized at human beings).
Question 77. What compounds are received at D-fructose oxidation? Are they optically active?
Answer. Ketose oxidation is performed under strong oxidizers action and it is accompanied by carbon chain destruction. Bond breaking could appear by two methods: between the first and the second and between the second and the third carbon atoms. All terminal carbon atoms are oxidized with carboxylic group formation.
At D-fructose oxidation four reaction products are formed. At bond breaking between the first and the second carbon atoms formic and D-arabinaric acids are formed. At bond breaking between the second and the third carbon atoms oxalic and meso-tartaric acids are formed.

Oxalic and formic acids are optically inactive as there are no asymmetric carbon atoms in their molecules. Meso-tartaric acid contains two asymmetric carbon atoms but it has plane of symmetry that is why, it is not optically active. D-arabinaric acid follows all requirements to be optically active compound: in the molecule there are three asymmetric carbon atoms and there are no symmetry elements.
Question 78. What compounds are called glycosides?
Answer. Monose cyclic forms are built at interaction between aldehyde group or keto-group with alcohol group, as they are hemiacetals (hemi-ketals). Hemiacetals (hemiketals) at interaction with alcohols at acid medium give acetal (ketal):

Mono-saccharides also form acetals (ketals) which are called glycosides:

Glycosides are not only carbohydrate acetals formed at alcohol interaction. Glycosides are also products of hemi-acetal hydroxyl interaction with other compounds. Bond which is formed by hemi-acetal hydroxyl is also called hemi-acetal bond.
Glycosides could be classified according to the fact what aglycone atom is connected to saccharide part of glycoside: C-glycosides, O-glycosides, N-glycosides, S-glycosides.
According to the cycle type glycosides are subdivided into pyranosides and furanosides.
Glycoside non-carbohydrate part is called aglycone (could be translated as non-sugar). At this example it is metoxy group: methyl alcohol residue (marked by italics). Glycoside names are built in the following way: aglycone + glycoside. Carbohydrate ending “-ose” is changed into “-oside”. At the beginning they mark atom through which aglycone is attached to carbohydrate part.
Example of C-glycoside is pseudo-uridine, example of S-glycoside is sinigrin which is in mustard. Nucleosides are ribose and deoxyribose N-glycosides with nucleic bases which are nucleotide, DNA and RNA components.



Question 79. What is the process of ester formation from mono-saccharides?
A nswer.
Esters
could be received by organic acid anhydrides action on
mono-saccharides. For example, with acetic anhydride acetyl
mono-saccharide derivatives are received.
nswer.
Esters
could be received by organic acid anhydrides action on
mono-saccharides. For example, with acetic anhydride acetyl
mono-saccharide derivatives are received.
Esters are hydrolyzed at acid and alkaline medium.
Phosphoric acid esters are very important, they are called phosphates and they are at all plant and animal organisms. They include D-glucose phosphates: D-glucose 1-phosphate is received at glycogen hydrolysis with the help of enzyme called phosphorylase; glucose 6-phosphate is formed at glycolysis first stage (glucose catabolism in the organism).


After that there is glucose 6-phosphate isomerization into D-fructose 6-phosphate, which, in its turn, is phosphorylated into D-fructose 1,6-phosphate with the help of ATP.
D-ribose and 2-deoxyribose phosphates are structural elements of nucleic acids and several co-enzymes.
Sulfuric acid esters – sulfates – are in structure of poly-saccharides at connective tissue.
Question 80. Write di-saccharide structural formula, it is formed by -D-galactose two residues which are attached to each other by 1,4-glycoside bond. Write structural formula of di-saccharide interaction product with methyl alcohol if there is chlorine-hydrogen in the mixture. Could these compounds participate at “silver mirror test”? Write necessary equations of reactions.
Answer. -D-galactose molecules could form bonds between each other with the participation of 1st and 4th carbon atoms:


Glycoside hydroxyl is marked with a star, it can be substituted by –ОСН3 group at interaction with methanol:

Due to glycoside hydroxyl this di-saccharide could be transformed in linear form with aldehyde group at aqueous solution, so, it can give positive silver mirror test (equation is given in simplified form):
С12Н22О11
+ Аg2О
![]() С12Н22О12
+ 2Ag
С12Н22О12
+ 2Ag
Di-saccharide -D-galactose mono-methyl ether does not have glycoside hydroxyl and could not exist at linear form and it cannot participate at silver mirror test.
Question 81. What is sucrose molecule structure?
A nswer.
At
hydrolysis sucrose molecule splits into α-D-gluco-pyranose
molecule and β-D-fructo-furanose
molecule, thus, it is di-saccharide, in other words it comprises two
mono-saccharide residues:
nswer.
At
hydrolysis sucrose molecule splits into α-D-gluco-pyranose
molecule and β-D-fructo-furanose
molecule, thus, it is di-saccharide, in other words it comprises two
mono-saccharide residues:
These residues are attached to each other by glycoside bond between hydroxyl groups at anomer carbon atoms (1st atom at α-D-glucopyranose and 2nd atom at β-D-fructofuranose).
At sucrose molecule there are no free OH-groups at 1st carbon atom at α-D-glucose residue and at 2nd carbon atom at β-D-fructose residue (hemi-acetal or hydroxyl hydroxyls). That is why, sucrose molecule could not transform into open (aldehyde) form and give positive silver mirror test, and also it could not be oxidized by copper hydroxide, i.e. it does not reduce silver and copper, it is non-reducing carbohydrate.
Sucrose sources are sugar cane, beet root, plant and fruit juices.
Question 82. Is sucrose the sweetest substance?
Answer. Carbohydrate sweet taste makes our life brighter and tastier. The sweetest substance is not sucrose and it is not carbohydrate at all. Sweeter carbohydrate in comparison to sucrose is fructose. If relative sweet taste of sucrose is 100 than fructose taste is 173, glucose sweetness is 74, maltose and galactose taste is 32 and lactose sweetness is 16.
Mono-saccharides could be tasteless and even bitter and non-carbohydrate substances could be sweet. For example, beryllium and lead compounds could be sweet. Lead acetate is called lead sugar, but it is not recommended to use it as sugar due to lead compounds high toxicity.
Many organic substances also have sweet taste but they have nothing in common with sugars, for example, saccharin which sweetness exceeds sucrose sweetness 500 times. 4-Etoxy-phenyl-urae (dulcin) 200 times sweeter than sucrose and it was used as sucrose substitute until it was discovered that it is harmful for the organism.
Acesulphame and aspartame (di-peptide L-aspartyl-L-phenyl-alanine methyl ester) is 200 times sweeter than sucrose. L-aspartyl-amino-malonic acid methyl-phenchyl ester is 33000 times sweeter than sucrose and sucronic acid is 200000 times sweeter than sucrose.
Some proteins have very strong sweet taste. For example, protein monellin from tropic plant Dioscoreophyllum cumminsii is 3000 times sweeter than sucrose and protein taumatin from tropic plant Thaumacoccus daniellii is 750-1000 times sweeter than sugar and its complex with aluminium ions – tallin – is 35000 times sweeter than sucrose. Protein miraculin from Synsepalum dulcificum does not have sweet taste but could change sour taste of products into sweet.
Question 83. What oligo-saccharides are most-widely spread in nature?
Answer. Sucrose (cane sugar, beet root sugar) is widely spread at plants. It is non-reducing di-saccharide.
Maltose (malt sugar) is made from two -D-gluco-pyranose residues which are attached to each other by -1,4–glycoside bond. Maltose is formed at starch hydrolysis under the action of enzyme called amylase; if you are chewing bread for a long time you can feel maltose sweet taste, it is formed from bread starch under the action of saliva amylase. It is reducing di-saccharide.
Cellobiose is made from two -D-gluco-pyranose residues which are attached to each other by -1,4-glycoside bond. It is structural element of cellular tissue (cellulose). At free form it is at some plant juice. It is reducing di-saccharide.
Lactose (lactic sugar) is made from -D-galacto-pyranose residues and -D-gluco-pyranose residues which are attached to each other by -1,4-glycoside bond. Lactose is only at mammal milk and it is reducing di-saccharide.
In the organism lactose hydrolysis is performed with the help of enzyme which is called lactase. If there is small amount of lactase produced in the organism then there is lactase deficiency and inability to digest lactose. That is why, at milk use at people with lactase deficiency lactose is not digested but there is its putrefaction with intestinal micro-flora with unpleasant consequences (flatulence, diarrhea). At majority of people and mammals lactase gene is switched off due to maturation. This fact is explained by information that mammals use milk only at early age and later there is no use spending energy and amino acids on lactase synthesis. As a result of mutation at one of regulatory genes which product switched off lactase gene, this enzyme (lactase) started synthesizing all though the life cycle. About 70% of European people can digest milk at mature age, though in Africa, Oceania and Central Asia only 30% of all people can synthesize lactase at mature age. Peoples from Africa with traditionally high milk use (tutsy, tuaregs etc.) do not have any problems with lactose digestion.
Tregalose (fungus sugar) comprises two -D-gluco-pyranose residues attached to each other by hemi-acetal hydroxyl groups, thus, tregalose is non-reducing di-saccharide.
It is at fungus and some plants. At yeast tregalose amount reaches 18% on dry substance.
Other di-saccharides such as melibiose, hentibiose, turanose, primverose etc. are rare.
Tri-saccharides are rare. Tri-saccharide rafinose which comprises galactose, glucose and fructose is at sugar beet root. It is non-reducing tri-saccharide. Other tri-saccharides (hencianose, melecitose, manninotriose, cellotriose, planteose) are very rare.
Tetra-saccharide stachyose is made from two galactose residues and one glucose residue and one fructose residue. Stachyose is at Stachys roots, at yellow-gold, soya, pea seeds and it is non-reducing tetra-saccharide.
Cyclic oligo-saccharides – cyclo-dextrins (Shardinger’s dextrins) are formed at starch hydrolysis under the action of amylase from Bacillus macerans. They contain 6-10 D-glucose residues attached to each other by -1,4-glycoside bonds. Cyclo-dextrins have catalyst properties, for example, they are able to catalyze phenol ester hydrolysis. They form coloured complexes with iodine and iodine molecules are inside cyclo-dextrin cavity.
Question 84. What carbohydrates are called poly-saccharides?
Answer. Poly-saccharides or polyoses are high-molecular carbohydrates. According to their chemical nature poly-saccharides are poly-glycosides.
At poly-saccharide molecule there are many mono-saccharide residues which are attached to each other by glycoside bonds. To form a bond with previous residue, each new residue gives alcohol hydroxyl group usually at 4th or 6th carbon atoms. To form a bond with each new residue, each previous residue gives glycoside (hemi-acetal) hydroxyl.
At poly-saccharides with plant origin there are mainly (14)- and (16) bonds. Poly-saccharide chains could be branched and non-branched (linear).
Poly-saccharides are hydrolyzed at acid medium and are not exposed to hydrolysis at alkaline medium. Complete hydrolysis leads to mono-saccharide formation or their derivatives formation, whereas incomplete hydrolysis leads to intermediate oligo-saccharides formation, including di-saccharides.
Question 85. What is the difference between homo-poly-saccharides and hetero-poly-saccharides?
Answer. Poly-saccharides are subdivided into two large groups: homo-poly-saccharides and hetero-poly-saccharides.
Homo-poly-saccharides are made from one mono-saccharide residues, for example, starch, cellulose, glycogen etc.
Starch is a mixture of two poly-saccharides made from -D-gluco-pyranose residues: amylose (10-20 %) and amylopectin (80-90 %).
Amylose chain is un-branched, it comprises 200-1000 glucose residues (molecular mass is 160000).
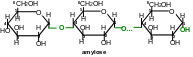
Amylose molecules wind in spiral, inner channel could be occupied by particular-sized molecules (for example, iodine molecules), forming complexes which are called intrusion complexes. Amylose complex with iodine is blue. It is used as qualitative reaction on starch (iodine-starch reaction).
Amylopectin, in opposition to amylose, is branched spiral. At its main chain α-D-gluco-pyranose resides are attached to each other by α(14)-glycoside bonds and at branch points they are attached by α(16)-glycoside bonds. Between branch points there are 20-25 α-D-gluco-pyranose residues. Amylopectin molecular mass is several millions.

Starch is white amorphous substance. It is not dissolved at cold water, at hot water it is swelling and dissolves only partially.
Starch is formed at plants at photosynthesis process and it is accumulated at roots, tubers and seeds. At digestive tract there is starch hydrolysis under the action of enzymes, hydrolysis final product is α-D-glucose.
Glycogen is plant starch structural and functional analogue which is at animal organisms. Glycogen is similar to amylopectin in its structure but it has more branched character of its chains. Between branch points usually there are 10-12 glucose parts, sometimes this number is 6. Glycogen molecular mass is really high (up to 100 millions). In the organism glycogen is reserve carbohydrate. If it is necessary enough, amount of glucose splits from it and energy reserve is increased in the organism. Amylopectin plays the role of reserve poly-saccharide at plants.
Cellulose or cellular tissue is the most-widely spread plant poly-saccharide. It plays supportive function at plants. At cotton there is about 100% of cellulose and in wood it is about 50-70 %.
Cellulose is made of β-D-gluco-pyranose residues which are attached to each other by β(14)-glycoside bonds. The chain is not branched, it has 2500-12000 β-D-glucose residues (molecular mass is 0,4-2 million).
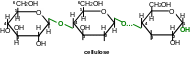
Cellulose linear structure is supported by hydrogen bonds which are formed inside carbohydrate chains and between separate chains. Cellulose is not digested at human gastro-intestinal tract and could not be nutrition substance but it is necessary for normal nutrition, as it facilitates gastro-intestinal tract regulation and it stimulates large intestine peristalsis.
Pectin substances are at fruits and vegetables. If there are organic acids these substances form jelly which is applied at food industry. Pectin substance molecules include pectic-poly-galacturonic acid which is made of D-galacturonic acid residues attached to each other by a-(1®4)-glycoside bond. Some pectin substances have anti-ulcer action and form the basis of some medicines, for example, plantaglucide produced from plantain.

Hetero-poly-saccharides are made from different mono-saccharide residues. Hetero-poly-saccharides are attached to proteins in the organism and form complex supra-molecular complexes. Examples of hetero-poly-saccharides are hyaluronic acid and heparin. Hyaluronic acid is made from D-glucuronic acid residues and N-acetyl-D-glucosamine attached to each other by β-(13)-glycoside bond.

Hyaluronic acid has high molecular mass and high viscosity. It provides connective tissue impermeability for pathogenic micro-organisms. Usually hyaluronic acid is not presented at free form, it is usually attached to poly-peptide chains. Mixed biopolymers are the basis for cells and liquids at animal organisms.
Heparin biopolymer structure contains D-glucosamine residues and two uronic acid residues: D-glucuronic acid and L-iduronic acid. L-iduronic acid predominates in quantity. Amino group at majority of glucosamine residues is sulfonated (contains SО3Н-group), at some residues it is acetylated (attached to СН3СО-group). In addition there are SО3Н groups at some L-iduronic acid residues. Heparin and its derivative – heparitine sulfate – in the organism are attached to protein. Heparin prevents blood coagulation, heparitine sulfate is in the structure of blood vessel walls.
