
Molecular Heterogeneous Catalysis, Wiley (2006), 352729662X
.pdf386 Chapter 9
cell. A few essential ingredients have to be part of the design. The catalytic system has to self organize, replicate and adapt itself. The system should not only self assemble around a template, but should also be able to replicate the template or the template structure. It should also behave as a reproductive and self-organizing reactor.
The property of reproduction and self organization will only exist for su ciently complex systems. Towards the design of the artificial catalytic cells a combinatorial, evolutionary process will have to be used. The realization of such an evolutionary adaptive process requires system conditions far from equilibrium and at a state near a critical point, such as those near a dynamic phase transition, where the developments in time and space are undecidable and hence, very sensitive to the choice of initial conditions.
9.6 Biomineralization, the Synthesis of Mesoporous Silicas
In the previous sections, we focused on chemical theories for the evolutionary origin of life-like systems. We learned that an important condition in the initiation of early life is a microporous or cell-type enclosed environment that can sustain local concentration variations.
Biomineralization is a biological synthesis process that exploits the unique interactions at the interface of the inorganic and organic material to produce composite materials with unique properties and inorganic materials with a unique structure, regular over nanometer length scales to micrometers and millimeters[33a]. The essence of biomineralization processes is that mineral is deposited around or within a mould, prestructured by organic building blocks that may consist of various types of molecules such as sugars or fatty esters. The key feature that characterizes these molecule is that they are amphiphilic. Such molecules can organize as double layers, as presented in cell membranes, and can have the shape of spheres, tubes or three-dimensional structures. These phases are known as liquid crystals. In the biomineralization process, minerals are deposited at the interfaces of the preorganized structures formed by the liquid crystal-type amphiphilic systems. This process that can be seen as a fixation of a fluidous system.
An extensive research activity in biomineralization has developed that investigates the structure-directing principles described above, but now chemically without the use of biological systems. This is of great interest in the context of designing the microporous cell-type enclosed conditions for protocell systems. Second, conversion of protocell-type catalytic systems into heterogeneous catalytic systems requires mineralization chemistry similar to biomineralization.
In this section and also in the next sections, we will highlight aspects of the chemistry that biomimics biomineralization. The treatment of the topic material will be much more detailed than the previous sections dealing with origin of life-like systems. We need the detail to compare the synthesis of ordered materials prepared in biomineralization with the disordered, often fractal, materials with high surface areas synthesized with more conventional chemistry. This comparison will be done in later sections. Again, processes in such materials are important to strenghten the walls of such systems. Stochastic modeling of such systems is possible, as will be shown in final sections. The chapter will be concluded with a summarizing section, which is less detailed. Some readers may prefer to skip the more technical sections that follow and read the final summarizing section of this chapter.
Zeolites are well-defined microporous systems that we introduced earlier. They have micropores ≤ 1 nm. Their synthesis requires the use of organic template molecules. As we described in Section 8.7.2, these template molecules organize silicate oligomers into
Heterogeneous catalysis the origin of life, biomineralization |
387 |
precursor molecules for crystallization of the zeolite. These precursor molecules di er depending of the templating molecule or cation used. In these systems there is no preorganization of the template molecules before crystallization. Using biomineralization synthesis techniques, ordered mesoporous and microporous systems can be made with micropore dimensions that vary over many length scales. In contrast to the zeolites, with channel walls of coordinatively saturated oxygen atoms, the channel walls of those systems are hydroxylated.
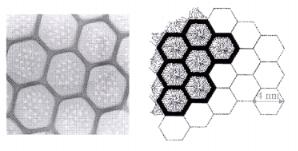
Biological systems that realize such ordered nanoand micrometer porosity include diatoms, which are monocellular algae. Figure 9.13 compares the micropore structure in a diatom with that of synthesized mesoporous materials.
Figure 9.13. Schematic drawings of (left) diatoms and (right) synthetic mesoporous MCM-41.
Here we will present the basic physicochemical principles on which the formation of such ordered microporosity is based. We limit ourselves here and in the following sec-
tions to the example of silica formation since this has been most extensively studied. The introduction in 1992 of the MCM-type periodic nanometer pore size materials[30], using surfactants in the synthesis instead of structure-directing template molecules, brought forward the development of methods to prepare mesoporous silicas with a great leap. In this approach, di erent lyotropic liquid crystal phases of a large variety of surfactants and amphiphilic polymers that can be organized in numerous mesophases are used to structure the developing silica phase. The silica phase is grown around the self assembled organic polymers, which act as a template. Surfactant molecules or amphiphilic polymers form micelles or liposome vesicles with a membrane that consists of a double layer of amphiphilic molecules. In an aqueous solution, their hydrophobic features point inwards, whereas their hydrophilic features are directed into the water phase. At increased concentration the micelles can become converted into tubular structures or even networks. Silicas with a well-defined network of uniform pore dimensions are formed by introducing silica precursors that oligomerize and subsequently solidify around the micelles. The pore sizes of as-made porous silica range from a few to tens of nanometers. Crystalline siliceous materials with larger pore dimensions ( 7.4 nm) can be made using non-ionic triblock copolymers of ethylene oxide and propylene oxide as templates under acidic conditions. The crystallinity of such materials can sometimes be directly deduced from the shape of the crystal particles. Figure 9.14 shows the rhombodecahedron shape of the corresponding crystals.
According to Zhao et al.[33a], a major breakthrough in biomineralization was the discovery of how to synthesize such materials at di erent pH. Kresge et al.[30] originally

388 Chapter 9
Figure 9.14. SEM images of mesoporous single crystal synthesized with F108 at 38 ◦C with 0.5 mol/L K2 SO4: a) at low magnification, b) at high magnification, c) index of a rhombdodecahedron model, the mesopore array in (110) plane is demonstrated. [32].
synthesized mesoporous materials in basic media with anionic silica species. The use of nearly neutral pH conditions, the same as in biological systems, was accomplished by use of [SiF6]2− and Triton X-100 as non-ionic surfactant, complementary to the use of conventional ethyltrimethylammonium cations, similar to those used in zeolite synthesis, as structure-directing agent[34]. The halide ion associates with the surfactant molecule. There is no need in this system to charge balance the surfactant charge with the negatively charged silica wall. The silica wall remains neutral. Tanev and Pinnavaia[35] were the first to use non-ionic surfactants such as poly(ethylene oxide) and poly(alkylene oxide) block copolymers with hydrophilic and hydrophobic parts. At neutral pH, disordered, worm-like pores are formed. If, on the other hand, low molecular weight block copolymer solutions
of relative high concentration are used at low pH, ordered mesoporous silica phases are synthesized[36,37] .
9.6.1 Biomimetic Approaches for Amorphous Silica Synthesis
Diatom biosilica formation can be mimicked by using bio-analogous reagents for silica synthesis. The diatom contains ordered micropores in a silica matrix which is amorphous. In this section, we show that amorphous silica can be considered as structures built for elementary particles. The packing of the elementary particles and their internal structure depend sensitively on the amphiphilic structure-directing molecules. The pH controls the preorganization of the structure-directing molecules. In this section we discuss materials made without preorganization of the structure-directing molecules.
Two types of enzymes have been identified as playing a role in the formation of biogenous silica. In a sponge, such enzymes are the silicateins[38] that catalyze the formation of Si–O–Si bonds from the corresponding monomer. Mechanistically the reaction
Heterogeneous catalysis the origin of life, biomineralization |
389 |
sequences in the enzyme fold are similar to the elementary reaction step in the papainlike hydrolysis reaction discussed in Chapter 4, section 4.4. The other enzymes, identified in diatoms, are the sila ns[39]. They are small peptides that consist of 15–18 amino acid residues linked to linear polyamines consisting of 5–11 N -methylamidpropylamine units. The sila ns induce the precipitation of silica under nearly neutral pH conditions. Synthetic polyamines such as polyethylenimine (PEI) along with natural species found in diatoms induce silica precipitation very e ciently. The reaction is generally too fast in the laboratory to investigate silica transformations and structure-directing processes. Polyethylene oxides (PEOs) and derivatives thereof are good alternatives, as they induce silica polymerization at a slower reaction rate. Consequently, it has been posible to perform and monitor time-resolved silica transformations, which eventually can be manipulated by temperature-controlled aging[40].
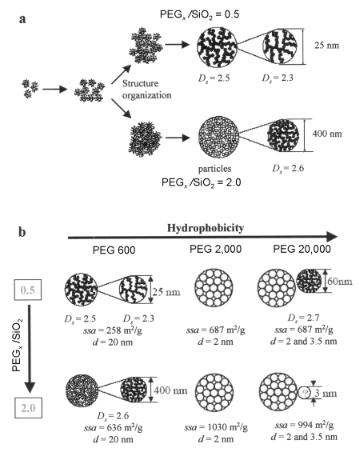
Figure 9.15. (a) Aggregation model for PEG600-mediated silica at PEO/ silica ratios 0.5 and 2.0. (b) Schematic presentation of the relation of PEO/silica ratio and hydrophobicity (which increases with size of the PEO monomer) and the physicochemical characteristics of the silica formed: specific surface area (ssa in m2g−1) determined by N2 sorption–desorption, fractal dimension (Ds) determined by SAXS, and pores diameter (d) determined from SAXS and pore volume contributions (derived from sorption– desorption isotherm)[41].
390 Chapter 9
When di erent hydrophylic homopolymers such as PEO600, PEO2000, PEO20,000 and PEI1,000,000, or triblock copolymers such as PEO76–PPO29–PEO76 or mixtures of them are used under mildly acidic conditions as additives in the biomimetic synthesis of silica from waterglass, well-defined spherical and smooth non-fractal and fractal silica particles (for definition fractals see Section 9.7.2) are formed. The silica aggregation behavior and pore formation behavior which are controlled by PEO homopolymers at various molecular weights and PEO/silica ratios have been studied with in situ (ultra) small-angle X-ray scattering [(U)SAXS]. The experimental results clearly indicate that PEO plays three di erent but important roles in silica aggregation. First, it serves as a flocculation agent in the formation of silica sols, similarly to PEI and the (analogues of) diatomaceous polyamines. Second, the hydrophobic silica–PEO interactions steer the silica polymerization. Finally, the presence of PEO induces phase separation during the reaction, in which PEO-rich and silica-rich phases are formed in the formerly homogeneous aqueous solution. As we will see later, phase separation processes play an important role in micromorphogenesis of the diatom cell wall.
From these studies, it becomes clear that the length of the PEO chain and the PEG/ silica ratio a ect the physicochemical properties of the silica formed with respect to a variety of pore dimensions from 2 up to 20 nm, their connectivity (determined as fractal dimensions with SAXS) and their specific surface areas. Consequently, di erent silicas can be created by choice (see also Section 9.7). A fractal dimension of <3 implies that a gel-type disordered, chaotic and open structure will form due to cluster–cluster aggregation processes that are di usion or reaction rate limited. A fractal packing of particles implies self similar scaling of the porosity distribution over several length scales. A fractal dimension of the order of 2.5 implies di usion-limited aggregation due to high sticking probabilities when reaction clusters meet. Figure 9.15a shows as a function of PEG[PEG = (poly ethylene oxide glycol)]x/SiO2, ratio that initially open or more dense gel-type structures are formed. They become converted into particles that themselves are built from smaller elementary particles that are fractally (PEGx/SiO2 = 0.5) or regularly packed. The elementary particles themselves can exist again with a fractal gel-inner structure as is the case for the silicas made with PEG600.Figure 9.15b summarizes the di erent types of end products as a function of polymer hydrophobicity and polymer/SiO2 ratio.
9.6.2 Micro-Emulsion Mediated Silica Formation
The diversified porous patterns of diatomaceous silicas are on the nanoto submicrometer scale ( 10–300 nm) and these mesoand macropores cannot be mediated by single macromolecules, not even proteins. To mimic these mesoand macroporous structures, a di erent approach can be applied based on a phase separation process as in the vesiclemediated macromorphogenesis processes extensivily reviewed in Pickett-Heaps et al.[42]. In this case oil-in-water (O/W) emulsions are applied as a model system. O/W emulsions are isotropic and thermodynamically stable liquid media with a continuous water domain and an oil domain, which are thermodynamically stabilized by a surfactant as micrometer-sized liquid entities.
Amphiphilic PEO–PPO–PEO triblock copolymers can form a variety of aggregates depending on the length of the respective polymer blocks. In such aggregates, the more hydrophobic PPO blocks are shielded from the aqueous envirement by hydrophilic PEO blocks that protrude into the aqueous phase. The triblock copolymer poly(ethylene oxide)– poly(propylene oxide)–poly(ethylene oxide) (PEO76/PPO29/PEO76: see Fig. 9.16a) can

Heterogeneous catalysis the origin of life, biomineralization |
391 |
Figure 9.16. (a) the chemical structure of the triblock copolymer poly(ethylene oxide)–poly(propylene oxide)–poly(ethylene oxide) (PEO76 –PPO29 –PEO76) and (b) the proposed structure of the emulsion droplets. TMB = 1,3,5-trimethylbenzene.
also be used with emulsions of 1,3,5-trimethylbenzene (TMB). These emulsion droplets act as templates in the biomimetic formation of silica (Fig. 9.16b), thereby exploiting the ability of the PEO chains on the aggregate surface to induce the precipitation of silica. After removal of the organics by high-temperature calcination, cavities of micrometer-size dimensions should remain in the as-made silicas.
Figure 9.17. Scanning (a, c) and high-resolution (b, d) micrographs of hollow silica spheres synthesized with the TMB/PEO–PPO–PEO/silica emulsion system at 80 ◦C[43].

392 Chapter 9
Figure 9.18. Schematic drawing of the templating mechanism by a downscaling phase separation model [(A) to (D)] in comparison with scanning electron micrographs of the valves from diatom Coscinodiscus wailesii [(E) to (H)]. (A) The monolayer of polyamine-containing droplets in close-packed arrangement guides silica deposition. (B and C) Consecutive segregations of smaller (about 300 nm) droplets open new routes for silica precipitation. (D) Dispersion of 300 nm droplets into 50 nm droplets guides the final stage of silica deposition. Silica precipitation occurs only within the water phase (white areas). The repeated phase separations produce a hierarchy of self-similar patterns. (E to H) SEM images of valves at the corresponding stages of development[44].
At pH 5.2, hollow spheres are formed with a uniform size distribution and an average diameter of approximately 1 µm. These silica spheres are thermally very stable and hollow (Fig. 9.17), possessing a relatively thin shell with a very regular thickness of 80–130 nm composed of 7–8 nm thick layers of silica with an interlamellar spacing of 3–4 nm[43] .
The biomimetic studies of biomineralization have also provided inspiration to explain biopolymer-mediated silica formation in the diatoms. As mentioned in the previous section, biogenic silica contain small proteins and polyamines such as the sila ns.
The sila n-1A proteins are small (2.4–3.1 kDa) polycationic proteins with highly modified amino acid residues. A particular well-characterized peptide contain seven phosphorylated serine residues and one phosphorylated trimethylhydroxylysine moiety.
The zwitterionic structure of the native sila ns leads to the self assembly of these molecules, which explains their extremely e cient induction of silica precipitation. Following up on this model, Sumper[44] proposed, on the basis of simulations, a model that consists of multiple steps in which phase separation processes occur. At the initial stage phase separation of protein phase and silica permits the formation of the large, honeycomb structures, followed by several intermittent steps of silica formation – each mediated
by phase separation processes – to create smaller structures (see Fig. 9.18).
In this “downscaling” model, the largest structures are formed at the start (Fig. 9.18a) followed by the formation of the smaller ones (Figs. 9.19b,c) and finally the more delicate
details (Fig. 9.18d).
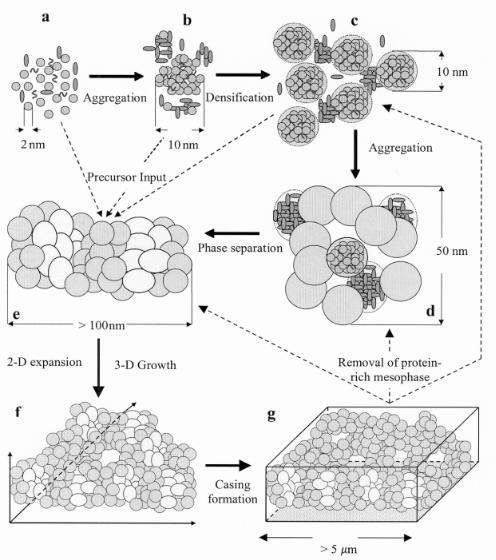
In contrast, Vrieling et al.[45] derived an “upscaling” model based on experimental data obtained from in situ time-resolved ultra-small angle X-ray scattering analysis of silica transformations mediated by synthetic polymers [poly(ethylene glycol), polyethylenimine)] and protiens (myoglobin, horseradish peroxidase). In the course of the polymerization and transformation processes (the latter of which is induced by aging), phase separation occurs. Silica-rich and structure-directing containing macromolecule phases appear, which continuously interact during the transformation of silica from smaller aggregates to the larger structures (see Fig. 9.19).
Ultimately, removal of the structure-directing-rich phase leads to the creation of a
Heterogeneous catalysis the origin of life, biomineralization |
393 |
Figure 9.19. Schematic presentation of the described upscaling model. At the onset of valve formation
(a) silica precursors and peptides are imported into the silica deposition vesicle (SDV), where precipitation of silica is induced by small organic molecules (sila ns and/or polyamines) to form silica sols (b). These sols further densify and grow to larger silica particles, while larger peptides start to interact with the silica
(c). When aggregation continues (d), silica forms larger particles (up to 50 nm). At this stage the silica and protein aggregates become transferred to silicaand protein-rich mesophases by phase separation
(e). This process proceeds until the SDV has reached it final two and three-dimensional size (f) and the protective casing has been formed (g). In order to leave the pores, the protein-rich mesophase is somehow removed prior to assembly of a protective casing before the wall leaves the cell[45].
394 Chapter 9
porous network ordered over several length scales. The two models can be reconciled by viewing the phase separation process proposed by Vrieling et al. as step A in the diatom formation model according to Sumper.
9.7 Aging of Silica Gels
9.7.1 Silica Gel Synthesis
After their initial formation, conventional silicas, formed by acidifying silicate solutions, can be significantly influenced by consecutive aging processes. When the fragile, lowdensity silicas as prepared in solution are dried, they often collapse. The surface tension of the water droplets disrupts the silica structure. Aging processes are important because in the process the walls of the micropores thicken and, hence, become resistant to drying processes.
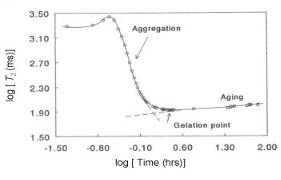
Here we will summarize the reaction steps that lead to silica gel formation, and focus on the physical chemistry of the aging processes. NMR proton spin–latice relaxation measurements[46] can be used to follow the morphological changes that occur in a silica gel as a function of time. Owing to the restricted movements and, hence, increased correlation times of the water molecules near to the silica–water interface, protons of water included in a porous solid have longer relaxation times than water in the bulk. A decrease in the spin–spin relaxation time is to be interpreted as an increase in the number of water molecules restricted in their movements due to the proximity of the silica structure. Figure 9.20 shows the spin–spin relaxation T2 for a silica system as a function of relaxation time. The decrease in T2 in the first 20 min of the reaction indicates the formation of increasing silica surfaces in contact with water.
Figure 9.20. Spin–spin relaxation behavior as a function of reaction time for a silica system. CSiO2 =
0.73 M, pH = 4 , T = 40 ◦C, F/Si = 0. The small initial increase is caused by initial warming up of the sample[46].
This surface formation is due to the polymerization of silica monomers and oligomers and subsequent aggregation of the particles formed into clusters. When the minimum value of T2 is reached, the beginning of gelation starts. After gelation, aging processes will become dominant. Silica dissolution leads to the formulation of convex soluble surfaces and redeposition of this material in the crevices and necks (concave surfaces) decreases the specific surface area and the average correlation time of the interacting water molecules increases slightly.
Heterogeneous catalysis the origin of life, biomineralization |
395 |
In acidified wate glass at pH = 4, the following di erent reaction steps may be discerned:
1.particle growth
2.aggregation
3.gelation
4.aging.
Steps 1, 2 and 4 are mass transformations successively in the reaction mixture. When gelation occus, a percolating network will be observed only above a certain silica concentration. First the monomers and oligomers present in the reaction medium will start to combine according to the condensation reaction
W |
|
Si O− + HO Si |
W W |
Si O Si |
|
W + OH− |
(9.1) |
|
|
− |
− |
|
− − |
|
|
||
resulting in a mixture of monomers, oligomers and small polymers.
Particle growth is favored at high pH, leading to larger sphere-like particles. At low pH, however, gel-type structures evolve.
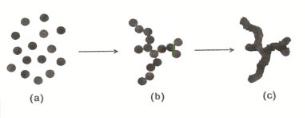
As soon as particles are formed, also particle–particle bonds may be formed according to Eq. (9.1), thus leading to open aggregates (Fig. 9.22) which may exhibit fractal properties, to be discussed in Section 9.7.2.
Figure 9.21. Aggregation of single particles (a) into agglomerates (b) and subsequent aging (c).
The structure of the aggregates formed is dependent on reaction parameters used during preparation of the silica gel. Di usion rates are inversely proportional to the size of the di using particles or aggregates.
There are two limiting cases of aggregation, namely reaction-limited and di usionlimited cluster–cluster aggregation. In the case of di usion-limited aggregation, the reaction between particles occurs upon collision, resulting in open, tenuous structures. For reaction-limited systems, particles will collide more often before reaction occurs and the primary particles are therefore able to penetrate deeper into the already existing aggregates, thus resulting in more compact, denser aggregates.
Aggregation continues until all primary particles have been incorporated into aggregates or, if the concentration is su cient, when a percolating network is formed throughout the entire reaction vessel. Gelation is characterized by the fact that the viscosity increases rapidly from the viscosity of the liquid to the viscosity of a solid.
Particles that are not part of the aggregates or network can continue to di use freely through the liquid and condense on this gel backbone, changing the properties of the gel. This is a form of aging (see Fig. 9.21), which may result in a substantial growth of the branches of the network long after aggregation and gelation.
