
ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 12
ʋ ɷɬɚɩɚ |
ɗɬɚɩɵ ɢɫɫɥɟɞɨɜɚɧɢɹ |
12Ɉɫɚɠɞɟɧɢɟ ɤɚɬɢɨɧɨɜ ȱȱȱ ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ, ɢ ɢɨɧɨɜ Pb2+ ɢɡ
ɰɟɧɬɪɢɮɭɝɚɬɚ 10 ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɚ H2SO4 ɜ ɩɪɢɫɭɬɫɬɜɢɢ C2H5OH ɩɪɢ ɧɚɝɪɟɜɚɧɢɢ:
H2SO4, ǻ
Ba2+  BaSO4p
BaSO4p
H2SO4, ǻ
Sr2+  SrSO4p
SrSO4p
H2SO4, ɋ2ɇ5Ɉɇ, ǻ
Ca2+  CaSO4p
CaSO4p
H2SO4, ǻ
Pb2+  PbSO4p
PbSO4p
ɉɪɨɦɵɜɚɧɢɟ ɨɫɚɞɤɚ ɜɨɞɨɣ, ɫɨɞɟɪɠɚɳɟɣ H2SO4
13Ɉɬɞɟɥɟɧɢɟ PbSO4p ɨɬ ɨɫɚɞɤɚ 12 ɞɟɣɫɬɜɢɟɦ 30% ɪɚɫɬɜɨɪɚ NH4CH3COO ɫ ɩɨɫɥɟɞɭɸɳɢɦ ɰɟɧɬɪɢɮɭɝɢɪɨɜɚɧɢɟɦ:
NH4CH3COO
PbSO4p  [PbSO4 · Pb(CH3COO)2]
[PbSO4 · Pb(CH3COO)2]
ɉɨɫɥɟ ɰɟɧɬɪɢɮɭɝɢɪɨɜɚɧɢɹ ɢɫɫɥɟɞɭɸɬ ɨɫɚɞɨɤ ɫɭɥɶɮɚɬɨɜ ɤɚɬɢɨɧɨɜ ȱȱȱ ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɩɨ ɫɯɟɦɟ ɫɢɫɬɟɦɚɬɢɱɟɫɤɨɝɨ ɯɨɞɚ ɚɧɚɥɢɡɚ ɫɦɟɫɢ ɤɚɬɢɨɧɨɜ ȱȱȱ ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
14ɂɫɫɥɟɞɨɜɚɧɢɟ ɨɫɚɞɤɚ 13 ɩɨ ɫɯɟɦɟ ɫɢɫɬɟɦɚɬɢɱɟɫɤɨɝɨ ɯɨɞɚ ɚɧɚɥɢɡɚ ɫɦɟɫɢ ɤɚɬɢɨɧɨɜ ȱȱȱ ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
15Ɉɬɞɟɥɟɧɢɟ ɤɚɬɢɨɧɨɜ IV ɝɪɭɩɩɵ ɨɬ ɤɚɬɢɨɧɨɜ V, VI ɚɧɚɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ ɞɟɣɫɬɜɢɟɦ ɢɡɛɵɬɤɚ 6Ɇ ɪɚɫɬɜɨɪɚ NaOH ɜ ɩɪɢɫɭɬɫɬɜɢɢ 3%
H2O2: |
|
|
|
|
|
|
|
|
|
|
|
|||||
|
NaOH |
|
|
ɢɡɛ. NaOH |
|
|
||||||||||
Al3+ |
|
|
|
|
Al(OH)3p |
|
|
|
|
|
|
[Al(OH)6]3- |
||||
|
|
|
|
|
|
|
|
|
|
|||||||
|
NaOH |
|
|
ɢɡɛ. NaOH |
|
|
||||||||||
Zn2+ |
Zn(OH)2p |
[Zn(OH)4]2- |
||||||||||||||
|
NaOH |
Cr(OH)3p |
ɢɡɛ. NaOH, H2O2 |
|||||||||||||
Cr3+ |
|
|
|
|
|
|
CrO42- |
|||||||||
|
NaOH |
Sn(OH)2p |
|
|
ɢɡɛ. NaOH, H2O2 |
|||||||||||
Sn2+ |
|
|
|
|
|
|
|
|
|
|
|
[Sn(OH)6]2- |
||||
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
NaOH |
|
|
ɢɡɛ. NaOH |
|
|
|||||||
Sn (IV) |
|
|
Sn(OH)4p |
[Sn(OH)6]2- |
||||||||||||
|
|
|
|
NaOH |
H2O2 |
|
|
|||||||||
As (III) |
|
|
|
AsO33- |
|
|
|
|
AsO43- |
|||||||
|
|
|
|
|
|
|
||||||||||
|
|
|
NaOH |
H2O2 |
|
|
||||||||||
As (V) |
|
|
|
AsO43- |
|
|
|
|
AsO43- |
|||||||
|
|
|
|
|
|
|
||||||||||
ɉɪɢ ɷɬɨɦ ɤɚɬɢɨɧɵ V, VI ɚɧɚɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ ɨɛɪɚɡɭɸɬ ɨɫɚɞɤɢ: Fe(OH)3, Mg(OH)2, MnO2 · nH2O, BiOCl, SbO2Cl, Cu(OH)2, HgO, Ni(OH)2, Co(OH)3
31
ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 12
ʋ ɷɬɚɩɚ |
ɗɬɚɩɵ ɢɫɫɥɟɞɨɜɚɧɢɹ |
16Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ As (V) ɜ ɨɬɞɟɥɶɧɨɣ ɩɨɪɰɢɢ ɰɟɧɬɪɢɮɭɝɚɬɚ 15 ɞɟɣɫɬɜɢɟɦ ɦɟɬɚɥɥɢɱɟɫɤɨɝɨ ɰɢɧɤɚ ɜ ɫɪɟɞɟ HCl (ɨɛɧɚɪɭɠɟɧɢɸ ɦɟɲɚɸɬ ɢɨɧɵ Sb (III), Sb (V)):
Zn; HCl
As (III), (V)  AsH3n AsH3
AsH3n AsH3
Ȼɭɦɚɠɤɚ, ɫɦɨɱɟɧɧɚɹ AgNO3  Agp (ɱɟɪɧɵɣ) (ɪɟɚɤɰɢɹ Ƚɭɬɰɚɣɬɚ)
Agp (ɱɟɪɧɵɣ) (ɪɟɚɤɰɢɹ Ƚɭɬɰɚɣɬɚ)
|
|
AsH2(HgCl) |
Ȼɭɦɚɠɤɚ, ɫɦɨɱɟɧɧɚɹ |
AsH3 |
AsH(HgCl)2 |
[HgCl ] |
|
|
(ɪɟɚɤɰɢɹ Ɂɚɧɝɟɪ-Ȼɥɟɤɚ) |
2 |
|
|
As(HgCl)3 |
|
|
|
|
|
|
As2Hg3 |
(ɠɟɥɬɨ-ɛɭɪɵɣ)
17Ɉɬɞɟɥɟɧɢɟ ɝɢɞɪɨɤɫɨɚɧɢɨɧɨɜ [Al(OH)6]3-; [Sn(OH)6]2- ɢɡ ɰɟɧɬɪɢɮɭɝɚɬɚ 15 ɞɟɣɫɬɜɢɟɦ ɤɪɢɫɬɚɥɥɢɱɟɫɤɨɝɨ NH4Cl ɩɪɢ ɧɚɝɪɟɜɚɧɢɢ:
NH4Cl, ǻ
[Al(OH)6]3-  Al(OH)3p
Al(OH)3p
NH4Cl, ǻ
[Sn(OH)6]2-  Sn(OH)4p
Sn(OH)4p
18 Ɋɚɫɬɜɨɪɟɧɢɟ ɨɫɚɞɤɚ 17 ɞɟɣɫɬɜɢɟɦ 2Ɇ HCl:
HCl Al(OH)3p  Al3+
Al3+
HCl
Sn(OH)4p  [SnCl6]2-
[SnCl6]2-
19Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Al3+ ɞɟɣɫɬɜɢɟɦ ɧɚ ɰɟɧɬɪɢɮɭɝɚɬ 18 ɪɚɫɬɜɨɪɨɜ ɚɥɢɡɚɪɢɧɚ ɢɥɢ ɧɚɬɪɢɹ ɚɰɟɬɚɬɚ:
HO OH
Al
O O
Ⱥɥɢɡɚɪɢɧ, NaOH OH
Al3+
O
CH3COONa
Al3+  Al(OH)2CH3COOp
Al(OH)2CH3COOp
32
ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 12
ʋ ɷɬɚɩɚ |
ɗɬɚɩɵ ɢɫɫɥɟɞɨɜɚɧɢɹ |
20Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Sn (IV) ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɚ ɫɨɥɢ ɪɬɭɬɢ (ȱȱ) ɧɚ ɩɪɟɞɜɚɪɢɬɟɥɶɧɨ ɩɪɨɤɢɩɹɱɟɧɧɵɣ ɪɚɫɬɜɨɪ 18 ɫ ɠɟɥɟɡɧɵɦɢ ɫɬɪɭɠɤɚɦɢ ɜ ɫɪɟɞɟ HCl:
|
Fe, HCl; ǻ |
|
HgCl2 |
|
[SnCl6]2- |
|
Sn2+ |
|
Hgp |
|
|
|||
21Ɉɛɧɚɪɭɠɟɧɢɟ ɤɚɬɢɨɧɨɜ Zn2+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɟ 17 ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɨɜ ɞɢɬɢɡɨɧɚ, ɢɥɢ K4[Fe(CN)6]:
|
ɞɢɬɢɡɨɧ |
C H |
-N |
|
|
|
C=N-NH-C6H5 |
||||
|
|
|
6 5 |
: |
|
S |
|
|
|
||
[Zn(NH3)4]2+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
N |
Zn |
N |
|
||||
|
|
|
|
S |
|
|
|
|
|||
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
:N |
-C H |
|||
|
C H -NH-N=C |
|
|||||||||
|
|
|
|
|
6 5 |
||||||
6 |
5 |
|
|
|
|
|
|
|
|
||
K4[Fe(CN)6]
[Zn(NH3)4]2+  K2Zn3[Fe(CN)6]2p
K2Zn3[Fe(CN)6]2p
22Ɉɬɞɟɥɟɧɢɟ ɢɨɧɨɜ Sb (V) ɨɬ ɞɪɭɝɢɯ ɤɚɬɢɨɧɨɜ V, VI ɚɧɚɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ ɞɟɣɫɬɜɢɟɦ HNO3 ɫ H2O2 ɧɚ ɨɫɚɞɨɤ 15:
HNO3; H2O2
SbO2Cl  HSbO3p
HSbO3p
ɉɪɢ ɷɬɨɦ ɞɪɭɝɢɟ ɧɟɪɚɫɬɜɨɪɢɦɵɟ ɫɨɟɞɢɧɟɧɢɹ ɤɚɬɢɨɧɨɜ V, VI ɝɪɭɩɩ ɪɚɫɬɜɨɪɹɸɬɫɹ ɫ ɜɨɫɫɬɚɧɨɜɥɟɧɢɟɦ ɫɥɟɞɭɸɳɢɯ ɤɚɬɢɨɧɨɜ:
HNO3, H2O2 MnO2 · nH2Op  Mn2+
Mn2+
HNO3, H2O2 Co(OH)3p  Co2+
Co2+
23 Ɋɚɫɬɜɨɪɟɧɢɟ ɨɫɚɞɤɚ 22 ɜ ɪɚɫɬɜɨɪɟ HCl: Hɋl
HSbO3p  [SbCl6]-
[SbCl6]-
24Ɉɛɧɚɪɭɠɟɧɢɟ Sb (V) ɜ ɪɚɫɬɜɨɪɟ 23 ɞɟɣɫɬɜɢɟɦ ɦɟɬɚɥɥɢɱɟɫɤɨɝɨ ɰɢɧɤɚ ɧɚ ɧɢɤɟɥɟɜɨɣ ɩɥɚɫɬɢɧɤɟ:
Zn
[SbCl6]-  Sbp
Sbp
33

ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 12
ʋ ɷɬɚɩɚ |
ɗɬɚɩɵ ɢɫɫɥɟɞɨɜɚɧɢɹ |
25Ɉɫɚɠɞɟɧɢɟ ɤɚɬɢɨɧɨɜ V ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɢɡ ɰɟɧɬɪɢɮɭɝɚɬɚ 22 ɞɟɣɫɬɜɢɟɦ ɤɨɧɰ. NH3 H2O:
NH3 · H2O
Mg2+  Mg(OH)2p
Mg(OH)2p
NH3 · H2O
Mn2+  Mn(OH)2p
Mn(OH)2p
NH3 · H2O
Fe3+  Fe(OH)3p
Fe(OH)3p
NH3 · H2O
Bi3+  BiONO3p
BiONO3p
ɉɪɢ ɷɬɨɦ ɤɚɬɢɨɧɵ VI ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɨɫɬɚɸɬɫɹ ɜ ɪɚɫɬɜɨɪɟ ɜ ɜɢɞɟ ɚɦɦɢɚɤɚɬɨɜ:
NH3 · H2O
Cu(OH)2p  [Cu(NH3)4]2+ NH3 · H2O
[Cu(NH3)4]2+ NH3 · H2O
HgOp  [Hg(NH3)4]2+
[Hg(NH3)4]2+
NH3 · H2O
Co(OH)2p  [Co(NH3)6]2-
[Co(NH3)6]2-
NH3 · H2O
Ni(OH)3p  [Ni(NH3)6]2+
[Ni(NH3)6]2+
26 Ɉɬɞɟɥɟɧɢɟ ɤɚɬɢɨɧɨɜ Mg2+ ɞɟɣɫɬɜɢɟɦ ɧɚ ɨɫɚɞɨɤ 25 ɪɚɫɬɜɨɪɚ
NH4Cl + 3% H2O2:
NH4Cl Mg(OH)2p  Mg2+
Mg2+
3% H2O2
Mn(OH)2  MnO2 nH2Op ɋɨɫɬɚɜ ɨɫɚɞɤɚ: MnO2 nH2Op, Fe(OH)3
MnO2 nH2Op ɋɨɫɬɚɜ ɨɫɚɞɤɚ: MnO2 nH2Op, Fe(OH)3
27Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Mg2+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɚɟ 26 ɞɟɣɫɬɜɢɟɦ Na2HPO4 ɜ ɩɪɢɫɭɬɫɬɜɢɢ ɚɦɦɢɚɱɧɨɝɨ ɛɭɮɟɪɧɨɝɨ ɪɚɫɬɜɨɪɚ:
Na2HPO4, NH3 H2O + NH4Cl
Mg2+ |
MgNH4PO4p |
28 Ɉɬɞɟɥɟɧɢɟ ɤɚɬɢɨɧɨɜ Bi3+ ɢ Fe3+ ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɚ HNO3 ɧɚ ɨɫɚɞɨɤ 26:
HNO3 BiONO3p  Bi3+
Bi3+
HNO3 Fe(OH)3p  Fe3+
Fe3+
ȼ ɨɫɚɞɤɟ MnO2 nH2Op
29Ɋɚɫɬɜɨɪɟɧɢɟ ɨɫɚɞɤɚ MnO2 · n H2O, ɩɨɥɭɱɟɧɧɨɝɨ ɩɨ ɩ.28, ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɚ HNO3 ɜ ɩɪɢɫɭɬɫɬɜɢɢ H2O2:
HNO3; H2O2
MnO2 · n H2Op  Mn2+
Mn2+
34
|
|
|
|
|
ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 12 |
||
|
|
|
|
|
|
|
|
ʋ ɷɬɚɩɚ |
|
ɗɬɚɩɵ ɢɫɫɥɟɞɨɜɚɧɢɹ |
|
|
|
||
|
|
|
|
||||
30 |
Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Mn2+ ɞɟɣɫɬɜɢɟɦ ɧɚ ɪɚɫɬɜɨɪ 29 (NH4)2S2O8: |
||||||
|
|
(NH4)2S2O8; HNO3; AgNO3 |
|||||
|
Mn2+ |
|
|
|
|
|
MnO4- |
|
|
|
|
|
|
||
31 |
Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Bi3+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɟ 28 |
ɞɟɣɫɬɜɢɟɦ ɫɜɟɠɟɩɪɢ- |
|||||
|
ɝɨɬɨɜɥɟɧɧɨɝɨ Na4[Sn(OH)6]: |
Na4[Sn(OH)6] |
|||||
|
|
|
Bi3+ |
|
|
Bip |
|
|
|
|
|
|
|||
32Ɋɚɡɪɭɲɟɧɢɟ ɚɦɦɢɚɤɚɬɨɜ ɜ ɪɚɫɬɜɨɪɟ 25 ɞɟɣɫɬɜɢɟɦ 2Ɇ H2SO4
33Ɉɬɞɟɥɟɧɢɟ ɢɨɧɨɜ Cu2+ ɢ Hg2+ ɨɬ ɨɫɬɚɥɶɧɵɯ ɤɚɬɢɨɧɨɜ VI ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɞɟɣɫɬɜɢɟɦ Na2S2O3 ɧɚ ɪɚɫɬɜɨɪ 32:
Na2S2O3; H2O; ǻ
Cu2+  Cu2Sp
Cu2Sp
Na2S2O3; H2O; ǻ
|
Hg2+ |
|
|
|
|
|
|
HgSp |
|
|
|
|
|
|
|
||
34 |
Ɉɬɞɟɥɟɧɢɟ Cu2S ɨɬ HgS ɢɡ ɨɫɚɞɤɚ 33 ɞɟɣɫɬɜɢɟɦ ɪɚɡɛ. HNO3 ɩɪɢ |
|||||||
|
ɧɚɝɪɟɜɚɧɢɢ: |
|
HNO3; ǻ |
|
|
|
||
|
Cu2Sp |
|
|
|
Cu2+ |
|||
|
|
|
|
|||||
|
HgS p ɜ ɷɬɢɯ ɭɫɥɨɜɢɹɯ ɧɟ ɪɚɫɬɜɨɪɹɟɬɫɹ. |
|||||||
35 |
Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Cu2+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɟ 34 ɞɟɣɫɬɜɢɟɦ ɤɨɧɰ. |
|||||||
|
NH3 H2O: |
NH3 H2O |
|
|
|
|||
|
Cu2+ |
|
|
|
[Cu(NH3)4]2+ |
|||
|
|
|
|
|||||
36Ɋɚɫɬɜɨɪɟɧɢɟ ɨɫɚɞɤɚ 34 ɞɟɣɫɬɜɢɟɦ ɛɪɨɦɧɨɣ ɜɨɞɵ ɜ ɩɪɢɫɭɬɫɬɜɢɢ HCl ɩɪɢ ɧɚɝɪɟɜɚɧɢɢ, ɢɥɢ ɜ ɰɚɪɫɤɨɣ ɜɨɞɤɟ:
Br2; HCl, ǻ
HgSp  [HgCl2] + Sp
[HgCl2] + Sp
ɤɨɧɰ. HNO3, ɤɨɧɰ. HCl
HgSp  [HgCl2]
[HgCl2]
37 ɈɛɧɚɪɭɠɟɧɢɟɢɨɧɨɜHg2+ ɜɰɟɧɬɪɢɮɭɝɚɬɟ36 ɞɟɣɫɬɜɢɟɦɪɚɫɬɜɨɪɚSnCl2: SnCl2
[HgCl2]  Hgp
Hgp
38 Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ ɋɨ2+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɟ 33 ɞɟɣɫɬɜɢɟɦ ɪɚɫɬɜɨɪɚ NH4SCN ɜ ɩɪɢɫɭɬɫɬɜɢɢ ɚɦɢɥɨɜɨɝɨ ɫɩɢɪɬɚ:
NH4SCN
ɋɨ2+  (NH4)2[Co(SCN)4]
(NH4)2[Co(SCN)4]
ɋ5ɇ11Ɉɇ
39 Ɉɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Ni2+ ɜ ɰɟɧɬɪɢɮɭɝɚɬɟ 33 ɞɟɣɫɬɜɢɟɦ ɪɟɚɤɬɢɜɚ ɑɭɝɚɟɜɚ (ɞɢɦɟɬɢɥɝɥɢɨɤɫɢɦɚ): O...HO
H3C-C=N N=C-CH3
|
|
ɪ-ɜ ɑɭɝɚɟɜɚ |
Ni |
|
|
|
H3C-C=N |
N=C-CH3 |
|
Ni |
2+ |
|
||
|
|
|
|
|
|
(ɞɢɦɟɬɢɥɝɥɢɨɤɫɢɦ) |
OH...O |
||
|
|
|
||
35
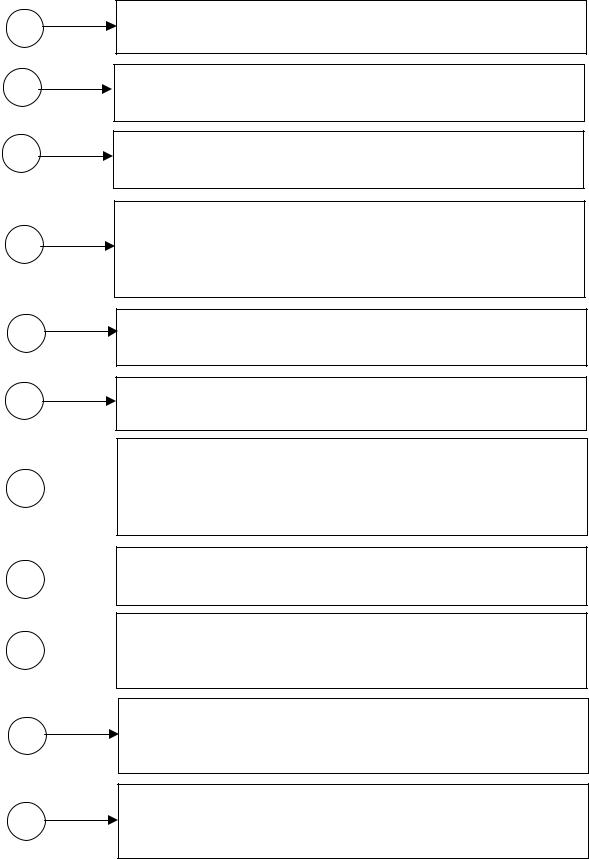
ɋɯɟɦɚ 7
ɋɏȿɆȺ ȺɇȺɅɂɁȺ ɋɆȿɋɂ ɄȺɌɂɈɇɈȼ I-VI ȺɇȺɅɂɌɂɑȿɋɄɂɏ ȽɊɍɉɉ ȻȿɁ ɈɋȺȾɄȺ
Ɉɛɧɚɪɭɠɟɧɢɟ ɜ ɨɬɞɟɥɶɧɵɯ ɩɪɨɛɚɯ ɤɚɬɢɨɧɨɜ NH4+; Fe2+;
1 Fe3+
2ɍɞɚɥɟɧɢɟ ɢɨɧɨɜ NH4+ ɢɡ ɨɬɞɟɥɶɧɨɣ ɩɪɨɛɵ ɞɟɣɫɬɜɢɟɦ Na2CO3 ɢɥɢ NaOH ɢ ɨɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ K+ ɜ ɪɚɫɬɜɨɪɟ
3ɍɞɚɥɟɧɢɟ ɢɨɧɨɜ NH4+ ɢɡ ɨɬɞɟɥɶɧɨɣ ɩɪɨɛɵ ɞɟɣɫɬɜɢɟɦ K2CO3 ɢɥɢ KOH ɢ ɨɛɧɚɪɭɠɟɧɢɟ ɢɨɧɨɜ Na+ ɜ ɪɚɫɬɜɨɪɟ
Ɉɫɚɠɞɟɧɢɟ ɤɚɬɢɨɧɨɜ II ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɢɡ ɢɫ-
4ɯɨɞɧɨɝɨ ɪɚɫɬɜɨɪɚ ɞɟɣɫɬɜɢɟɦ 2 Ɇ ɪɚɫɬɜɨɪɚ HCl ɢ ɢɫɫɥɟɞɨɜɚɧɢɟ ɩɨɥɭɱɟɧɧɨɝɨ ɨɫɚɞɤɚ ɧɚ ɤɚɬɢɨɧɵ II ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
5Ɉɫɚɠɞɟɧɢɟ ɤɚɬɢɨɧɨɜ III ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ ɢɡ ɰɟɧɬɪɢɮɭɝɚɬɚ ɩɨɫɥɟ ɨɬɞɟɥɟɧɢɹ II ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
6Ɉɬɞɟɥɟɧɢɟ ɨɬ ɨɫɚɞɤɚ 5 PbSO4 ɢ ɢɫɫɥɟɞɨɜɚɧɢɟ ɨɫɬɚɜɲɟɝɨɫɹ ɨɫɚɞɤɚ ɧɚ ɤɚɬɢɨɧɵ III ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
7
8
9
10
11
Ɉɬɞɟɥɟɧɢɟ ɤɚɬɢɨɧɨɜ IV ɝɪɭɩɩɵ ɨɬ ɤɚɬɢɨɧɨɜ V ɢ VI  ɝɪɭɩɩ ɞɟɣɫɬɜɢɟɦ 6 Ɇ ɪɚɫɬɜɨɪɚ NaOH ɜ ɩɪɢɫɭɬɫɬɜɢɢ 3% ɪɚɫɬɜɨɪɚ H2O2 ɢɢɯɨɛɧɚɪɭɠɟɧɢɟɜɩɨɥɭɱɟɧɧɨɦɪɚɫɬɜɨɪɟ
ɝɪɭɩɩ ɞɟɣɫɬɜɢɟɦ 6 Ɇ ɪɚɫɬɜɨɪɚ NaOH ɜ ɩɪɢɫɭɬɫɬɜɢɢ 3% ɪɚɫɬɜɨɪɚ H2O2 ɢɢɯɨɛɧɚɪɭɠɟɧɢɟɜɩɨɥɭɱɟɧɧɨɦɪɚɫɬɜɨɪɟ
Ɉɬɞɟɥɟɧɢɟ ɢɨɧɨɜ Sb(V) ɨɬ ɞɪɭɝɢɯ ɤɚɬɢɨɧɨɜ V ɢ VI ɚɧɚ-  ɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ
ɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ
Ɋɚɡɞɟɥɟɧɢɟ ɤɚɬɢɨɧɨɜ V ɢ VI ɚɧɚɥɢɬɢɱɟɫɤɢɯ ɝɪɭɩɩ ɞɟɣ-  ɫɬɜɢɟɦ ɧɚ ɪɚɫɬɜɨɪ 8 ɤɨɧɰɟɧɬɪɢɪɨɜɚɧɧɵɦ NH3·H2O
ɫɬɜɢɟɦ ɧɚ ɪɚɫɬɜɨɪ 8 ɤɨɧɰɟɧɬɪɢɪɨɜɚɧɧɵɦ NH3·H2O
(V ɝɪɭɩɩɚ ɜ ɨɫɚɞɤɟ; VI ɝɪɭɩɩɚ ɜ ɪɚɫɬɜɨɪɟ)
ɂɫɫɥɟɞɨɜɚɧɢɟ ɨɫɚɞɤɚ 9 ɧɚ ɤɚɬɢɨɧɵ V ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ (ɢɫɤɥɸɱɚɹ Fe2+ ɢ Fe3+)
ɂɫɫɥɟɞɨɜɚɧɢɟ ɪɚɫɬɜɨɪɚ 10 ɧɚ ɤɚɬɢɨɧɵ VI ɚɧɚɥɢɬɢɱɟɫɤɨɣ ɝɪɭɩɩɵ
36
Ɍɚɛɥɢɰɚ 13
ȺɇȺɅɂɌɂɑȿɋɄȺə ɄɅȺɋɋɂɎɂɄȺɐɂə ȺɇɂɈɇɈȼ,
ɈɋɇɈȼȺɇɇȺə ɇȺ ɊȺɁɅɂɑɇɈɃ ɊȺɋɌȼɈɊɂɆɈɋɌɂ ɋɈɅȿɃ ȼɚ2+ ɂ Ag+
Ⱥɧɚɥɢɬɢ- |
|
|
|
|
|
Ƚɪɭɩɩɨɜɨɣ |
|
ɱɟɫɤɚɹ |
Ⱥɧɢɨɧɵ |
|
ɋɜɨɣɫɬɜɚ ɨɫɚɞɤɨɜ |
|
|
ɪɟɚɝɟɧɬ |
|
ɝɪɭɩɩɚ |
|
|
|
|
|
|
|
I |
SO42–, SO32–, S2O32–, |
BaCl2 |
Ɉɫɚɞɤɢ ɫɨɥɟɣ ɛɚɪɢɹ ɧɟ |
|
CO32–, AsO43–, AsO33–, |
ɢɥɢ |
ɪɚɫɬɜɨɪɢɦɵ ɜ ɜɨɞɟ, ɧɨ |
|
C2O42–, CrO42–, (Cr2O72–), |
Ba(NO3)2 |
ɪɚɫɬɜɨɪɢɦɵ ɜ ɤɢɫɥɨɬɚɯ |
|
SiO32–, BO2– (B4O72–), |
|
(ɤɪɨɦɟ BaSO4) |
|
F–, IO3–, IO4–, PO43–, |
|
|
|
C4H4O62– |
|
|
II |
Cl– , Br– , I– , CN–, |
AgNO3 ɜ |
Ɉɫɚɞɤɢ ɫɨɥɟɣ ɫɟɪɟɛɪɚ ɧɟ |
|
SCN–, C6H5COO–, S2- |
HNO3 |
ɪɚɫɬɜɨɪɢɦɵ ɜ ɜɨɞɟ ɢ ɚɡɨɬ- |
|
|
|
ɧɨɣɤɢɫɥɨɬɟ |
|
|
|
|
III |
NO3–, NO2–, CH3COO–, |
ɧɟɬ |
Ɉɫɚɞɤɢ ɫɨɥɟɣ ɛɚɪɢɹ ɢ ɫɟ- |
|
ClO4–, BrO3– |
|
ɪɟɛɪɚ ɪɚɫɬɜɨɪɢɦɵ ɜ ɜɨɞɟ |
Ɍɚɛɥɢɰɚ 14
ȺɇɂɈɇɕ, ɈȻɊȺɁɍɘɓɂȿ ȽȺɁɈɈȻɊȺɁɇɕȿ ɉɊɈȾɍɄɌɕ ɉɊɂ ȾȿɃɋɌȼɂɂ ɋɂɅɖɇɕɏ ɄɂɋɅɈɌ
Ⱥɧɢɨɧ, ɫɨɞɟɪɠɚ- |
ȼɵɞɟɥɹɸɳɢɣɫɹ ɝɚɡ |
|
|
(ɩɪɨɞɭɤɬ) |
ɋɩɨɫɨɛ ɨɛɧɚɪɭɠɟɧɢɹ |
ɳɢɣɫɹ ɜ ɪɚɫɬɜɨɪɟ |
|
|
CO32–; HCO3– |
CO2 |
ɉɨɦɭɬɧɟɧɢɟ ɢɡɜɟɫɬɤɨɜɨɣ ɜɨɞɵ |
SO32–; S2O32– |
SO2 |
Ɂɚɩɚɯ ɝɨɪɹɳɟɣ ɫɟɪɵ |
NO2– |
NO2 |
Ʉɪɚɫɧɨ-ɛɭɪɵɟ ɩɚɪɵ |
S2–; SO32–; S2O32– |
H2S |
Ɂɚɩɚɯ ɬɭɯɥɵɯ ɹɢɰ |
CH3COO– |
CH3COOH |
Ɂɚɩɚɯ ɭɤɫɭɫɚ |
Br– (ɫ ɨɤɢɫɥɢɬɟɥɟɦ) |
Br2 |
Ʉɪɚɫɧɨ-ɛɭɪɵɟ ɩɚɪɵ |
Cl– |
HCl |
ɍɞɭɲɥɢɜɵɣ ɝɚɡ, ɩɨɦɭɬɧɟɧɢɟ |
|
|
ɪɚɫɬɜɨɪɚ AgNO3 |
37
|
|
|
|
|
|
|
|
|
|
|
Таблица 15 |
|
КЛАССИФИКАЦИЯ АНИОНОВ ПО ОКИСЛИТЕЛЬНО-ВОССТАНОВИТЕЛЬНЫМ СВОЙСТВАМ |
||||||||||
|
|
Анионы-окислители |
|
|
|
Анионы-восстановители |
|
||||
|
|
Cr2O72–; AsO43–; NO2–; IO4–; IO3– |
Br – ,I – , S2–, C2O42–, AsO33–, SO32–, S2O32–, NO2– |
||||||||
|
|
|
|
Проба на присутствие в растворе |
|
|
|
|
|||
|
При действии KI появляется окраска I2 |
|
При действии I2 или KMnO4 раствор обесцвечивается |
||||||||
|
|
|
|
|
Примеры |
|
|
|
|
|
|
|
+2e + AsO43– + 2H+ |
AsO33– + H2O |
1 |
-2e + SO32– + H2O |
|
SO42– + 2H+ |
1 |
||||
|
-2e + 3I – |
[I3] – |
|
1 |
+2e + [I3] – |
3I – |
|
SO42– + 2H+ + 3I – |
1 |
||
|
AsO43– + 2H+ + 3I – |
AsO33– + H2O + [I3] – |
SO32– + H2O + [I3] – |
|
|
|
|||||
|
(в среде конц. HCl) |
|
|
-2e + C2O42– |
2CO2↑ |
|
5 |
||||
|
+6e + Cr2O72– + 14H+ |
2Cr3+ + 7H2O |
|
Mn2+ + 4H2O |
|||||||
38 |
|
1 +5e + MnO4– + 8H+ |
|
|
2 |
||||||
-2e + 3I – |
[I3] – |
2Cr3+ + 7H2O + 3[I3] – |
3 5C2O42– + 2MnO4– + 16H+ |
10CO2↑+ 2Mn2+ + 8H2O |
|||||||
|
Cr2O72– + 14H+ + 9I – |
-2e + AsO33– + 2OH – |
|
|
AsO43– + H2O |
1 |
|||||
|
|
|
|
|
– |
|
|||||
|
|
|
|
|
+2e + [I3] |
– |
3I |
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
AsO33– + 2OH – |
+ [I3] – |
|
AsO43– + 3I – |
+ H2O |
||
|
|
|
|
|
(всредеNaHCO3) |
|
|
|
|
||
|
|
|
|
|
При действии конц. H2SO4 выделяется I2 и Br2: |
||||||
|
|
|
|
|
-2e + 2I– |
|
I2 |
|
|
|
4 |
|
|
|
|
|
+8e + SO42– + 10H+ |
|
|
H2S + 4H2O |
1 |
||
|
|
|
|
|
8 I – + SO42– + 10H+ |
|
|
4I2↑ + H2S↑+ 4H2O |
|||
|
|
|
|
|
-2e + 2Br – |
Br2 |
|
|
|
1 |
|
|
|
|
|
|
+2e + SO42– + 4H+ |
|
|
SO2 + H2O |
1 |
||
|
|
|
|
|
2 Br – + SO42– + 4H+ |
|
|
Br2 + SO2 + H2O |
|
||
Ɍɚɛɥɢɰɚ 16
ɊȿȺȽȿɇɌɕ, ɂɋɉɈɅɖɁɍȿɆɕȿ ȾɅə ȾɊɈȻɇɈȽɈ ɈȻɇȺɊɍɀȿɇɂə ȺɇɂɈɇɈȼ I-III ȽɊɍɉɉ ɂ ɋɈɈɌȼȿɌɋɌȼɍɘɓɂȿ
ɉɊɈȾɍɄɌɕ ɊȿȺɄɐɂɃ
|
Ɋɟɚɝɟɧɬɵ |
ɉɪɨɞɭɤɬ ɪɟɚɤɰɢɢ, ɚɧɚɥɢɬɢɱɟɫɤɢɣ |
Ⱥɧɢɨɧɵ |
(ɭɫɥɨɜɢɹ ɪɟɚɤɰɢɢ) |
ɷɮɮɟɤɬ |
SO42– |
Ba2+ (ɜ ɫɪɟɞɟ ɦɢɧɟ- |
BaSO4Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɧɟɪɚɫɬɜɨɪɢɦ ɜ ɤɢɫ- |
|
ɪɚɥɶɧɨɣ ɤɢɫɥɨɬɵ) |
ɥɨɬɚɯ ɢ ɳɟɥɨɱɚɯ) |
|
Sr2+ (ɜ ɫɪɟɞɟ ɦɢɧɟ- |
SrSO4Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɧɟɪɚɫɬɜɨɪɢɦ ɜ ɤɢɫ- |
|
ɪɚɥɶɧɨɣ ɤɢɫɥɨɬɵ) |
ɥɨɬɚɯ) |
SO32– |
H+ |
SO2Ĺ (ɡɚɩɚɯ) |
|
[I3]– |
I– (ɨɛɟɫɰɜɟɱɢɜɚɧɢɟ ɪɚɫɬɜɨɪɚ ɣɨɞɚ) |
S2O32– |
H+ |
SO2Ĺ (ɡɚɩɚɯ) + SĻ (ɛɟɥɵɣ ɨɫɚɞɨɤ) |
|
[I3]– |
I– (ɨɛɟɫɰɜɟɱɢɜɚɧɢɟ ɪɚɫɬɜɨɪɚ ɣɨɞɚ) |
|
Ag+ (ɢɡɛɵɬɨɤ) |
Ag2S2O3Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɩɪɢ ɪɚɡɥɨɠɟɧɢɢ |
|
|
ɱɟɪɧɟɟɬ Ag2SĻ) |
ɋO32– |
H+ |
ɋO2Ĺ |
|
Mg2+ |
MgCO3Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ) |
|
ɮɟɧɨɥɮɬɚɥɟɢɧ |
ɦɚɥɢɧɨɜɨɟ ɨɤɪɚɲɢɜɚɧɢɟ |
|
|
|
ɇɋO3– |
Mg2+ (ɩɪɢ ɤɢɩɹɱɟ- |
MgCO3Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ) + ɋO2Ĺ |
|
ɧɢɢ) |
|
|
ɮɟɧɨɥɮɬɚɥɟɢɧ |
ɧɟ ɞɚɟɬ ɨɤɪɚɲɢɜɚɧɢɹ |
|
|
|
PO43– |
MgCl2; NH4OH; |
MgNH4PO4Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ) |
|
NH4Cl (ɦɚɝɧɟɡɢɚɥɶ- |
|
|
ɧɚɹ ɫɦɟɫɶ) |
|
|
Ag+ |
Ag3PO4Ļ (ɠɟɥɬɵɣ ɨɫɚɞɨɤ, ɪɚɫɬɜɨɪɢɦ ɜ |
|
|
HNO3; NH4OH) |
C2O42– |
Ca2+ |
CaC2O4Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɧɟɪɚɫɬɜɨɪɢɦ ɜ ɭɤ- |
|
|
ɫɭɫɧɨɣ ɤɢɫɥɨɬɟ) |
|
MnO4– (ɜɫɪɟɞɟH2SO4) |
CO2Ĺ (ɨɛɟɫɰɜɟɱɢɜɚɧɢɟ ɪɚɫɬɜɨɪɚ KMnO4) |
39
|
|
|
ɉɪɨɞɨɥɠɟɧɢɟ ɬɚɛɥ. 16 |
|
|
|
|
|
Ɋɟɚɝɟɧɬɵ |
|
ɉɪɨɞɭɤɬ ɪɟɚɤɰɢɢ, ɚɧɚɥɢɬɢɱɟɫɤɢɣ |
Ⱥɧɢɨɧɵ |
(ɭɫɥɨɜɢɹ ɪɟɚɤɰɢɢ) |
ɷɮɮɟɤɬ |
|
AsO43– |
MgCl2+ NH4OH + |
MgNH4AsO4Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ) |
|
|
NH4Cl (ɦɚɝɧɟɡɢɚɥɶ- |
|
|
|
ɧɚɹ ɫɦɟɫɶ) |
|
|
|
Ag+ |
|
Ag3AsO4Ļ (ɲɨɤɨɥɚɞɧɵɣ ɨɫɚɞɨɤ, ɪɚɫɬɜɨɪɢɦ |
|
|
|
ɜ HNO3 ɢ NH4OH) |
|
I– (ɜ ɫɪɟɞɟ HCl |
ɜ |
[I3]– – ɤɪɚɫɧɨ-ɮɢɨɥɟɬɨɜɵɣ ɰɜɟɬ ɯɥɨɪɨɮɨɪɦ- |
|
ɩɪɢɫɭɬɫɬɜɢɢ CHCl3) |
ɧɨɝɨ ɫɥɨɹ |
|
AsO33– |
S2– |
|
As2S3Ļ (ɠɟɥɬɵɣ ɨɫɚɞɨɤ, ɧɟɪɚɫɬɜɨɪɢɦ ɜ |
|
|
|
ɤɨɧɰ. HCl; ɪɚɫɬɜɨɪɢɦ ɜ NH4OH) |
|
Ag+ |
|
Ag3AsO3Ļ (ɠɟɥɬɵɣ, ɪɚɫɬɜɨɪɢɦ ɜ NH4OH ɢ |
|
|
|
ɤɨɧɰ.HNO3) |
|
[I3]– (ɜɫɪɟɞɟNaHCO3) |
I– (ɨɛɟɫɰɜɟɱɢɜɚɧɢɟ ɪɚɫɬɜɨɪɚ ɣɨɞɚ) |
|
CrO42– |
ȼɚ2+ |
|
BaCrO4Ļ (ɠɟɥɬɵɣ ɨɫɚɞɨɤ) |
(Cr2O72–) |
I– (ɜ ɫɪɟɞɟ HCl |
ɜ |
[I3]– – ɤɪɚɫɧɨ-ɮɢɨɥɟɬɨɜɵɣ ɰɜɟɬ ɯɥɨɪɨɮɨɪɦ- |
|
ɩɪɢɫɭɬɫɬɜɢɢ CHCl3) |
ɧɨɝɨ ɫɥɨɹ |
|
SiO32– |
Ba2+ |
|
BaSiO3Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɩɪɢ ɞɟɣɫɬɜɢɢ ɤɢɫ- |
|
|
|
ɥɨɬ ɪɚɡɥɚɝɚɟɬɫɹ ɫ ɨɛɪɚɡɨɜɚɧɢɟɦ ɇ2SiO3Ļ) |
ȼ4Ɉ72– |
H2SO4; C2H5OH |
|
(C2H5O)3B – ɨɤɪɚɲɢɜɚɟɬ ɩɥɚɦɹ ɜ ɡɟɥɟɧɵɣ |
|
|
|
ɰɜɟɬ |
|
|
|
|
F– |
Ba2+ |
|
BaF2Ļ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɪɚɫɬɜɨɪɢɦ ɜ NH4OH, |
|
|
|
ɜ ɦɢɧɟɪɚɥɶɧɵɯ ɤɢɫɥɨɬɚɯ) |
|
|
|
|
|
H2SO4, (SiO2·H2O) |
|
H2SiO3Ļ (ɝɟɥɶ) |
Cl– |
Ag+ |
|
AgClĻ (ɛɟɥɵɣ ɨɫɚɞɨɤ, ɪɚɫɬɜɨɪɢɦ ɜ |
|
|
|
(NH4)2ɋɈ3 ɢ NH4OH) |
Br– |
Ag+ |
|
AgBrĻ (ɠɟɥɬɵɣ ɨɫɚɞɨɤ, ɱɚɫɬɢɱɧɨ ɪɚɫɬɜɨ- |
|
|
|
ɪɢɦ ɜ NH4OH) |
|
Cl2 (ɜ ɤɢɫɥɨɣ ɫɪɟɞɟ |
|
Br2 (ɯɥɨɪɨɮɨɪɦɧɵɣ ɫɥɨɣ ɨɤɪɚɲɢɜɚɟɬɫɹ ɜ |
|
ɜ ɩɪɢɫɭɬɫɬɜɢɢ |
|
ɛɭɪɵɣ ɰɜɟɬ) |
|
CHCl3) |
|
|
40
