
Лабораторные работы по химии
..pdf
|
|
, |
|
|
|
|
|
( |
|
|
), |
|
|
. |
|
|
|
|
|
|
|
|
|
: |
л |
|
хи и |
а ии ( . . и |
и |
ал ии и |
) а и и |
л |
|||
|
а ал |
и |
и ( |
ал |
ии) а |
их |
а |
ии |
и |
а и и |
|
х а и ( |
и |
а и ) |
|
а. |
|
|
|
|
|
|
|
: |
|
|
|
|
1. |
|
|
|
|
|
|
|
|
|
( |
|
|
|
|
|
). |
|
|
|
|
1. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
: 2МР( ) + |
2( ) = 2МР ( ) + |
|
( |
), |
|
, |
|
|
|
|
|
2 МР |
|
|
|
|
|
-393,5 |
-601,8 |
/ |
л , |
|
МР |
|
( |
) |
|
0 ( |
|
|
). |
|
|
|
|
|
|
|
: |
|
|
|
|
|
|
|
|
∆ |
0 = 2∆ |
0МР |
- ∆ 0 |
2 |
= 2(-601,8) + 393,5 = -810 |
|
|
||
|
|
|
2. |
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
, |
|
|
|
|
, |
|
|
|
, |
|
|
|
. |
|
|
|
|
|
( |
, |
|
, |
, |
), |
|
|
|
|
|
|
. |
|
|
|
|
|
|
|
2. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N2 ( ), |
|
|
|
|
|
|
|
|
: |
|
|
|
|
1) |
( |
) + 2N2 |
( ) = 2 + 2N2( ), |
∆ |
01 = -557,5 |
; |
|||
|
2) |
( |
) + |
2( ) = |
2( ), |
|
∆ |
02 = -393,5 |
. |
|
:
|
|
|
|
|
|
|
|
N2 |
|
: |
|
|
|
|
|
|
|
3) N2( ) + 1/2 2 = N2 ( ), |
|
∆ 03 = ? |
|
|||||
|
|
|
(3) |
|
|
|
(1) |
(2), |
|
(1) |
|
(-1/2), |
|
|
|
(2) |
|
|
|
|
|
|
: |
|
|
|
1) 1/2 |
2( ) + N2( ) = 1/2 ( |
) + N2 |
( ); |
|
|
|||
2) 1/2 ( |
) +1/2 2( ) = 1/2 |
2( ) |
|
|
|
|||
|
|
|
|
|
|
: |
|
|
3) N2( ) + 1/2 2 = N2 ( ) |
|
|
|
|
||||
|
|
|
∆ |
03 |
|
|
|
|
|
|
|
|
: |
|
|
|
|
∆ |
03 = -1/2 ∆ |
01 + 1/2∆ |
02 = 1/2·557,5 - 1/2·393,5 = 82,0 |
/ л |
||||
|
|
|
|
|
|
|
|
( |
|
|
|
) |
|
|
|
|
: |
|
|
|
|
H10 H 2980 T CP dT, |
|
|
||
|
|
|
|
|
298 |
|
|
|
HT0 - |
|
|
|
|
|
, |
|
|
H2980 - |
|
|
|
= 298 К. |
|
|
|
|
∆ |
|
|
|
|
|
: а |
+ b = |
+ НD |
: |
|
|
CP |
cCP (C ) |
dCP (D) |
aCP ( A) |
bCP (B) , |
|
|
( ),, |
( ), |
( ), (D) – |
|
|
|
|
|
, |
, |
, D; a, b, c, d – |
|
|
|
|
|
|
|
|
|
. |
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
, |
|
|
. |
|
, |
|
, |
, |
|
|
. |
|
|
|
|
1 |
|
1 К. |
|
|
|
(1/2),
;
.
,

,
1 л |
1 К. |
|
|
– |
, |
|
1 |
2. |
|
|
Q |
|
C |
T2 T1 |
: Cи dQdT ,
dQ, |
Н |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
T2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
C |
|
|
Cи |
dT |
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
T |
T |
|
|
|
|
||||||
|
|
|
|
|
|
|
2 |
1 T |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
V. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
= const Qp = ∆H, |
|
|
V = const QV = ∆U |
|
|||||||||||
|
CP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
dH |
|
|
|
|
|
|
|
CP |
|
dU |
|
|
||
|
|
|
dT |
P |
|
|
|
|
|
|
|
dT |
V |
|
||
|
V |
|
|
|
|
|
|
|
|
|
|
|
: |
|
|
|
|
|
|
|
|
|
|
- |
V = R, |
|
|
|
|
|
|||
R = 8,314 |
/ л ∙К ( |
|
|
|
|
|
|
|
|
|
|
|
|
). |
|
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
: 1) |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
( |
) ∆ <0 |
2) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, . . |
( |
|
|
|
|
|
|
|
|
|
|
|
|
). |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
S: |
|
|
|
|
|
|
|
|
S = RlnW, |
|
|
|
|
|
|||
R – |
|
|
|
|
|
|
|
|
|
|
|
; |
|
|
|
|
W |
– |
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
, |
|
|
|
|
( |
|
|
|
|||
|
|
|
|
|
|
|
|
), |
|
|
|
|
|
( |
, |
|
|
, |
|
|
|
). |
|
|
|
|
|
Э |
|
|
|
|
|
|
|
|
|
|
( |
|
|
|
|
|
|
, |
|
|
|
|
|
|
, |
||
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
(∆S>0). |
|
|
|
||
|
|
, |
|
|
|
|
|
, |
|
|
|
|
|
|
( |
|
|
|
|
, |
|
, |
|
, |
|||
|
|
|
|
|
|
|
|
|
|
(∆S<0). |
|
|
|
|
|
3. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
: |
|
|
|
|
|
|
|
|
|
1) N 4N |
3( ) = N2 |
( ) + 2 2 ( ); |
|
|
|
||||||||
2) 2 2( ) + |
2( ) = 2 |
2 |
( ); |
|
|
|
|
|
|||||
3) 2 2( ) + |
2( ) = 2 |
2 |
( ). |
|
|
|
|
|
|||||
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) |
1 |
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
, ∆S>0. |
(2) |
(3) |
||||
|
, |
|
|
|
|
|
|
|
|
|
|
, |
|
|
∆S3 |
|
|
|
|
|
|
|
|
|
|
|
|
SH 2O( ) |
SH 2O( ) . |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
S |
|
л |
, |
|
|
|
- S |
|
и |
|
|
|
|
л |
|
|
|
|
и |
|||||
|
|
|
|
|
|
|
|
|
|
|
|
||
∆ |
л ∆ |
и . |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
T2 |
|
|
|
||
|
|
|
|
|
|
|
: S T |
CP |
dT ( |
1 |
|
|
|
|
|
|
|
|
|
|
T |
|
|
||||
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
)
,
,
,
),
3
∆S2 < 0 ∆S3 < 0.
, ∆S2,
:
,
.
).

S Rln V2 ,
V1
S Rln p1 .
p2
,
:
n1 n2 – V1 V2 –
,
> 0.
V2>V1, |
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
: |
S T2 |
Cp |
dT Rln |
V2 |
. |
|
|
||||||
T |
V |
|
|
|||||||||
T |
|
|
|
|
|
|
1 |
|
|
|
||
1 |
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
V |
V |
|
|
V |
V |
|
||||||
S n1R ln |
|
1 |
2 |
n2 R ln |
|
1 |
2 |
, |
||||
|
|
|
V |
|
|
V |
||||||
|
|
|
|
1 |
|
|
|
|
|
|
2 |
|
; |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
S0 |
|
|
|
|
|
|
|
|
|
|
|
|
298 |
. |
|
|
|
|
|
|
|
|
|
|
|
/ л ∙К. |
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
∆ : |
|
|
|
|
|
|
(∆S) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
||
. |
|
|
|
|
|
|
|
|
|
|
|
|
(Q = 0, |
= 0, U = |
|
st) |
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
, . . ∆S |
, . . |
|
|
|
|
∆S = 0. |
|
|
|
|
|
||
, |
|
|
|
|
|
|
|
|
|
|
|
|
, . . |
(∆ < 0), |
, . . |
|
(∆S > 0). |
|
|||||
|
, |
|
|
|
|
|
|
|
(∆ < 0, ∆S > 0) |
|
|
|
|
|
|
||
, |
, |
|
|
|
|
|
|
|
: |
|
|
|
|
|
|
|
|
|
G = |
|
|
– S. |
|
|||
- |
|
|
|
|
|
|
|
|
∆G = ∆ - ∆S. |
|
|
|
|
|
|
|
|
Э |
|
|
|
|
|
|
|
1 |
/ |
л ; |
|
|
|
∆G0 |
|
||
|
|
|
|
. |
|
|||
|
|
|
(∆G < 0). |
|
||||
, |
|
|
|
|
|
|
|
|
(Gmin). |
|
|
|
|
|
|
|
|
(∆G = 0). |
|
|
|
|
|
|
|
|
|
|
|
|
, |
, |
|||
. |
|
|
|
, |
|
|||
, |
|
|
|
|
|
|
|
. |
/ |
|
|
|
: |
|
|||
|
KC |
|
CB |
. |
|
|||
|
|
|
|
|||||
|
|
|
|
CA |
|
|||
|
|
|
|
, |
|
|||
|
|
|
|
|
|
|
|
: |
|
KP |
|
PB |
|
|
|||
|
P |
|
||||||
|
|
|
|
|
A |
|
||
– |
|
|
|
|
|
|
|
. |
|
– |
|
|
|
|
|
|
, |
|
|
|
, |
|
|
|
|
. |
:
|
|
|
GT0 RT ln KP .\ |
|
|
|
|
|
|
1.2 |
|
|
|
|
1. |
|
|
|
? |
|
|
2. |
|
|
|
? |
|
|
|
? |
|
|
|
|
|
|
|
|
|
|
|
|
3. |
|
? |
|
|
|
|
4. |
|
? |
|
, |
|
|
5. |
|
|
? |
|
|
|
|
|
|
|
||
|
6. |
|
. |
|
|
|
|
7. |
|
, |
|
. |
|
|
8. |
|
? |
|
|
|
|
9. |
|
|
|
? |
|
|
10. |
|
|
? |
|
|
|
11. |
|
|
|
|
? |
|
12. |
|
|
, |
|
, |
|
, |
|
? |
|
|
|
|
13. |
|
? |
|
|
|
|
14. |
|
? |
|
|
|
|
15. |
|
|
) |
|
|
|
|
U |
T S |
|
||
|
, |
) |
? |
|
|
|
|
16. |
|
|
|
|
|
) |
0, ) |
|
|
|
|
|
S |
S 0. |
|
|
|
||
|
17. |
|
? |
|
|
|
|
|
|
|
|
|
|
1.3 |
|
|
|
|
|
“ |
|
|
|
|
|
|
” |
: |
|
. |
|
|
( |
|
|
) |
|
|
. |
|
|
: |
|
, |
, |
|||
|
|
|||||
|
(1 |
), |
(2 |
) |
, |
|
. |
|
|
|
|
|
|
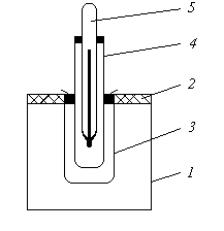
( 1) |
1 ( |
), |
2 |
. |
|
3, |
|
4 |
5.
|
|
|
|
|
1.1 – |
|
, |
|
|
|
|
. |
|
|
|
. |
|
|
, |
, |
|
|
|
|
|
|
|
1. |
|
|
|
|
, |
|
|
|
|
|
|
|
. |
|
|
|
: |
. |
|
|
|
) |
|
|
|
|
|
|
|
|
|
: |
– 40 |
, |
20 |
NaOH. |
|
3 |
|||
. |
|
|
|
|
1 |
|
. |
. |
|
|
|
NaOH |
|
|
5–10 |
2 3 |
, |
|
||
30 |
2 |
3 |
. |
|
||
,
, |
– |
. |
. 1.2 |
|
. |
|
|
( |
). |
Э |
– |
|
||
|
. |
|
||
|
|
|
|

|
|
|
|
2 – |
|
|
|
|
|
|
|
|
|
|
|
|
= |
– . |
|
|
|
|
|
|
|
|
, |
1 |
|
|
1 |
||||
|
|
. |
|
|
|
: |
|
|
|
|
|
|
|
|
|
H2SO4+NaOH=NaHSO4+H2O, H1. |
). |
|
|||||
|
|
) |
. |
|
|
( |
1 |
|
|||
|
|
20 |
|
NaOH, |
|
|
|||||
|
|
, |
|
|
|
|
|
|
|||
|
|
|
NaOH ( |
|
|
|
). |
|
|
||
|
|
|
|
. |
|
|
|
||||
|
|
: |
|
|
|
|
H2. |
|
|
||
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
NaHSO4+NaOH=Na2SO4+H2O, |
|
|
|||||
|
|
|
: |
|
( |
1 |
). |
|
|
||
|
|
|
|
|
|
|
|
|
|
||
|
|
1. |
|
|
|
|
|
|
|
. |
|
|
|
2. |
|
|
. |
|
|
||||
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
V, |
|
|
, 0 |
|
d, / |
|
/ |
|
H, |
|
|
H2SO4 |
|
NaOH |
|
CV1 |
|
||||
1 |
|
40 |
20 |
|
|
1,031 |
|
1,54 |
|
|
|
|
|
– |
20 |
|
|
1,034 |
|
1,65 |
|
|
|
2 |
|
40 |
40 |
|
|
1,034 |
|
1,65 |
|
|
|
|
|
2. |
|
|
|
|
|
|
. |
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
: |
3 |
. |
|
, |
|
|||
|
|
|
40 |
. |
|
|
|||||
|
|
|
|
|
|
. |
|||||
|
|
|
|
|
|
|
|
|
|
||
NaOH
.

,
:
1.
2.
( 1, 3. 2, 3)
V – d –
– – 0 ; 25 –
4.
3 5.
6.
7.
.
, |
1 |
|
2 |
|
: |
H3 |
|
H2SO4+2NaOH=Na2SO4+2H2O, |
. |
||
1 |
|
2 |
|
|
. |
|
|
2 |
. |
|
|
: =V d C T 25 0,001,
,;
|
, / ; |
|
); |
|
|
|
|
, |
/( |
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
; 0,001 – |
|
1+ |
|
|
( |
). |
|
2 |
|
1+ |
. |
||
|
. |
|
|
2 |
||
|
|
|
|
|
|
|
H |
3 |
( |
H |
H |
2 |
) |
100% . |
K = |
|
|
H13 |
|
|
|||
|
|
|
|
|
|
