
3139
.pdf, |
№ 2 (17), 2018 |
621.377
|
|
. . |
|
*, . . |
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
я |
я, 394006, . |
, |
. 20- |
я |
я я,84 |
|
|
* |
я |
: |
|
. . E-mail:b.a.spiridonov@yandex.ru |
|
|||
|
|
|
|
|
|
|
|
, |
|
|
- |
|
– |
|
( |
). |
, |
|
|
|
|
, |
|
|
|
- |
|
|
|
. |
|
1,5 |
2 |
/ |
|
|
|
|
|
, |
, |
|
|
- |
|
. |
|
|
|
|
|
|
|
|
|
|
. |
|
|
( |
1,0 1,7) |
- |
|
β–Co, |
( |
3,0) – |
α–Co. |
|
|
|
|
|
: |
|
, |
|
, |
|
|
, |
|
, |
, |
, |
|
, |
|
|
|
© |
. ., |
. ., 2018 |
100
, |
№ 2 (17), 2018 |
INVESTIGATION OF THE PROCESS OF ELECTRODEPTION OF
COBALT COATINGS FROM SULFURIC ELECTROLYTEES WITH
MONOETANOLAMINE
B.A. Spiridonov*, M.A. Zavalishin
Voronezh State Technical University, Russian Federation, 394006, Voronezh, the 20th Anniversary of October, 84
*Corresponding author: Spiridonov B.A., E-mail: b.a.spiridonov@yandex.ru
The kinetics of electroreduction of cobalt from sulphate electrolytes containing a surfac- tant-monoethanolamine (MEA) is studied. It has been established that cobalt does not precipitate from the electrolyte without MEA, since a film of cobalt hydroxide is formed on the surface of the cathode. In the presence of MEA from 1.5 to 2 ml / l, the quality of cobalt coatings improves significantly, gloss appears, and microhardness increases. X-ray diffraction method established the relationship between the phase composition of cobalt coatings and the pH of the electrolyte. Of acid electrolytes (pH 1.0 and 1.7), β-Co is precipitated, and less acidic (pH 3.0) is mainly α-Co.
Keywords: electrolysis, cobalt coatings, sulphate electrolyte, monoethanolamine, kinetics, structure, internal stresses, microhardness
. |
|
|
|
|
|
, |
, |
|
- |
, |
|
|
|
- |
|
|
. |
|
- |
|
|
|
|
- |
, |
|
– |
|
- |
Д1-6]. |
|
|
|
|
, |
|
|
|
|
. |
|
|
, |
, |
, |
, |
|
|
Д4Ж. |
101
, |
№ 2 (17), 2018 |
|
|
|
, |
- |
, |
|
|
|
|
. |
|
|
|
- |
, |
|
|
|
|
ik > 10 / |
2. |
|
|
- |
|
- |
|
, |
|
|
|
|
Д7Ж. |
- |
|
, |
|
|
ik |
Д9,10]. |
|
|
|
- |
( |
, |
, |
.) |
|
, |
|
|
. |
|
|
|
|
|
Д2, |
3Ж, |
. |
|
|
- |
|
|
|
CoSO4 |
|
. |
|
|
|
|
( ), |
|
|
- |
. |
|
. |
- |
[6], |
|
|
|
, |
, |
, |
|
Д6Ж. |
|
|
|
|
|
|
- |
. |
|
, |
- |
, |
|
|
- |
Д5Ж. |
|
, |
, |
3000 . |
|
|
|
, |
|
, |
- |
102
, |
№ 2 (17), 2018 |
|
|
|
|
|
|
|
|
|
|
|
. |
|
|
|
|
|
|
, |
|
|
|
|
- |
|
|
|
|
( |
). |
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
: α- |
|
|
|
|
|
|
|
|
|
( |
|
|
a = 2,5063 |
= 4,0795 й) |
β- |
|
- |
|
|
||
(a = 3, 5441 й). |
|
|
|
|
|
|
|
|
|||
|
|
|
4500 . |
|
|
|
4770 |
|
|
|
- |
|
β- |
|
Д6Ж. |
|
|
|
|
|
|
|
- |
|
, |
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
Д4Ж. |
|
|
|
|
|
|
- |
|
|
|
|
, |
, |
|
|
|
|
|
. |
|
|
, α-Co |
|
, |
|
|
500 |
/ |
CoSO4 |
45 |
|
/ |
3BO3 |
5, |
600 |
ik = 1, 5 / |
2. |
|
|
|
|||
= 10 / |
|
β-Co. |
|
|
1,3-1,9, |
|
|
200 |
ik |
||
2 |
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
. |
|
, |
|
β-Co |
|
- |
|
|
|
|
, |
, |
|
, |
α-Co |
|
|
|
|
|
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
- |
|
|
|
|
|
|
|
( |
) |
|
|
|
|
|
ik |
|
|
|
|
|
Д8, 19]. |
|
||
|
|
|
– |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
CoSO4 |
|
( / |
): - 2, 10, 30 |
100. |
|
|
NaCl – 5 |
/ |
H3BO3 – 15 |
/ . |
|||
|
|
|
|
0,5 ÷ 3 |
/ ; t = 20-250 C; |
2-2,5; |
- |
||||
|
|
|
– 2 |
8 / |
2 . |
– |
Co, |
|
– |
. |
|
103

, |
№ 2 (17), 2018 |
( |
) |
|
|
|
. |
|
- |
(2 |
/ ) |
-5827 |
, |
|
. |
|
- |
, |
|
– |
. |
|
|
-3 |
100 . |
|
|
|
, |
|
α |
|
|
10 |
|
/ 2. |
|
|
. |
1 |
|
|
|
, |
|
- |
|
|
( 2 |
/ ) |
|
|
CoSO4. |
, |
|
|
|
|
CШSτ4 |
|
, |
|
|
|
|
|
|
|
|
. |
, |
|
|
|
|
CШSτ4 |
100 2 / ( |
4) |
|
(ΔE) |
|
- |
0,46 (i |
= 2 |
/ 2). |
|
, |
|
|
|
- |
( |
CoSO4 = 2 / |
) |
|
|
|
, |
- |
|
|
|
|
|
|
|
Co(OH)2 ( |
Co(OH)2 |
= |
1,6·10-15) [10]. |
|
|
|
|
|
|
|
|
. 1.
, |
. |
CШSτ4 ( / ): 1 – 100; 2 – 50; 3 – 20; 4 – 2.; 2 / |
; pH 2,1 |
104

, |
№ 2 (17), 2018 |
|
|
|
|
|
|
|
( |
.2) |
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
. |
|
- |
, |
|
|
|
|
( |
0,5 |
2 |
/ |
|
= 1 |
/ |
- |
|
|
|
|
|
2 - 4), |
|
|
i |
2 |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0,22 |
. |
, |
, |
|
|
|
|
|
|
|
- |
|
|
|
|
Co2+ |
|
NH2, |
|
|
|
|
|
|
|
|
Д11Ж. |
|
|
|
|
|
|
, - |
|||
, |
3 |
/ |
|
|
5 |
|
|
|
|
|
|
- |
|
|
, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
. |
|
|
|
|
|
, |
|
|
|
( |
Co) |
ik, |
|
pH |
||
/ |
. |
|
ik |
2 |
8 / |
CoSO4 100 |
/ |
45 |
|
2 |
||
|
|
Co |
|
|
62% |
|||||||
|
|
|
|
|
|
2 |
|
|
|
|
|
|
( = 30 |
, pH 2,1). |
|
|
|
CoSO4 |
50 |
/ , |
|
Co |
|||
|
31% (ik |
=2 / |
2, |
= 30 |
, pH 2,1). |
|
|
|
|
|
||
|
|
|
2-4 |
/ |
2, |
|
|
pH (2,5) |
|
|
|
|
|
|
|
, |
|
pH > 3 – |
. |
|
|
|
|
|
|
. 2. |
, |
, |
2 / |
CШSτ4, pH 2,1. |
|
( / ): 1- |
; 2 – 0,5; 3 – 1,0; 4 – 2,0; 5 – 3,0 |
|
-
, |
CoSO4 100 / , |
- 2 / .
105
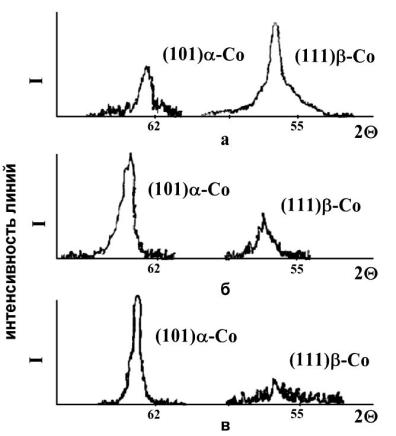
, |
№ 2 (17), 2018 |
|
, |
Co- |
, |
pH (1,0; 1,7; 3,0) |
- |
α- |
(α–Co) ( .2), |
. - |
β-Co |
, |
|
(pH 1,0 |
1,7). |
|
|
. 3. |
|
|
, |
α- β-Co, |
|
|
|
|
|
pH: |
|
– 1,0; |
– 1,7; |
– 3,0. |
CoSO4 100 / |
2 / |
|
. 3 |
, |
|
, |
pH |
1,7 |
β- |
Co |
, |
pH 1,0, |
pH |
|
3,0 |
|
|
, |
- |
|
|
β–Co. |
|
|
|
|
|
|
|
CoSO4 100 / |
pH 2,5-3,0 |
|
|
|
|
, |
pH > 4 |
|
|
|
. |
|
- |
|
|
|
|
( ). |
|
106
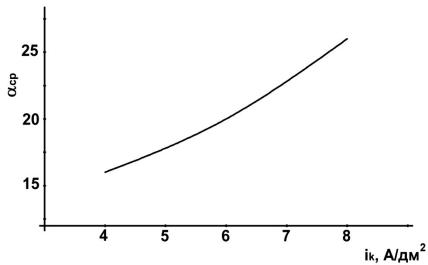
, |
№ 2 (17), 2018 |
. 4. |
(α ) |
і . |
|
: CoSO4 -100 / , |
-2 / |
, |
|
4 |
8 A/ |
|
|
( |
. |
4). |
|
i |
2 |
|
1,6 |
, |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
. |
- |
|
|
і |
|
|
|
|
|
|
|
- |
Д12]. |
|
|
|
|
|
|
|
|
. |
|
Т |
1 |
|
4 |
/ |
2 |
|
- |
|
|
|
|
|
|
|
|
|
|
0,83·105 |
1,42·105 |
|
, . . |
з 1,7 |
|
. |
3000 |
||
|
|
|
|
|
|
|
, |
|
|
|
. |
Т |
> 1,5 |
|
, |
|
, |
, |
- |
|
|
/ |
2 |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1-2 |
|
. |
|
|
|
|
|
|
|
|
|
, |
|
- |
|
|
|
|
|
|
|
|
|
- |
c |
|
(100 / |
oSO4) |
|
|
- |
|||
- |
– |
|
|
|
|
|
1,5-2 |
/ |
, |
|
|
|
|
|
|
|
2-5 / |
2. |
|
107
, |
№ 2 (17), 2018 |
1. |
. ., |
|
|
. . |
|
|
|
|
. |
.: |
- |
|
|
|
. 1957. 647 . |
|
|
|
|
|
|
||
2. |
|
|
|
/ |
|
. . . |
|
. |
.: |
, |
|
1975. 552 . |
|
|
|
|
|
|
|
|
|
|
|
3. Moral F.R. [Electroplating of cobalt and cobalt alloys a review]. Plating, |
|||||||||||
1967, vol. 5, no 6, P. 693-701. |
|
|
|
|
|
|
|
|
|||
4. |
. |
|
|
|
|
|
|
|
|
|
|
. .: |
|
|
, |
. |
., 1983. 96 . |
|
|
|
|
||
5. |
. . |
|
|
|
|
// |
|
|
|
|
- |
|
. 2004. |
.12. № 1. .16-18. |
|
|
|
|
|
|
|||
6. |
. |
|
|
|
. |
.: |
, 1996. |
.2. 836 . |
|
||
7. |
. ., |
|
. ., |
|
. ., |
|
. . |
|
|
|
- |
|
|
. |
: |
|
, 1978. 267 . |
|
|
|
|
||
8. |
|
. ., |
|
|
. ., |
|
|
. ., |
|
|
. . |
|
|
|
|
|
|
|
// |
|
|
|
- |
|
|
|
|
|
|
. |
. |
. |
. |
, |
2006. |
. 97-100. |
|
|
|
|
|
|
|
|
|
|
|
9. |
№ 2340709 |
, |
|
23 b 5/08. |
|
|
|
|
|
||
|
/ |
|
. ., |
|
|
. ., |
|
|
. . ( |
). № |
|
20077110620/02; |
. 22.03.2007.; |
|
. 10.12.2008, |
. № 34. 2 . |
|
||||||
10. |
. . |
|
|
|
|
|
|
. |
.: |
. |
|
1979. 480 . |
|
|
|
|
|
|
|
|
|
|
|
11. |
|
. |
., |
. |
., |
|
. . |
|
|
|
|
|
|
|
(II) |
S- |
|
|
|
|
|
|
|
// |
. |
|
. |
|
. 2008. |
.51, №11. |
. 7-10. |
|
|||
12. |
|
. . |
|
|
|
|
|
|
|
|
- |
|
- |
|
|
|
|
|
// |
|
|
|
|
. 2003. . 76. . 67-72. |
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
– |
. |
. |
, |
|
|
|
|
- |
|
|
|
– |
|
|
|
111, |
|
|
|
|
108

, |
№ 2 (17), 2018 |
543.4:691
|
. . |
, |
. . |
, . . |
, . . |
|
, |
|
|
|
|
|
|
|
. . |
* |
|
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
я |
|
я, 394006, . |
, . 20- |
я |
я я, 84 |
|
|
* |
я |
: |
|
|
|
, E-mail: robi57@mail.ru |
|
||
|
|
|
|
|
|
|
|
RGB |
- |
|
( |
I 42,5 |
), |
, |
, |
|
|
, |
|
|
, |
|
|
|
|
, |
|
|
- |
|
|
|
|
|
. |
|
|
|
|
|
5-30%. |
|
|
|
|
|
|
, |
|
|
|
|
|
, |
|
|
|
|
|
Pixel |
Picker |
1.3.0.23. |
|
, |
|
|
|
|
|
|
|
|
|
|
, |
|
|
|
- |
|
|
. |
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
, |
|
|
|
|
|
, |
, |
|
|
|
- |
|
|
|
|
|
|
. |
|
|
- |
|
|
|
|
|
. |
|
|
|
|
|
|
: |
|
, |
|
, |
|
RGB, |
, |
|
|
, |
|
|
, |
|
|
|
|
© |
. ., |
. ., |
. ., |
. ., |
. ., 2018 |
109
