
PS-2020a / part16
.pdf
DICOM PS3.16 2020a - Content Mapping Resource Page 171
Table TID 3108. Intervention
|
NL |
Rel with Parent VT |
Concept Name |
VM |
Req ConditionValue Set Constraint |
||
|
|
|
|
|
|
Type |
|
1 |
|
|
CODE |
EV (122090, DCM, |
1 |
M |
DCID 3412 |
|
|
|
|
"Intervention Action") |
|
|
“Intervention Actions |
|
|
|
|
|
|
|
and Status” |
2 |
> |
HAS |
CODE |
EV (363704007, SCT, |
1 |
M |
DCID 3604 “Arterial |
|
|
PROPERTIES |
|
"Procedure site") |
|
|
Lesion Locations” |
3 |
>> |
HAS CONCEPT CODE |
EV (106233006, SCT, |
1 |
U |
DCID 3019 |
|
|
|
MOD |
|
"Topographical modifier") |
|
|
“Cardiovascular |
|
|
|
|
|
|
|
Anatomic Location |
|
|
|
|
|
|
|
Modifiers” |
4 |
> |
HAS |
TEXT |
EV (121154, DCM, |
1 |
M |
Up to 3 numeric |
|
|
PROPERTIES |
|
"Intervention attempt |
|
|
characters |
|
|
|
|
identifier") |
|
|
|
5 |
> |
HAS |
CODE |
EV (116682006, SCT, "Uses |
1-n |
U |
BCID 3411 |
|
|
PROPERTIES |
|
Equipment") |
|
|
“IntracoronaryDevices” |
6 |
>> |
HAS CONCEPT CODE |
EV (122111, DCM, "Primary |
1 |
MC IF DeviceDCID 230 “Yes-No” |
||
|
|
MOD |
|
Intervention Device") |
|
|
is Primary |
|
|
|
|
|
|
|
for this |
|
|
|
|
|
|
|
Lesion |
7 |
> |
HAS |
NUM |
DCID 3425 “Intervention |
1-n |
U |
|
|
|
PROPERTIES |
|
Parameters” |
|
|
|
8 |
> |
HAS |
IMAGE |
BCID 7003 “Diagnostic |
1 |
U |
|
|
|
PROPERTIES |
|
Imaging Report Purposes of |
|
|
|
|
|
|
|
Reference” |
|
|
|
9 |
> |
|
INCLUDE DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
|
|
Qualifiers” |
|
|
|
Content Item Descriptions |
|
|
|
|
|
||
Row 4 |
Intervention attempt Identifier is specified as a numeric text string, and shall be treated as the ordinal of the |
||||||
|
|
recorded attempted intervention within this procedure (i.e., "1" for the first attempted intervention, "2" for the |
|||||
|
|
second, etc.). |
|
|
|
|
|
TID 3109 Measurements
The Measurements Template allows recording of significant measurements, such as vital signs, laboratory results, hemodynamic measurements, or quantitative analysis measurements. These measurements are often quoted from another source, which would be documented in the included TID 3010 “Log Entry Qualifiers”.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
|
Table TID 3109. Measurements |
|
NL |
RelwithParent |
VT |
Concept Name |
VM |
Req TypeConditionValue Set |
|
|
|
|
|
|
|
|
Constraint |
1 |
|
|
NUM |
|
1 |
U |
|
2 |
> |
|
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
|
|
Qualifiers” |
|
|
3 |
> |
HAS |
INCLUDE |
DTID 310 “Measurement |
1 |
U |
|
|
|
PROPERTIES |
|
Properties” |
|
|
|
- Standard -

Page 172 |
|
DICOM PS3.16 2020a - Content Mapping Resource |
|
|||
|
NL |
RelwithParent |
VT |
Concept Name |
VM |
Req TypeConditionValue Set |
|
|
|
|
|
|
Constraint |
4 |
|
CODE |
|
1 |
U |
|
5 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
|
Qualifiers” |
|
|
TID 3110 Impressions or Findings
The Impressions or Findings Template allows the recording of unconfirmed (provisional) impressions or findings noted during the procedure. It is not intended to convey the Cath Lab Clinical Report (the formal report from the performing physician), although it may be used (like any Procedure Log entry) for the subsequent construction of the Cath Lab Clinical Report.
A finding that is supported by a specific image frame may reference that image in the INFERRED FROM / IMAGE row of the included
TID 3010 “Log Entry Qualifiers” Template.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
|
Table TID 3110. Impressions or Findings |
|
NL Rel with Parent VT |
Concept Name |
VM |
Req Condition Value Set |
|||
|
|
|
|
|
|
Type |
Constraint |
1 |
|
|
CODE |
EV (121071, DCM, "Finding") |
1 |
U |
BCID 3728 “Cath |
|
|
|
|
|
|
|
Findings” |
2 |
> |
HAS |
CODE |
EV (246112005, SCT, |
1 |
U |
DCID3716“Severity” |
|
|
PROPERTIES |
|
"Severity") |
|
|
|
3 |
> |
HAS |
CODE |
EV (363698007, SCT, |
1 |
U |
|
|
|
PROPERTIES |
|
"Finding Site") |
|
|
|
4 |
>> |
HAS CONCEPTCODE |
EV (106233006, SCT, |
1 |
U |
DCID 3019 |
|
|
|
MOD |
|
"Topographical modifier") |
|
|
“Cardiovascular |
|
|
|
|
|
|
|
Anatomic Location |
|
|
|
|
|
|
|
Modifiers” |
5 |
> |
|
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
|
Qualifiers” |
|
|
|
6 |
|
|
TEXT |
BCID 3419 “Findings Titles” |
1 |
U |
|
7 |
> |
|
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
|
Qualifiers” |
|
|
|
Content Item Descriptions |
|
|
|
|
|
||
Row 3 |
Finding Site has no Baseline Context ID specified. Typically terms would be drawn from coronary segments, |
||||||
|
|
other arterial segments, myocardial segments, etc. |
|
|
|
||
TID 3111 Percutaneous Entry
The Percutaneous Entry Template allows recording of the opening or closing of invasive access ports.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
- Standard -

DICOM PS3.16 2020a - Content Mapping Resource Page 173
Table TID 3111. Percutaneous Entry
|
NL RelwithParent VT |
Concept Name |
VM Req TypeConditionValue Set Constraint |
|||
1 |
|
CODE |
EV (121156, DCM, |
1 |
M |
DCID 3403 |
|
|
|
"PercutaneousEntryAction") |
|
|
“Percutaneous Entry” |
2 |
> |
HASCONCEPTCODE |
EV (272741003, SCT, |
1 |
U |
DCID 244 “Laterality” |
|
|
MOD |
"Laterality") |
|
|
|
3 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
Qualifiers” |
|
|
|
TID 3112 Specimen Obtained
The Specimen Obtained Template allows recording of obtaining a specimen, and the identifiers for that specimen. This is particularly designedforbloodsamplesthatwillbeanalyzedforbloodoxygen-relatedmeasurements.Theanalysisofthesamplemayberecorded in one or more log entries using TID 3109 “Measurements”, or in a separate Structured Report SOP Instance referenced by a log entry using TID 3103 “Referenced Object”.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
Table TID 3112. Specimen Obtained
|
NL Rel with Parent VT |
Concept Name |
VM |
Req |
Condition Value Set Constraint |
|||
|
|
|
|
|
|
Type |
|
|
1 |
|
|
CODE |
EV (121123, DCM, "Patient |
1 |
M |
|
DCID 3515 “Specimen |
|
|
|
|
Status or Event") |
|
|
|
Collection” |
2 |
> |
HAS ACQ |
CODE |
EV (371439000, SCT, |
1 |
UC |
IFF |
DCID 3520 “Blood |
|
|
CONTEXT |
|
"Specimen Type") |
|
|
specimen isSource Type” |
|
|
|
|
|
|
|
|
bloodsample |
|
3 |
> |
HAS ACQ |
CODE |
EV (363704007, SCT, |
1 |
U |
|
BCID 3630 |
|
|
CONTEXT |
|
"Procedure site") |
|
|
|
“Cardiovascular |
|
|
|
|
|
|
|
|
Anatomic Locations” |
4 |
> |
HAS |
INCLUDE DTID 1009 “Subject |
1 |
U |
|
|
|
|
|
PROPERTIES |
|
Context, Specimen” |
|
|
|
|
TID 3113 Patient Support
The Patient Support Template allows recording of the use of various support technologies, including oxygen, ventilation, pacing, etc.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
|
Table TID 3113. Patient Support |
NL |
Rel with |
VT |
Concept Name |
VM |
Req Condition |
Value Set |
|
Parent |
|
|
|
Type |
Constraint |
1 |
|
CODE |
DCID 3530 “Oxygen |
1 |
U |
DCID 3531 “Oxygen |
|
|
|
Administration Actions” |
|
|
Administration” |
2 > |
HAS |
NUM |
EV (121160, DCM, "Oxygen |
1 |
MC IFF Row 1 |
UNITS = DT (l/min, |
|
PROPERTIES |
Administration Rate") |
|
Concept is |
UCUM, "l/min") |
|
|
|
|
|
|
(121161, DCM, |
|
|
|
|
|
|
"Begin Oxygen |
|
|
|
|
|
|
Administration") |
|
- Standard -
Page 174 |
DICOM PS3.16 2020a - Content Mapping Resource |
|
||||
|
NL Rel with |
VT |
Concept Name |
VM |
Req Condition |
Value Set |
|
Parent |
|
|
|
Type |
Constraint |
3 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
Qualifiers” |
|
|
|
4 |
|
CODE |
DCID 3550 “Circulatory |
1 |
U |
DCID 3553 |
|
|
|
Support Actions” |
|
|
“CirculatorySupport” |
5 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
Qualifiers” |
|
|
|
6 |
|
CODE |
DCID 3551 “Ventilation |
1 |
U |
DCID 3554 |
|
|
|
Actions” |
|
|
“Ventilation” |
7 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
Qualifiers” |
|
|
|
8 |
|
CODE |
DCID 3552 “Pacing Actions” |
1 |
U |
DCID 3555 “Pacing” |
9 |
> |
INCLUDE |
DTID 3010 “Log Entry |
1 |
U |
|
|
|
|
Qualifiers” |
|
|
|
TID 3114 Patient Assessment
ThePatientAssessmentTemplateallowsrecordingoftheassessmentofthepatient'scardiovascular,neurological,and/orrespiratory condition. A particular use of this Template is for "vital signs", which are a specific subset of mandatory patient assessment measure- ments.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
|
Table TID 3114. Patient Assessment |
|
NL Rel with |
VT |
Concept Name |
VM Req |
Condition |
Value Set Constraint |
|
|
|
Parent |
|
|
Type |
|
|
1 |
|
|
CODE |
EV (121123, DCM, |
1 M |
|
DT (121165, DCM, "Patient |
|
|
|
|
"Patient Status or |
|
|
Assessment Performed") |
|
|
|
|
Event") |
|
|
DT (61746007, SCT, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
"Observation of Vital Signs") |
2 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = EV |
||
|
|
PROPERTIES |
“Measurement” |
|
(61746007, SCT, |
(271649006, SCT, "Systolic |
|
|
|
|
|
|
|
"Observation of Vitalblood pressure") |
|
|
|
|
|
|
|
Signs") |
$Units = DCID 3500 “Pressure |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Units” |
|
|
|
|
|
|
|
$Method = BCID 3560 “Blood |
|
|
|
|
|
|
|
Pressure Methods” |
3 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = EV |
||
|
|
PROPERTIES |
“Measurement” |
|
(61746007, SCT, |
(271650006, SCT, "Diastolic |
|
|
|
|
|
|
|
"Observation of Vitalblood pressure") |
|
|
|
|
|
|
|
Signs") |
$Units = DCID 3500 “Pressure |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Units” |
4 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = EV (8867-4, |
||
|
|
PROPERTIES |
“Measurement” |
|
(61746007, SCT, |
LN, "Heart rate") |
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
Signs") |
$Units=EV({H.B.}/min,UCUM, |
|
|
|
|
|
|
|
"BPM") |
- Standard -
|
|
|
|
DICOM PS3.16 2020a - Content Mapping Resource |
Page 175 |
|||
|
NL Rel with |
VT |
Concept Name |
VM Req |
Condition |
Value Set Constraint |
||
|
|
Parent |
|
|
|
Type |
|
|
5 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = EV (8310-5, |
|||
|
|
PROPERTIES |
“Measurement” |
|
|
(61746007, SCT, |
LN, "Body temperature") |
|
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
|
Signs") |
$Units = EV (Cel, UCUM, "C") |
6 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = DCID 3526 |
|||
|
|
PROPERTIES |
“Measurement” |
|
|
(61746007, SCT, |
“Blood Gas Saturation” |
|
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
|
Signs") |
$Units = EV (%, UCUM, "%") |
7 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement=EV(86290005, |
|||
|
|
PROPERTIES |
“Measurement” |
|
|
(61746007, SCT, |
SCT, "Respiration rate") |
|
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
|
Signs") |
$Units = EV (/min, UCUM, |
|
|
|
|
|
|
|
|
"breaths/min") |
8 |
> |
HAS |
INCLUDEDTID 300 |
1-n MCIF Row 1 value = |
$Measurement = EV (122195, |
|||
|
|
PROPERTIES |
“Measurement” |
|
|
(61746007, SCT, |
DCM, "Pulse Strength") |
|
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
|
Signs") |
$Method = BCID 3442 |
|
|
|
|
|
|
|
|
“Peripheral Pulse Methods” |
|
|
|
|
|
|
|
|
$TargetSite = BCID 3440 |
|
|
|
|
|
|
|
|
“Peripheral Pulse Locations” |
|
|
|
|
|
|
|
|
$Units = DT ({0:4}, UCUM, |
|
|
|
|
|
|
|
|
"range 0:4") |
9 |
> |
HAS |
INCLUDEDTID 300 |
1 MCIF Row 1 value = |
$Measurement = EV |
|||
|
|
PROPERTIES |
“Measurement” |
|
|
(61746007, SCT, |
(225908003,SCT,"PainScore") |
|
|
|
|
|
|
|
|
"Observation of Vital |
|
|
|
|
|
|
|
|
Signs") |
$Units = DT ({1:10}, UCUM, |
|
|
|
|
|
|
|
|
"range 1:10") |
10 |
> |
HAS |
CODE |
DT (8884-9, LN, |
1 |
U |
|
BCID 3415 “Cardiac Rhythms” |
|
|
PROPERTIES |
"Cardiac Rhythm") |
|
|
|
|
|
11 |
> |
HAS |
CODE |
DT (9304-7, LN, |
1 |
U |
|
BCID 3416 “Respiration |
|
|
PROPERTIES |
"Respiration |
|
|
|
Rhythms” |
|
|
|
|
|
Rhythm") |
|
|
|
|
12 |
> |
HAS |
CODE |
DT (364062005, |
1 |
U |
|
BCID3448“AirwayAssessment” |
|
|
PROPERTIES |
SCT, "Respiration |
|
|
|
|
|
|
|
|
|
Assessment") |
|
|
|
|
13 |
> |
HAS |
CODE |
DT (364528001, |
1-n |
U |
|
BCID 3446 “Skin Condition” |
|
|
PROPERTIES |
SCT, "Skin |
|
|
|
|
|
|
|
|
|
condition") |
|
|
|
|
14 > |
HAS |
CODE |
DT (363871006, |
1 |
U |
|
|
|
|
|
PROPERTIES |
SCT, "Patient mental |
|
|
|
||
|
|
|
|
state assessment") |
|
|
|
|
15 |
> |
HAS |
TEXT |
BCID 3441 “Patient 1-n |
U |
|
|
|
|
|
PROPERTIES |
Assessments” |
|
|
|
|
|
Content Item Descriptions |
|
|
|
|
|
|||
Row 8 |
Pulse Strength allows the assessment of the patient's pulse at multiple locations using the Topographical |
|||||||
|
|
concept modifier. It may also be used for a single pulse strength measurement from an unspecified location, |
||||||
|
|
as is typical of vital signs assessments. |
|
|
|
|||
Row 16 |
Allows free text description of patient assessments that are not expressible by coded entries of Rows 10 to |
|||||||
|
|
14. |
|
|
|
|
|
|
- Standard -
Page 176 |
DICOM PS3.16 2020a - Content Mapping Resource |
TID 3115 ECG ST Assessment
The ECG ST Assessment Template allows recording of the assessment of changes in the patient ECG relative to baseline.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
Table TID 3115. ECG ST Assessment
|
NL Rel with Parent VT |
Concept Name |
VM |
Req |
ConditionValue Set Constraint |
||
|
|
|
|
|
|
Type |
|
1 |
|
|
CODE |
EV (121123, DCM, "Patient |
1 |
M |
DT (258181008, SCT, |
|
|
|
|
Status or Event") |
|
|
"ECG Analysis") |
2 |
> |
HAS |
NUM |
DT (122099, DCM, "ST |
1-n |
M |
UNITS = EV (uV, UCUM, |
|
|
PROPERTIES |
|
change from baseline") |
|
|
"uV") |
3 |
>> |
HAS CONCEPTCODE |
DT (122148, DCM, "Lead |
1 |
M |
BCID 3001 “ECG Leads” |
|
|
|
MOD |
|
ID") |
|
|
|
Quantitative Ventricular Analysis Report SR IOD Templates
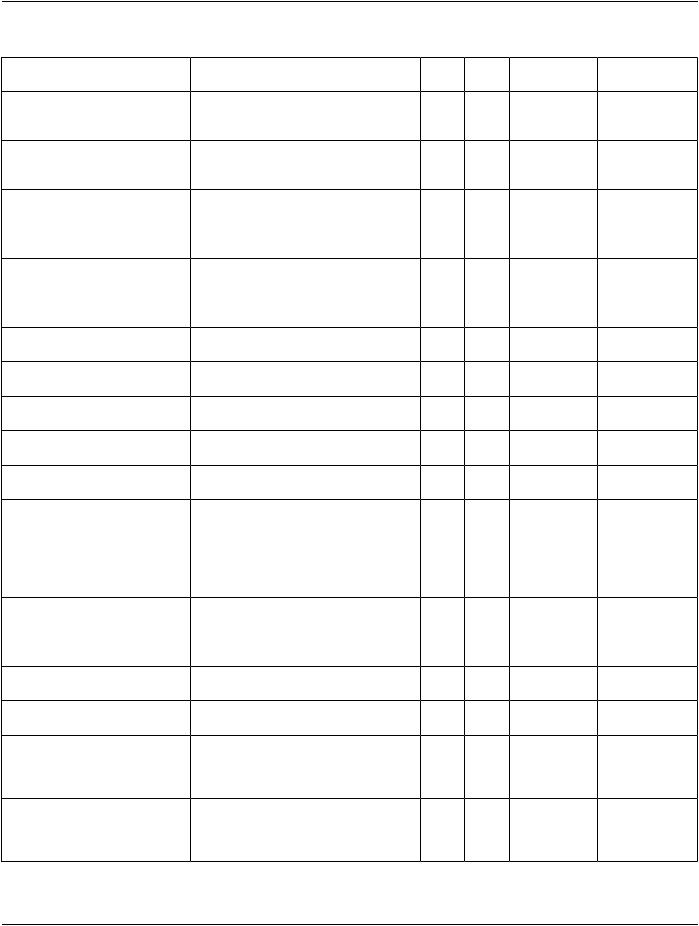
The Templates that comprise the Quantitative Ventricular Analysis SR are interconnected as in Figure A-3:
TID 3202 |
|
|
TID 1002 |
Ventricular |
|
|
Observer |
Analysis |
|
|
Context |
|
|
|
TID 1204 |
|
|
|
Language of |
|
|
|
Content Item and |
|
|
|
Descendants |
|
|
|
TID 3602 |
|
|
|
Patient |
|
|
|
Characteristics |
|
|
|
TID 3205 |
|
|
|
Calibration |
|
|
|
TID 3520 |
|
|
|
Hemodynamic |
|
|
|
Clinical Context |
|
|
|
TID 3206 |
|
|
|
AA Main Results |
|
|
|
TID 3208 |
|
|
|
Frame to Frame |
|
|
|
Results |
|
|
|
TID 3209 |
|
|
|
Centerline Wall |
|
|
|
Motion |
|
|
|
TID 32011 |
|
|
|
Landmark Based |
|
|
|
Wall Motion |
Figure A-3. Quantitative Ventricular Analysis Report SR IOD Template Structure
TID 3202 Ventricular Analysis
The Ventricular Analysis Template provides a CONTAINER with a structure for reporting the result of the ventricular analysis.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
- Standard -
DICOM PS3.16 2020a - Content Mapping Resource Page 177
Table TID 3202. Ventricular Analysis
|
NL |
Rel with Parent VT |
Concept Name |
VM |
Req |
Condition |
Value Set |
|
|
|
|
|
|
|
Type |
|
Constraint |
1 |
|
|
CONTAINEREV (122292, DCM, |
1 |
M |
|
|
|
|
|
|
|
"Quantitative |
|
|
|
|
|
|
|
|
Ventriculography Report") |
|
|
|
|
2 |
> |
HAS CONCEPTINCLUDE |
DTID 1204 “Language of |
1 |
M |
|
|
|
|
|
MOD |
|
Content Item and |
|
|
|
|
|
|
|
|
Descendants” |
|
|
|
|
3 |
> |
HAS OBS |
INCLUDE |
DTID 1002 “Observer |
1-n |
M |
|
|
|
|
CONTEXT |
|
Context” |
|
|
|
|
4 |
> |
CONTAINS |
INCLUDE |
DTID 3602 “Cardiovascular |
1 |
U |
|
|
|
|
|
|
Patient Characteristics” |
|
|
|
|
5 |
> |
CONTAINS |
CONTAINEREV (122144, DCM, |
1-n |
M |
|
|
|
|
|
|
|
"Quantitative Analysis") |
|
|
|
|
6 |
>> |
HAS OBS |
INCLUDE |
DTID 1002 “Observer |
1-n |
U |
|
|
|
|
CONTEXT |
|
Context” |
|
|
|
|
7 |
>> |
HAS OBS |
TEXT |
EV (111001, DCM, |
1 |
M |
|
|
|
|
CONTEXT |
|
"Algorithm Name") |
|
|
|
|
8 |
>> |
HAS OBS |
TEXT |
EV (111003, DCM, |
1 |
M |
|
|
|
|
CONTEXT |
|
"Algorithm Version") |
|
|
|
|
9 |
>> |
HAS OBS |
TEXT |
EV (122405, DCM, |
1 |
M |
|
|
|
|
CONTEXT |
|
"Algorithm Manufacturer") |
|
|
|
|
10 |
>> |
CONTAINS |
IMAGE |
EV (121112, DCM, "Source 1-n |
M |
|
|
|
|
|
|
|
of Measurement") |
|
|
|
|
11 |
>>> |
HAS CONCEPTCODE |
EV (246092007, SCT, |
1 |
M |
|
DCID 12233 |
|
|
|
MOD |
|
"Cardiac Phase") |
|
|
|
“Cardiac Phase” |
12 |
>>> |
HAS CONCEPTCODE |
EV (111031, DCM, "Image |
1 |
MC If Biplane |
DCID3466“Plane |
||
|
|
MOD |
|
View") |
|
|
Analysis |
Identification” |
13 |
>> |
HAS ACQ |
INCLUDE |
DTID 3205 “Calibration” |
1-2 |
U |
VM = 1: Single$CalibrationPlane |
|
|
|
CONTEXT |
|
|
|
|
plane analysis,= DCID 3466 |
|
|
|
|
|
|
|
|
VM=2:Biplane“Plane |
|
|
|
|
|
|
|
|
analysis |
Identification” |
14 |
>> |
HAS ACQ |
INCLUDE |
DTID 3520 “Hemodynamic |
1 |
U |
|
|
|
|
CONTEXT |
|
Clinical Context” |
|
|
|
|
15 |
>> |
CONTAINS |
INCLUDE |
DTID 3206 “VA Main |
1 |
M |
|
|
|
|
|
|
Results” |
|
|
|
|
16 |
>> |
CONTAINS |
INCLUDE |
DTID 3207 “AA Main |
1 |
U |
|
|
|
|
|
|
Results” |
|
|
|
|
17 |
>> |
CONTAINS |
INCLUDE |
DTID3208“Frame-to-Frame |
1 |
U |
|
|
|
|
|
|
Results” |
|
|
|
|
18 |
>> |
CONTAINS |
INCLUDE |
DTID 3209 “Centerline Wall 1-2 |
U |
VM = 1: Single |
||
|
|
|
|
Motion” |
|
|
plane analysis, |
|
|
|
|
|
|
|
|
VM=2:Biplane |
|
|
|
|
|
|
|
|
analysis |
|
19 |
>> |
CONTAINS |
INCLUDE |
DTID 3210 “Radial Based |
1-2 |
U |
VM = 1: Single |
|
|
|
|
|
Wall Motion” |
|
|
plane analysis, |
|
|
|
|
|
|
|
|
VM=2:Biplane |
|
|
|
|
|
|
|
|
analysis |
|
- Standard -
Page 178 |
|
|
DICOM PS3.16 2020a - Content Mapping Resource |
|
|
|||
NL |
Rel with Parent |
VT |
Concept Name |
VM |
Req |
Condition |
Value Set |
|
|
|
|
|
|
|
Type |
|
Constraint |
20 >> |
CONTAINS |
INCLUDE |
DTID 3211 “Landmark |
1-2 |
U |
VM = 1: Single |
|
|
|
|
|
|
Based Wall Motion” |
|
|
plane analysis, |
|
|
|
|
|
|
|
|
VM=2:Biplane |
|
|
|
|
|
|
|
|
analysis |
|
Content Item Descriptions |
|
|
|
|
|
|
|
|
Row 7 |
|
Identifies the Ventricular Analysis program |
|
|
|
|
||
Row 8 |
|
Identifies the Ventricular Analysis program version |
|
|
|
|||
Row 9 |
|
Identifies the Ventricular Analysis program manufacturer |
|
|
||||
Row 10 |
|
Identifies the ES and ED images on which the analysis is based, for frame by frame analysis the |
||||||
|
|
analyzed image are specified in the frame by frame results (3208) Template |
|
|||||
TID 3205 Calibration
The Calibration Template consists of a CONTAINER, with a structure for reporting of the calibration of images used in the analysis.
|
|
|
|
Table TID 3205. Parameters |
|
|||
|
|
Parameter Name |
|
|
|
Parameter Usage |
||
$CalibrationPlane |
|
XA Imaging plane |
|
|||||
Type: |
|
Extensible |
|
|
|
|
||
Order: |
|
Significant |
|
|
|
|
||
Root: |
|
No |
|
|
|
|
|
|
|
|
|
|
Table TID 3205. Calibration |
|
|||
|
NL |
Rel with |
VT |
Concept Name |
VM |
Req |
Condition |
Value Set Constraint |
|
|
Parent |
|
|
|
Type |
|
|
1 |
|
|
CONTAINEREV (122505, DCM, |
1 |
M |
|
|
|
|
|
|
|
"Calibration") |
|
|
|
|
2 |
> |
HAS |
CODE |
EV (111031, DCM, |
1 |
U |
|
$CalibrationPlane |
|
|
CONCEPT |
|
"Image View") |
|
|
|
|
|
|
MOD |
|
|
|
|
|
|
3 |
> |
HAS OBS |
TEXT |
EV (111001, DCM, |
1 |
MC IF different from |
|
|
|
|
CONTEXT |
|
"Algorithm Name") |
|
|
Analysis program |
|
|
|
|
|
|
|
|
specified in the |
|
|
|
|
|
|
|
|
invoking Template |
|
4 |
> |
HAS OBS |
TEXT |
EV (111003, DCM, |
1 |
MC IF different from |
|
|
|
|
CONTEXT |
|
"Algorithm Version") |
|
|
Analysis program |
|
|
|
|
|
|
|
|
specified in the |
|
|
|
|
|
|
|
|
invoking Template |
|
5 |
> |
HAS OBS |
TEXT |
EV (122405, DCM, |
1 |
MC IF different from |
|
|
|
|
CONTEXT |
|
"Algorithm |
|
|
Analysis program |
|
|
|
|
|
Manufacturer") |
|
|
specified in the |
|
|
|
|
|
|
|
|
invoking Template |
|
6 |
> |
CONTAINS |
CODE |
EV (122422, DCM, |
1 |
M |
|
DCID 3452 “Calibration |
|
|
|
|
"Calibration Method") |
|
|
|
Methods” |
7 |
> |
CONTAINS |
CODE |
EV (122421, DCM, |
1 |
MC If row 6 value |
DCID 3451 “Calibration |
|
|
|
|
|
"Calibration Object") |
|
|
specifies CalibrationObjects” |
|
|
|
|
|
|
|
|
Object Used |
|
- Standard -

|
|
|
DICOM PS3.16 2020a - Content Mapping Resource |
Page 179 |
||||
|
NL |
Rel with |
VT |
Concept Name |
VM |
Req |
Condition |
Value Set Constraint |
|
|
Parent |
|
|
|
Type |
|
|
8 |
> |
CONTAINS |
NUM |
EV (122423, DCM, |
1 |
MC If row 6 value |
DCID3510“CatheterSize |
|
|
|
|
|
"Calibration Object |
|
|
specifies CalibrationUnits” |
|
|
|
|
|
Size") |
|
|
Object Used |
|
9 |
> |
CONTAINS |
INCLUDE |
DTID 300 |
1 |
M |
|
$Measurement = EV |
|
|
|
|
“Measurement” |
|
|
|
(111026, DCM, |
|
|
|
|
|
|
|
|
"HorizontalPixelSpacing") |
|
|
|
|
|
|
|
|
$Unit = DT (mm/{pixel}, |
|
|
|
|
|
|
|
|
UCUM, "mm/pixel") |
|
|
|
|
|
|
|
|
$ImagePurpose = EV |
|
|
|
|
|
|
|
|
(121112,DCM,"Sourceof |
|
|
|
|
|
|
|
|
Measurement") |
10 |
> |
CONTAINS |
INCLUDE |
DTID 300 |
1 |
M |
|
$Measurement = EV |
|
|
|
|
“Measurement” |
|
|
|
(111066, DCM, "Vertical |
|
|
|
|
|
|
|
|
Pixel Spacing") |
|
|
|
|
|
|
|
|
$Unit = DT (mm/{pixel}, |
|
|
|
|
|
|
|
|
UCUM, "mm/pixel") |
|
|
|
|
|
|
|
|
$ImagePurpose = EV |
|
|
|
|
|
|
|
|
(121112,DCM,"Sourceof |
|
|
|
|
|
|
|
|
Measurement") |
11 |
> |
CONTAINS |
IMAGE |
|
1 |
U |
|
|
Content Item Descriptions |
|
|
|
|
|
|||
Row 3 |
|
Identifies the Calibration program |
|
|
|
|
||
Row 4 |
|
Identifies the Calibration program version |
|
|
|
|||
Row 5 |
|
Identifies the Calibration program manufacturer |
|
|
||||
Row 7 |
|
Besides a Sphere and a Catheter, a Distance can be identified as a Calibration Object. In this case a |
||||||
|
|
|
distance measurement of a known dimension of the object is used to calculate the pixel size. |
|||||
Row 8 |
|
The catheter size units is also used to specify the size of other calibration objects (e.g., sphere) |
||||||
Row 9, 10 |
|
Spacinginthepatientbody.Pointtoasingleframecontainingtheimageusedforcalibrationifapplicable, |
||||||
|
|
|
theactualmeasurementsmaybeindicatedbyaSCOORD(seeTID320“ImageorSpatialCoordinates”, |
|||||
|
|
|
row 3) |
|
|
|
|
|
Row 11 |
|
Secondary Capture image with calibration position. No purpose of reference is specified. |
||||||
TID 3206 VA Main Results
The VA Main Results Template consists of a CONTAINER with a structure for reporting the main ventricular analysis measurements.
Type: |
Extensible |
Order: |
Significant |
Root: |
No |
|
Table TID 3206. VA Main Results |
NL Rel with |
VT |
Concept Name |
VM ReqCondition |
Value Set Constraint |
Parent |
|
|
Type |
|
1 |
CONTAINEREV (121070, DCM, |
1 M |
|
|
|
|
"Findings") |
|
|
- Standard -

Page 180 |
|
DICOM PS3.16 2020a - Content Mapping Resource |
|
|||||
|
NL |
Rel with |
VT |
Concept Name |
VM ReqCondition |
Value Set Constraint |
||
|
|
Parent |
|
|
|
Type |
|
|
2 |
> |
HAS |
CODE |
EV(363698007,SCT, 1 |
M |
DCID 3462 “Chamber Identification” |
||
|
|
CONCEPT |
|
"Finding Site") |
|
|
|
|
|
|
MOD |
|
|
|
|
|
|
3 |
> |
CONTAINS CODE |
EV (122429, DCM, |
1 |
M |
DCID 3453 “Cardiac Volume Methods” |
||
|
|
|
|
"Volume Method") |
|
|
|
|
4 |
> |
CONTAINS NUM |
EV (122435, DCM, |
1 |
U |
Unit = DT (1, UCUM, "no units") |
||
|
|
|
|
"Regression Volume |
|
|
|
|
|
|
|
|
Exponent") |
|
|
|
|
5 |
> |
CONTAINS NUM |
EV (122431, DCM, |
1 |
U |
Unit = DT ({ratio}, UCUM, "ratio") |
||
|
|
|
|
"Regression Slope |
|
|
|
|
|
|
|
|
ED") |
|
|
|
|
6 |
> |
CONTAINS NUM |
EV (122432, DCM, |
1 |
U |
Unit = DT (ml, UCUM, "ml") |
||
|
|
|
|
"Regression Offset |
|
|
|
|
|
|
|
|
ED") |
|
|
|
|
7 |
> |
CONTAINS NUM |
EV (122433, DCM, |
1 |
U |
Unit = DT ({ratio}, UCUM, "ratio") |
||
|
|
|
|
"Regression Slope |
|
|
|
|
|
|
|
|
ES") |
|
|
|
|
8 |
> |
CONTAINS NUM |
EV (122434, DCM, |
1 |
U |
Unit = DT (ml, UCUM, "ml") |
||
|
|
|
|
"Regression Offset |
|
|
|
|
|
|
|
|
ES") |
|
|
|
|
9 |
> |
CONTAINS INCLUDE |
DTID 300 |
1 |
M |
$Measurement = DCID 3467 “Ejection |
||
|
|
|
|
“Measurement” |
|
|
Fraction” |
|
|
|
|
|
|
|
|
$Unit = DT (%, UCUM, "%") |
|
10 |
> |
CONTAINS INCLUDE |
DTID 300 |
1 |
U |
$Measurement = DCID 3468 “ED |
||
|
|
|
|
“Measurement” |
|
|
Volume” |
|
|
|
|
|
|
|
|
$Unit = DT (ml, UCUM, "ml") |
|
11 |
> |
CONTAINS INCLUDE |
DTID 300 |
1 |
U |
$Measurement = DCID 3469 “ES |
||
|
|
|
|
“Measurement” |
|
|
Volume” |
|
|
|
|
|
|
|
|
$Unit = DT (ml, UCUM, "ml") |
|
12 |
> |
CONTAINS INCLUDE |
DTID 300 |
1 |
U |
$Measurement = EV (90096001, SCT, |
||
|
|
|
|
“Measurement” |
|
|
"Stroke Volume") |
|
|
|
|
|
|
|
|
$Unit = DT (ml, UCUM, "ml") |
|
13 |
> |
CONTAINS NUM |
EV (8867-4, LN, |
1 |
U |
Unit = DT ({H.B.}/min, UCUM, "BPM") |
||
|
|
|
|
"Heart Rate") |
|
|
|
|
14 |
> |
CONTAINS INCLUDE |
DTID 300 |
1 |
U |
$Measurement = DCID 3468 “ED |
||
|
|
|
|
“Measurement” |
|
|
Volume” |
|
$ModType = EV (121425, DCM, "Index")
$ModValue = DCID 3455 “Index
Methods”
$Unit = DT (ml/m2, UCUM, "ml/m2")
- Standard -
