
ostrovskaya_a_v_garifullina_a_r_abdullin_i_sh_tekhnologiya_i
.pdf
|
|
|
|
|
|
|
|
N H C H 2 O H |
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
C |
|
|
|
|||||||||||
|
|
|
|
|
|
N |
|
N |
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H O H 2 C H N |
|
C |
|
C |
|
N |
|
C H 2 |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
||||||||||||||||
H |
|
|
|
|
|
|
|
N |
|
|
|
|
O H |
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
n |
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
2 7 7
.
' * !. 0 6
, ,
,
.
0
, 6,
6. * 6, 7 6 6 6
|
|
|
|
|
|
NH(CH2OH) |
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
C |
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
N |
|
|
N |
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(CH2OH)HN |
|
C |
|
|
C |
|
NH |
|
CH2 |
|
|
|
|
|
|
|
N |
|
N |
|
...Kp |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
|
|
N |
|
|
|
|
, |
|||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
– 6 .
4 &' * !. 6 6 .3, .3#,
.32.
3 7 ,
6 6. # .
.3 .
.3# – , 6
.
.32 – 210-16 ,
6.
H |
|
|
|
|
|
|
|
|
SO 2 |
|
|
|
|
|
|
|
CH2 |
|
|
|
O H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
HO |
|
|
|
|
|
|
|
|
O H |
|
|
|
|
||||||
|
|
|
|
|
|
|
|||||||||||||||
|
|
|
|
CH2O C O R |
CH2O CO R |
|
|
|
|
||||||||||||
n
1 &' ! ! # * !. :
. # 6
-151-
, 7 6 , 6 . ' 6
6, ,
, , .
6 ) ! 1 - ,
6
, ) ! * 2 – . # , 6,
. %
, , . 0
6 6 |
6 6 6: |
|
|
|
+ |
OH |
OH |
|
CH2 |
CH2 |
CH2 NH3 SO42- |
|
n-1 |
|
|
|
2 |
( * ' . . 6
6 6 6 (6 6). 6 6 7 6: 6 6
6 (6), 6 6 6.
6 9 6.
6 6:
|
|
|
|
|
|
|
|
NHCH2OH |
|
|
|
|
|||
|
|
|
O |
|
C |
|
|
|
|
||||||
|
|
|
|
|
|||||||||||
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
N |
|
CH2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
H |
|
|
|
|
|
|
|
|
|
|
n |
OH |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
6 6 6:
|
|
|
|
|
|
|
|
N HCH2O H |
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
C |
|
|
|
|||||||||||
|
|
|
|
|
|
N |
|
N |
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
HO H2CHN |
|
C |
|
C |
|
N |
|
CH2 |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
||||||||||||||||
H |
|
|
|
|
|
|
|
N |
|
|
|
|
O H |
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
n |
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
# 6- 6 6 6 6 6 6 ,
( ) 6 6
.
0 9 6 6 6 6
95°C, 6 7 6.
-152-
. # 6
6. 6
,
. 2 6
6. 0 :
1)6 6
6 6 6;
2)6 6 . ( 9 ,
6 6 6;
3)6 6
,
6.
0 7 .
0 6 6 6 6 6 6. "6
6 6. "
6 6 6 6 6. ' 6
6 , .
!6 6 7 7
.
1. . . 0 .
7 7. % . " 6 6 6, .
2.6
6 6 .
3.' 6 6 :
, .
4. . 6 .
5. 2 6 6 .
. " 6 6 . 6.* .
* 7 7. ' . 7. . 6 6, , .
8. # 7 7. # 7 . .7
. 2 6 . 9. .7 .
-153-
10. 2
. ) 6 6 6 . # , 7 ..
11. . 6 6 6. '. 2 7 .
12. 2 (6, 6
, ) 13. .7 6
#-
-*' ! * ! # % %.
# 6 ,
. # 6 ,
6 . 6 : ,
, , , , ,
, 6
6 .
0 6 6
7 6 9 . * :
-6 ;
- |
6 6 |
- |
6 |
; |
|
-7.
0 6 :
1.0, - 6 6 6;
2.#, 7 6
6 .
6 : , , ,
, , , ,
; 6 – , , ,
, , ,
, ( ) 6, , 6 7.
-154-
: 6
6 6 .
|
|
|
< 7 |
|
6 6 |
6, 6 7 |
. |
|
! # , . . |
||
( ) 9 6 9. |
|
|
$ 6 , 7
6 . %6 400 760 6. 0 6 6
6 9 6 7 7
.
0 6 6 6 6
9 6 6
. " 6
6 6, 6, 6
6 6. (
7 6, 6.
- (% !) ,
7 6 6
7 6 . (, 6,
7 580-595 6,
7 6 ,
6 6 400 580 595 760 6
7 7 .
7 7 ?
6 6 (7) 6 6
9. 0 7 9 , 6 6
6 . 0
7 . ; 6 (10-5 ) 6
6 7 6 -
. # 7 9 6 9 ΔΕ
ħγ, ΔΕ = ħγ, ħ –
0, γ – 6
9 6 .
2 7, ,
, 6
-155-
9 158
– 300 ./6. : 9 9 6 6
.
0 9 6 (()
9 9 (( ), 9 (( ) 7
(( ) 9 6 6:
( = ( + ( + ( 0 6 ( 6 6 7 9
6.
σ – 9, 7 6
6 6. .
9
800 ./6. 2, 9 6
, 6 6 7
.
π – 9 . # 9 . ;
.
%, ! ,
7 7
9 9 6 ( 9) 6
. # 6
7 6 (6 6, 6).
) ! % ! !
-6 6 6 6 6
6 !. . 5 6,
|
6, 1868 . |
5 7 6, 7 |
|
6. |
|
0.0. ! (1876 .) 6, |
|
|
6, |
7 6 6, 6 6 (NO2),
(NO), (2=#),
6, (#*) 6 (NH2). |
|
||||
: |
|
|
|
|
|
6 6 , 6 #." 6
1876 .
-156-
) 6, 6 #." : (NO2),
(NO), (2=#) - (N=N),
7 6 6 6.
) 6 6, . .
" 7 6
« 6» ( – , - ). 2,
6 .
6 6 " 7 : NH2, OH, SH .
) 6 6
6 , 6 5.%. 2 6.
" ! !,
(=)
, 6 6 6
. ) 6
6, 7 7 9 ,
6, NH2, NHR, NR2, OH. : 9. 6 6
6, 6
9 – 9 . 6 NO2 , 2=#. %
.
" 6 6
. . 9 6, . . 6 6 6 7 9 . # ,
6 9 6 9 6
, 7 6
9 .
" ! # ! #.
6 6 , , . 6 6 –
, .
) 6 % ! !, # ! ! % ! (7) ! & ( ).
9 ! ! 6 7 – λ * 6, 580-595 6.
+ ! ! % !
. 6 6 6 6
. 0 6- 6
.
-157-
3 ! – 9 ,
6 6 . * 6, , – .
) % !. " 6
! ! ( )
! ! #.
2 6 2 : # &
! # &.
# 6 ,
6 . 2 , 15
: -, -, 6, , 6,
6, , 6 , -
-, , , . * -6 6
3 : , 6
.
1.!, 7 (-N=N-) - 6 . 5 50 % 9 .
9, 6, , .
2.) 6 – 9 - 6 -
6 6 7
6 6 6 6.
3.! – 6 - 6 . < 6
6. ', 6 α- (1,4,5,8)
, 6 β- (2,3,6,7). 1,4 –
, 6 1,5 1,8 – 6 7.
|
O |
8 |
1 |
7 |
2 |
6 |
3 |
5 |
4 |
O
: 6
6 6.
# ) % !. 2
, 7 :
1. (RSO3N ) – 6
, R – - ,
. 7 6 6 7
-158-
6 6 .
66 . # 7 . #
6 6 6,
.
6, 9 6
7 6 6 .
,
:
RSO3Na ↔ RSO3- + Na+
6-
. - 9 6 ,
.
. – 9, 6 6,
. # 6 6 7 (* 4,5
– 5,0).
– 9
. 4 , 6
.
' 6 – 9
, .
6 6 ( ) 6 6
( 6 ). 6
, 6,
- . " 6 ,
# ( )
- - ( ).
6 6
6 :
6 6, . 0 6
6 6 .
2.0 6 – 6 6,
6 6 , 6 6. )
6 .
, 6 , . .
. 0 6
.
-159-
3.# – 6
6 : RNH2.HX, R –
. #
7 . #
. 0 9 6 6 6 6
6 6 6 . # . #
6 7 6, 6, 6
6 6 6, 9 6 6 7 6
6 6 6 .
4.0 -
6 6. # .
; 7 6 6.
5. 7 -
6 6. " 6 6. #
, . . . # . 0 6 6 6
6 6, 6
6 6. 0 9
6, 6.
2 6 6 6 7
6:
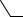 N
N  N
N 
O  O
O
H2O Cr OH
C N C
R H O
6. ! . # 6 6,
6 6 6 6
6 6 . :
, ,
, . " 7 6 6 6
6 7 6: S- – ZX,
S – , 7 6 ,
9 ;
-160-
