
ostrovskaya_a_v_garifullina_a_r_abdullin_i_sh_tekhnologiya_i
.pdf7 6 6 6
6 7 .
. 6 6 6 7:
6 6 7,
6 . * 6
6 6
6 7 . "
6 7 7 . 0 9 6 7 6-8 %. +
7 . "
6, .
+ - 9
- :
, . ;
7 6
.
, 7 6
, 6
6 6. 0
6 6 6. * 9 6
6. ',
, 21,5 %,
– 47 % - 31,5 %.
*
6
.
"
$ , 6-
6 ,
(~4 %) 6, 6
6 (~1,7 %). - 6
. 2 6 6
- , 7
.
<
. #
-101-

6 , 9 6
, .
. 6 6
, 7 6 6
. # 6 6
NH2 |
|
|
|
|
NH2 |
B |
Ca + 2 HA |
|
2 B |
+ CaA2 |
|
|
|||||
COO- |
2 |
|
|
COOH |
|
|
|
|
|
|
|
B-5, !
. 6 ( , ,
) (6 .) . %
6 . 4
6 7 . " 6
pH 5 6 ,
6 pH 5.
0 6 . #
.
0 6, 6 6 66 6,
7 6: (NH4)2SO4 + H2O 
 NH4OH + H2SO4
NH4OH + H2SO4
NH4OH
 NH3
NH3 + H2O
+ H2O
|
N H 2 |
|
|
|
|
N H 2 |
B |
C O O - |
C a + H 2 S O 4 |
|
2 B |
+ C a S O 4 |
|
|
||||||
|
2 |
|
|
C O O H |
||
|
|
|
|
|
|
|
0 6 7 66 6,
6 , . .
6. " 7 66 ,
7
. .
66, 9 6 6
(NH4)2Ca(SO4)2. 2 (CaSO4) 6.
. 66. )
, 7 9 6, 6 . #
-102-
, 6 66, 6 6 -
6.
) 66 6
9 , ,
,
6
.
% - 6
6 7, 6, 6 66 6 6,
7 6 .
6 6 . * -6 . 0
7 6 . * ,
- , 6
- 6 .
! " $ , ,
6. "
6 ,
6 6, 6 6,
.
6
6 6, 6
9 6. " 6
6 6 ,
, , .
# 6
,
6 .
- 6 0.1 66 .
0.1 ,
0.1 6 . 0
. ! 6
6 0.1 ,
1 2 : != 1- 2. 6
-103-
, 7 6 0.1 NaOH, 6 6
1 .
0 6 200 / ,
0,4 63 0.1 NaOH, – 500 / ,
1 63 0.1 NaOH, -
6 - 6 16 / ,
– 70 / .
6 6 6 pH . # 6 6 pH 6 7,8- 8,7, $- – 7,4-7,6. 2 6 6
40 2 6 , 6 6 6
50 2, 6 . 0 6 . 5 6 6 6
- . —
. 0 6 6
. " 6
6 , 6 .
6
6
( ). * 6 6
.
0 6 ,. . 6 ,
6 6, 6. #
, 6
66.
* -
6 6 6,
. ; 7 6 , 7
,
(6, , ) .
2 7 6, 6
( ). 6 pH
. # 5080 /63. " ,
-104-

7 7 6 = 6
6 6 6 7 7
6 , .
0 pH 2,8–3,2 7. 0 6 pH ,
.
0 7 6 6 6
|
|
|
|
|
|
|
|
+ |
|
COOH |
|
|
|
|
COOH |
||
|
|
|
|
|
|
|||
B |
+ HA |
|
|
B |
NH3 |
A- |
||
|
||||||||
|
||||||||
|
NH |
|
|
|
|
|
||
|
2 |
|
|
|
|
|
|
|
) 6 6
6 6 6 6 ,
, 6 6,
6 7. 0 6 = 6 6. 2
9 6 , 6. 0 18-23 °2, . . 0,6-1.
0 :
6 2 – 5-7 ,
– 2,5-3 , . ., 6 7 , 6
. .
2 , 6 . $
6 7, 6 . " 6 6
, , 6
. 7 * 2,2-2,8.
0
6 . 0
6 6
6 .
$ * * ! *
0 6 6 :
-6 7, 7
;
;
-.
-105-
1. 0
6 . # 6 6. * 9
. 0 . " 0!" . 0 6 6. 6. " , 6 .
2.# . - . - 6 . * 6 . 6
. . 0,
6 6 .
3.* . 0 . (6
, 9 , -
6).
4.# 6. <. , 6
6. 0
6. .
5.0, 7. , 6
. 0 .
6.#. < 6 6
. .
!"
-*' ! % *. 0 - 9
6 , - 6 6
, 6
.
%
7 . 0
6, 6 6, 6
6, 7 6 6 . 0
6 , . '
6 7
.
. 6
7 6 7 6. 2, 6,
6
,
7 6 -6 6.
, 6
-106-
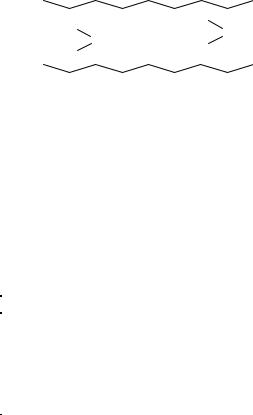
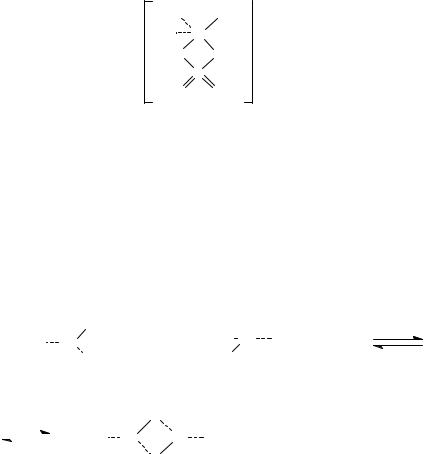
: ( 80 %) 9. 6 ,
: 6, .
6 6,
6 . "
7 7 6, 6
6 6 ,
6 6 . 0
6 6 . 2 6 9 6 :
|
|
|
|
|
|
|
|
- |
N H 2 |
||
C O O |
|||||
|
|
D |
|||
|
D |
|
|
||
|
|
C O O - |
|||
N |
H 2 |
|
|||
|
|
|
|||
|
|
|
|
|
|
- D - 6 6 .
0 6,
. 4
,
6 6.
# 7
. 4 7
6 . ' 6
6 (' .) ( . 2),
6 (6) 6 6 .
* % 2. 8$ ! ! # ! $! !
* |
|
. 6 |
' ., 02 ($#2' 938.25-73) |
. |
60-65 |
0 : |
|
6 |
70-90 |
6 6 |
90-140 |
2.6 |
65-75 |
6 6 |
85-90 |
' 6 6, 6 7
( ,
, - ). +
. 6 6 ,
6, 6.
0 6. . 6
. " 6
-107-

6 6 6
6 7 6, 6 ,
6 6.
% " & &
6 7 6 7 6
6 6, 6, , , .
. 7
6 6
6 6 . * 6 7 6
6 6.
( * '
" 6 6
6 +3 6 6 6,
#*. 2 6 +6 7 .
0 6 6 6 6 6 FeO.Cr2O3. 6
6
6 - K2Cr2O7 Na2Cr2O7.2H2O,
6 6 .
. 7 6 6 6
6 6 (+6) 6 ,
:
4K2Cr2O7 + 12 H2SO4 + C6H12O6  4K2SO4 + 8Cr(OH)SO4 + 6CO2 + 14H2O.
4K2SO4 + 8Cr(OH)SO4 + 6CO2 + 14H2O.
0
. 0 . # - . $ 6
6 2r2(SO4)n(OH)6-2n . 2 6 6
6 . # 150170 /63. $ 6 6
6 (+6), 7 . ) 6 (+3)
6. " 6
, 6. -108-

6 6 ,
6 6 6 6. ) 6 6 6
« ». + 6 6 6 H2O, NH3, OH-, SO42-, SO32-, HCOO-, CO32- . # 6
6. : 6
6, . 0
6 7 : Cl <
SO42- < SO32- < HCOO- < CH3COO- < C6H5COO- < (COO)2- < HOC(CH2COOH)2COO- < (HOCHCOO)2- < OH-. 2 6 6 6
, 7
( ). * 6, -
6 6 6 7 -
6, 6 6, 6, 6, 6, 6
6, 7 6 6 7
.
# , 7
, 6
(6, 6 6 6 6) . #
6 6, 6
, 9 6 6 6
.
6 6 6 6
6. # 6 6 , 6 7 6,
6 - , 6 7
.
" 6 6 6 ,
:
|
|
|
|
|
3+ |
|
|
H 2 O |
H 2 O |
||
|
|
H2O |
H2O |
|
|
||||||
|
|
|
|
|
|||||||
|
|
|
|
|
H 2 O C r |
|
O H |
||||
|
H O Cr |
H O |
Cl |
- |
|
|
|||||
|
|
O |
O |
||||||||
|
2 |
2 |
|
|
|
|
|||||
|
|
H2O |
H2O |
|
|
|
S |
|
|
||
|
|
|
|
|
O |
O |
|||||
|
|
|
|
|
|
|
|
||||
|
|||||||||||
|
|
|
|||||||||
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
-109- |
|
|
|
|
|
H2O |
OH |
|
- |
|
|
||||
|
|
|||
H2O Cr |
|
OH |
|
Na+ |
|
|
|||
O |
O |
|
||
|
|
|||
S
OO
6
6 . 5 6
6 , 9 6 #*
6, 6 . " 6 6
6 .
0 , 7 6 6
, . 0 9 6
6 6, 6
6 6 6 6 6:
|
|
|
|
|
|
|
OH |
|
2+ |
|
|
|
|
|
|
|
|
|
2+ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
2- |
|
H2O |
|
Cr (H2O)4 |
SO 2- |
|||||
|
|
(H O) Cr |
|
SO |
+ |
|
|||||||||||||
|
|
|
|
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
4 |
||||||||
|
2 |
4 |
|
|
|
|
4 |
|
|
HO |
|
||||||||
|
|
|
|
|
|
|
H2O |
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OH |
|
|
|
|
4+ |
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
(H2O)4 |
|
Cr |
|
Cr |
|
(H2O)4 |
(SO42-)2 + 2H2O |
||||||
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
HO |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
- |
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
# ( – ) 6
6:
, – – . :
6:
-110-
